* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download met60-topic02
Future sea level wikipedia , lookup
Marine pollution wikipedia , lookup
Ocean acidification wikipedia , lookup
Blue carbon wikipedia , lookup
Arctic Ocean wikipedia , lookup
Anoxic event wikipedia , lookup
Effects of global warming on oceans wikipedia , lookup
Physical oceanography wikipedia , lookup
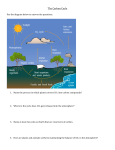
MET 60 Chapter 2: The “big picture” 9/1/09 MET 60 topic 02 1 • Read Chapter 2 ASAP. • The atmosphere is just one “sphere” on Earth. • There are several others. • They all interact! • Ultimately to understand climate, we need to understand all systems and interactions. 9/1/09 MET 60 topic 02 2 The systems… • The atmosphere. • The oceans (part of the hydrosphere): – Structure (thermal) – Content (salt) – Motions • The cryosphere – ice: – Polar regions – Greenland & Antarctica – Permafrost regions 9/1/09 MET 60 topic 02 3 • The biosphere – everything living (DNA-based): – Oceanic – Land-based • The “solid” earth: – Plate tectonics & paleoclimate – Volcanic gases – Carbon locked in (“sequestered” e.g., in rocks) 9/1/09 MET 60 topic 02 4 Various “cycles” are important for weather and climate, including: • The water cycle (2.2) • The carbon cycle (2.3) • Oxygen (2.4) Finally section 2.5 gives an overview of Earth’s climate history 9/1/09 MET 60 topic 02 5 The Oceans (part of the hydrosphere) • Cover 72% of planet – hence VIP • Enough water to cover planet with a layer 2.6 km deep • Salty! • On average 35 g of salts per kg of fresh water • Salt water is denser than fresh water – this matters because…see below… • Remember that fresh water is densest at 4C (hence – ice floats) 9/1/09 MET 60 topic 02 6 Vertical density structure… • Fig 2.2 • Maximum density gradient layer is called the thermocline (or pycnocline) – A density equivalent to an inversion – Inhibits vertical mixing (between upper and lower ocean) • Top layer of ocean is a mixed layer – Rain → salinity/density decrease – Evaporation → salinity/density increase 9/1/09 MET 60 topic 02 7 Water masses… • We talk of water masses, which retain their properties of temperature and salinity as they move. – A bit like air masses! • Use this idea to trace motions etc. in the oceans For the Atlantic… • Antarctic Bottom Water (AABW) – Very cold and dense (salty) – Formed as ice forms at the Antarctic and salt is expelled, leaving the surface water cold and dense – Eventually sinks → AABW 9/1/09 MET 60 topic 02 8 • North Atlantic Deep Water (NADW) – Also cold and dense – not quite as dense as AABW – Formed as ice forms around the Arctic • Mediterranean Outflow – Warm and very salty Fig 2.8 shows the arrangement of these masses: – – – – 9/1/09 AABW @ bottom (sinks down…) NADW above (sinks down…) “intermediate” water above Surface water above (wind-driven flows) MET 60 topic 02 9 Thermohaline circulation…Fig. 2.7 • http://en.wikipedia.org/wiki/Thermohaline_circulation • Circulation involving: – – – – sinking at high latitudes poleward drift at depth rising somewhere else return flow @ surface • Time involved? – Hundreds of years! 9/1/09 MET 60 topic 02 10 Wind-driven surface circulation…Fig 2.4 • Notice prevailing eastward flows in mid-latitude westerly regions • Notice ocean gyres Sea-surface temperatures…Fig 2.11 • SSTs • Pattern largely controlled by (net) radiative heating • At lower latitudes, surface circulations → warmer in western oceans, colder in eastern oceans • At higher latitudes, opposite (wind driven) 9/1/09 MET 60 topic 02 11 Sea-surface temperatures… • • • • • Upwelling is an important factor Due to the wind turning with depth (Ekman effect) Gives us in N. Cal. our cool water Cloud decks offshore also → cooling (Fig 1.7) http://www.ssec.wisc.edu/data/sst/latest_sst.gif • Often interested in SST anomalies, such as associated with El Nino • http://www.noaanews.noaa.gov/stories2009/20090709_elnin o.html 9/1/09 MET 60 topic 02 12 The Cryosphere • • Components – Table 2.1 & Fig. 2.12 – be aware of each one and its size (mass, area) What does the cryosphere do? 1. Provides thermal inertia to the climate system – Components warm/cool more slowly 2. Contributes to the albedo 3. Influences (partially drives) the thermohaline oceanic circulation – 4. 