* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 9
Homoaromaticity wikipedia , lookup
Marcus theory wikipedia , lookup
Heat transfer physics wikipedia , lookup
Electron scattering wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Woodward–Hoffmann rules wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Aromaticity wikipedia , lookup
Atomic theory wikipedia , lookup
Atomic orbital wikipedia , lookup
Molecular orbital wikipedia , lookup
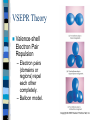
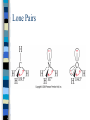
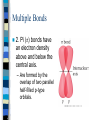
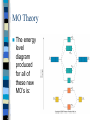
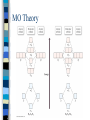
Chapter 10 Molecular Geometries and Bonding Theories – Lewis structures do not indicate the molecular architecture – the shape of the molecule. – The shape and structure of a molecule determines much of its physical and chemical characteristics. VSEPR Theory Valence-shell Electron Pair Repulsion – Electron pairs (domains or regions) repel each other completely. – Balloon model. Electron Regions 1. 2. 3. The number of electron regions around the central atom are counted as: Each single bond counts as a region. Each lone pair counts as a region. A multiple bond counts as a single region. Electron Regions How many? Electron Pair Geometry (EPG) Can be from two to six regions. Thus, only five EPG’s are possible. Two regions produces a linear EPG. Electron Pair Geometry Three regions produces a trigonal planar geometry. Planar = 2D. Ex) BF3 Electron Pair Geometry •Four regions becomes a three dimensional structure based on the tetrahedron. •Formally called tetrahedral with bond angles of 109.5o Electron Pair Geometry Tetrahedral is very common and symmetrical. An example is CF4 Electron Pair Geometry Five regions produces a trigonal bipyramidal geometry with two sets of bond angles. Electron Pair Geometry An example is PCl5 Electron Pair Geometry Six regions produces an octahedral geometry. Electron Pair Geometry An example is SF6 Molecular Geometry (MG) This is based on the shape of the electron pairs. When a molecule has no lone pairs, the EPG = MG. If the molecules DOES have one or more lone pairs, then the shape of the atoms is determined based off of the EPG. Molecular Geometry Molecular Geometry Molecular Geometry Examples Bent (120), SO2 Trigonal pyramidal, NH3 Bent (109.5), H2O Seesaw, SF4 T-shaped, ClF3 Linear, I3Square pyramidal, BrF5 Square planar, XeF4 Sketching the Molecules Simple = Ball and Stick figures Representing the 3D shapes: – Put as many of the molecules in the same plane as possible including the central atom. Use straight lines for bonds connected to atoms in plane. – For atoms in front of the plane, use a solid wedge. – For atoms behind the plane, use a hashed wedge. 3D Representations Lone Pairs A non-bonding pair will always take up more space. This compresses the normal bond angles. Lone Pairs Lone Pairs This also explains the MG’s for the trigonal bipyramidal family. Shapes of Larger Molecules A molecule like acetic acid has three central atoms. Shapes of Larger Molecules Polarity A molecule can contain very polar bonds, but can be non-polar. An example is CO2. Polarity On the other hand, sometimes polar bonds DO make a molecule polar. An example of a polar molecule is H2O. Polarity Polarity A molecule with a symmetrical distribution of polar bonds will be nonpolar. A molecule with an un-symmetrical distribution of polar bonds will be polar. – presence of lone pairs – different external atoms Polarity Polarity Polar molecules are attracted to other polar molecules Because water is a polar molecule, other polar molecules dissolve well in water – and ionic compounds as well Non-polar molecules do NOT dissolve in water. Valence Bond Theory How can we explain the formation of the bonds in a molecular compound? A bond occurs when a valence orbital on one atom overlaps with a valence orbital of another atom. Valence Bond Theory The H2 molecule – a closer look. nuclear repulsion no interaction minimum energy Valence Bond Theory Three (or more) atom molecules cannot be explained by simple overlap of orbitals. Fact: a bond generally forms between two half-filled orbitals. Fact: an s-type orbital is spherical, so it could form a bond in any direction. Fact: the three p-type orbitals are at 90 degree angles to each other. Valence Bond Theory CH4 – has an EPG and MG of tetrahedral with bond angles of 109.5o. Valence diagram for C and H before any bonding is: Valence Bond Theory Solution: promote the paired electron from the s orbital to the empty p orbital. Solution: mix the one s and three p orbitals together to get a new set of four orbitals all equal in energy. This is called _____________________. Valence Bond Theory Each hybrid orbital has some s and some p characteristics. Thus, they look different! Types of Hybrids Determined from the EPG. EPG Atomic orbitals Linear s+p = sp Trigonal s+p+p = planar sp2 Tetrahedral s+p+p+p = sp3 Hybrid diagram Examples BeF2 BF3 CH4 Types of Hybrids Atoms in the third period and beyond have empty d orbitals that can potentially be used for hybridization. PCl5 – requires five bonds, so need a set of five orbitals. Once again, must first promote the s electron to an empty d orbital. Types of Hybrids EPG Atomic orbitals Hybrid diagram Examples Trigonal bipyramidal s+p+p+p+d = sp3d PCl5 Octahedral s+p+p+p+d+d = sp3d2 SF6 Molecules with Lone Pairs Ex) NH3 Ex) H2O Ex) BrF3 Multiple Bonds Two types of bonds are possible. 1. Sigma (s) bonds have a cylindrical shape of electron density along the central axis between the two nuclei. s bond Multiple Bonds 2. Pi (p) bonds have an electron density above and below the central axis. – Are formed by the overlap of two parallel half-filled p-type orbitals. Multiple Bonds The majority of bonds are sigma bonds. When a double bond is present, the first bond is a sigma and the second is a pi. Pi bonds Multiple Bonds For any pi bonds, you MUST use an unhybridized half-full p-type orbital. Ex) C2H4 Ex) CO2 Multiple Bonds Pi Bond Significance Sigma bonds have free-rotation about the central axis. Ex) C2H4Cl2 Pi bonds have NO free-rotation due to the fact that they must overlap above and below the central axis. Ex) C2H2Cl2 Pi Bond Significance Isomers When two compounds share the exact same formula but are different either structurally or spatially, then they are said to be isomers. Structural isomers – C5H12 – C2H6O Isomers Geometric isomers are different spatially. This can occur for our carbon-carbon double bond. X Y Y Y C=C C=C Y X X X Trans Cis Isomers The last molecule in your packet has three possible structures. One is structural and two are geometric isomers. One other geometry can have cis/trans isomerism – is it tetrahedral or square planar? Ex) CH2Cl2 or Pt(NH3)2Cl2? Limitations of V.B. Theory Valence Bond Theory does not adequately explain molecules with resonance structures nor some other observed properties. Ex) O2 or molecular oxygen is paramagnetic (unpaired electrons). Lewis structure for O2 Molecular Orbital Theory A more sophisticated and complex model of bonding. Atomic orbitals from each atom contribute to new MO’s. Like atomic orbitals, each MO can hold up to two electrons. A MO, though, is spread out over the entire molecule. MO Theory For each atomic orbital contributed we get one MO. Half of the MO’s become bonding and the other half become anti-bonding. – Waveforms add either constructively or destructively like light! For the n=1 period, each atom contributes a 1s atomic orbital. MO Theory MO Theory MO Theory The Bond Order in MO theory is found by: BO = ½ (Bonding e- - Anti-bonding e-). Any bond order = 0 implies that the molecule is not possible. Odd number of electrons will produce half-integer BO’s. MO Theory Period 2 elements have both the 2s and 2p atomic orbitals to contribute towards MO’s. Thus, two atoms from period 2 will have how many atomic orbitals total? How many MO bonding orbitals will be produced? Anti-bonding? MO Theory The two 2s orbitals overlap just like the two 1s orbitals did in period 1. This produces the s2s and s2s* MO’s. The six 2p orbitals overlap differently. – Two will overlap end-on-end and produce a s2p type MO. – Four will overlap sideways and produce two p MO’s that are equal in energy. MO Theory The energy level diagram produced for all of these new MO’s is: MO Theory Diagram assumes that no 2s-2p orbitals interactions occur. For B2 , C2 , and N2 the interactions cause the s2p and p2p order to trade places on the diagram. Since these are filled for O2 , F2 , and Ne2 the diagram can be written with those two always reversed to simplify. MO Theory MO Theory It is possible for some molecules and ions to be paramagnetic – one or more unpaired electrons. Most will be diamagnetic – all paired electrons. n=2 Diatomic Molecules n=2 Diatomic Molecules Molecular Oxygen According to MO theory, it has a BO = 2 and it is paramagnetic! As liquid O2 is poured between the poles of a magnetic, it will have a strong attraction. Clip. Heteronuclear Diatomics For period 2, we can mix and match other elements and apply the same MO diagram. Simply add up the total valence electrons that each contribute and place in the diagram. For ions, a positive charge means we would decrease by one electron and a negative charge means we would increase by one electron. Heteronuclear Diatomics More electronegative element has lower energy orbitals. Can produce bond orders with ½ values. B.O. = ___________ Polyatomic Molecules When many atoms are combined together, the atomic orbitals of all the atoms are combined to make a set of molecular orbitals, which are delocalized over the entire molecule Gives results that better match real molecule properties than either Lewis or valence bond theories This is why resonance structures cannot be explained by valence bond theory. Ozone, O3 The structure of O3 includes two resonances. M.O. showing delocalized pi bonds