9/1/09 Freezing @ ice edges Impacts sea levels via storage MET 60 topic 02 13 • Over land, ice flows towards edges of ice cap – e.g., off Greenland – Fig. 2.13 – Greenland ice cap thickness → ice 100,000 years old @ bottom – Antarctic - 500,000 years old – Cores → information on past climate (see later) • Alpine glaciers – Smaller – Move faster – Shrinking rapidly! 9/1/09 MET 60 topic 02 14 Sea Ice • • Only 1-3 m thick Once ice starts to form, the ocean is insulated (very cold above), so additional ice forms more slowly • • • Not a solid layer! Floes and leads! Fig. 2.15 Heat exchange through the leads → complicated picture 9/1/09 MET 60 topic 02 15 Permafrost • • Global Climate Change effects are enhanced at high latitudes Hence – lots of interest now in permafrost • Fig. 2.17 – understand the curves! 9/1/09 MET 60 topic 02 16 The Biosphere Marine biosphere… • • • • • Top layer of ocean = euphotic zone = where light can penetrate = photosynthesis …provided nutrients are provided… E.g., via upwelling • Fig. 2.10 → active zones and biological deserts • Note the impact of El Nino! 9/1/09 MET 60 topic 02 17 Terrestrial biosphere… • • A biome is an area with a given climate that can support a give combination of animal and plant life Fig. 2.19 • • • • • • • Distribution depends on: Latitude (→ insolation) Annual average temperature (e.g., > or < 0C) Annual range of temperature (summer – winter) Diurnal range of temperature (day – night) Precipitation amount and distribution Cloudiness amount and distribution (_______________) 9/1/09 MET 60 topic 02 18 How does the terrestrial biosphere impact climate (and weather?) 1. Impact on hydrological cycle • Example: on hot days, plants transpire via evaporating water from leaves • Cooling of plants • Reduced heating of the soil as a result 2. Impact on albedo 3. Impact on surface roughness 9/1/09 MET 60 topic 02 19 The solid earth Make sure you know about plate tectonics and continental drift • Both play a role in climate • Plate tectonics → carbon sequestration • Continental drift → role in climate • Fig. 2.20 9/1/09 MET 60 topic 02 20 The hydrological cycle Spend time looking at Table 2.2 Huge amount in the mantle Expelled via volcanic eruptions Large amount in oceans Considerable amount in ice sheets (Greenland, Antarctica) Teeny amount in atmosphere! short residence time! 9/1/09 MET 60 topic 02 21 Two things of interest: 1. Precipitation (P) – Fig. 1.25 2. Evaporation (E) On the average globally: EP Where the overbar denotes an average (always!) Not true at every location/time! 9/1/09 Over the Sahara, E>P Along the ITCZ, P>E MET 60 topic 02 22 In a column of air somewhere: P E 9/1/09 MET 60 topic 02 23 And… Tr P Tr Tr E Tr is the water vapor flux or transport into/out of the column 9/1/09 MET 60 topic 02 24 Note that for land… E P atmosphere soil/rock T Where now T is transport into rivers, lakes, aquifers 9/1/09 MET 60 topic 02 25 The carbon cycle Imagine following a carbon atom through the carbon cycle: 9/1/09 MET 60 topic 02 26 9/1/09 MET 60 topic 02 27 Atmosphere: CO2 and CH4 Terrestrial biosphere: locked in plants via photosynthesis CO2 + H2O CH2O + O2 carbodydrate Oceans: dissolved CO2 in sea water (→carbonic acid, H2CO3) absorbed into ocean biota precipitated onto ocean floor (in dead stuff) 9/1/09 MET 60 topic 02 28 Oceans… precipitated onto ocean floor (in dead stuff) Solid earth: sedimentary rocks (from previous line!) in/into the mantle via subduction of plates released back into atmosphere in volcanic eruptions weathered from the surface soils and rocks fossil fuels 9/1/09 MET 60 topic 02 29 The carbon cycle Some processes go very fast (photosynthesis) Some go very slow (subduction etc.) See Table 2.3 & Fig. 2.23 9/1/09 MET 60 topic 02 30 Atmospheric carbon Most in CO2 (also a greenhouse gas) Well mixed (away from leaves) Amounts increasing (Fig. 1.3) Some in CH4 (also a greenhouse gas) Emitted from rice farming & livestock Emitted from natural gas (CH4) production Amounts increasing (Fig. xxx) 9/1/09 MET 60 topic 02 31 Biospheric carbon Terrestrial part…Photosynthesis… Plants take in sunlight (visible) + CO2 + H2O → “food” and O2 Upon decay, plants release CO2 and heat Fig. 2.24 shows where this is effective (surprising!) p.43: “if a large quantity of CO2 were injected into the atmosphere instantaneously…” – the biosphere could not quickly absorb it 9/1/09 MET 60 topic 02 32 Oceanic part…Photosynthesis… Marine biosphere absorbs carbon in euphotic zone Transports it downward as animals and plants die and sink 9/1/09 MET 60 topic 02 33 Oceanic (non-biosphere) carbon 1) 2) 3) Dissolved CO2 (carbonic acid, H2CO3) Carbonate ions (CO32-) Bicarbonate ions (HCO3-) – largest reservoir Bicarbonate is incorporated into shells etc. Carbon in the crust “Currently, the burning of fossil fuels is returning as much carbon to the atmosphere in a single year as weathering would return in hundreds of thousands of years.” – p.45 9/1/09 MET 60 topic 02 34 Oxygen Read the main text - basically on the accumulation of O2 in the atmosphere over time Oxygen (and other) isotopes What are they? Why do we care? Because isotope data are proxies for climate data: temperature and other quantities 9/1/09 MET 60 topic 02 35 Isotopes a) The Springfield baseball team (!) http://en.wikipedia.org/wiki/Springfield_(The_Simpsons) b) 9/1/09 atomic things (see (a)) MET 60 topic 02 36 Isotopes http://ie.lbl.gov/education/info.htm http://www.google.com/imgres?imgurl=http://www.emporia.edu/eart hsci/student/tinsley1/isotopes.gif&imgrefurl=http://www.empor ia.edu/earthsci/student/tinsley1/webpage1.html&h=142&w=350 &sz=408&tbnid=DBhGIF367Fj6mM:&tbnh=49&tbnw=120&p rev=/images%3Fq%3Disotopes%2Bof%2Boxygen&hl=en&usg =__7ylrVMpAzqQUpx2uYXb-7hKLv20=&ei=N7vSpGuGYXwsgOm0e24Cw&sa=X&oi=image_result&resnum= 7&ct=image http://www.nature.com/news/2009/090909/full/news.2009.901.html 9/1/09 MET 60 topic 02 37 Example: Oxygen-18 (18O) In cores of ocean sediments, 18O amounts are enhanced in times when surface ocean waters are cooler Thus we examine the ratio 18O/ 16O over time (carbon dating → time) 9/1/09 MET 60 topic 02 38 18O also changes in response to continental ice sheet volume. 16O preferentially evaporates (from the ocean) - leaving more 18O in the ocean If ice sheets grow (from snow enriched in 16O) then the ocean & sediments are enriched in 18O 9/1/09 MET 60 topic 02 39 Example: Carbon-13 (13C) Low amounts of 13C in organic deposits indicate high levels of CO2 at the time of plant growth Thus we examine the ratio 18O/ 16O over time (carbon dating → time) 18O 9/1/09 also changes in response to continental ice sheet volume MET 60 topic 02 40 No class Tuesday Furlough (all faculty) 9/1/09 MET 60 topic 02 41 Climate History Much is reconstructed from • • • 9/1/09 isotope data geology modeling! MET 60 topic 02 42 Something to remember: faint young sun luminosity has increased 30% since planet formed so??? we should be getting hotter!!! but… planet was NOT frozen most of the time “snowball earth”?? resolution? atmospheric greenhouse effect 9/1/09 MET 60 topic 02 43 Past 100 million years… timeline at http://www.seafriends.org.nz/books/geotime.htm Warmer before/cooler after “K-T boundary” iridium Glaciation began afterwards 9/1/09 MET 60 topic 02 44 Past 1 million years… Glacial and interglacial epochs we are in an interglacial Isotope data reveals links between temperatures and CO2 and CH4 levels (Fig. 2.31) LGM = “last glacial maximum” – about 20,000 ya - colder much lower sea levels (125m) much lower CO2 concentrations (180 ppmv) 9/1/09 MET 60 topic 02 45 Variations strongly linked to orbital variations • • • Obliquity (tilt…currently 23 ½ ) Eccentricity (small) Precession Fig. 2.34 9/1/09 MET 60 topic 02 46 Past 20,000 years… Interglacial Younger-Dryas (cold) event – see also paper in Asst 3 – see Fig. 2.35 Little Ice Age - 14th – 19th centuries 9/1/09 MET 60 topic 02 47