* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Lab 1 activity, AMINO ACIDS - Cal State LA
Survey
Document related concepts
Citric acid cycle wikipedia , lookup
Butyric acid wikipedia , lookup
Protein–protein interaction wikipedia , lookup
Western blot wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Two-hybrid screening wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Point mutation wikipedia , lookup
Metalloprotein wikipedia , lookup
Genetic code wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Biosynthesis wikipedia , lookup
Proteolysis wikipedia , lookup
Transcript
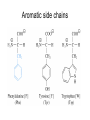
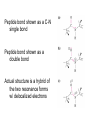
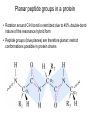
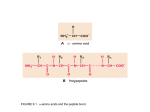
On a FREE AMINO ACID, what functional groups will accept or donate protons at pH 7, and hence are normally charged in water? side chain groups -OH -CH3 -C O -NH2 -COOH AA’s are zwitterions at pH 7, because they have oppositely charged groups the group “COOH” would be a very strong acid in fact, this group is always deprotonated, and thus exists as a weak base, COO- amino acids are salts with high melting points, because as they crystallize, ionic interactions stabilize the crystal lattice Titration curve for alanine pK1 = 2.4 pK2 = 9.9 pIAla = isoelectric point pKa values of amino acid ionizable groups Which amino acid side chains are positively charged at pH 7? Which amino acid side chains are negatively charged at pH 7? Which amino acid side chains can act as H-bond donors? Which amino acid side chains can act as H-bond acceptors? Aliphatic side chains Aromatic side chains Proline has a nitrogen in an aliphatic ring system • Proline (Pro, P) - has a three carbon side chain bonded to the a-amino nitrogen • The heterocyclic pyrrolidine ring restricts the geometry of polypeptides Methionine and cysteine Cys can participate in S-S bonds that covalently link different parts of a polypeptide chain to stabilize the 3D structure of a protein Side Chains with Alcohol Groups - can donate & accept H-bonds, but are NOT charged! - can be phosphorylated by kinase enzymes Acidic or polar side chains Basic amino acids Side chain of arginine Due to resonance, all lone pairs are partially occupied by the proton in its normal, positively charged state Peptide bond between two amino acids Peptide bond shown as a C-N single bond Peptide bond shown as a double bond Actual structure is a hybrid of the two resonance forms w/ delocalized electrons Planar peptide groups in a protein • Rotation around C-N bond is restricted due to 40% double-bond nature of the resonance hybrid form • Peptide groups (blue planes) are therefore planar; restrict conformations possible in protein chains features of the peptide backbone What parts of this polypeptide are charged at pH 7 ? What groups can accept H-bonds? What groups can donate H-bonds? [ Hint: what is different for a peptide versus a free amino acid? ] draw your amino acids as part of a peptide backbone, not as free aa’s, on your homework and quizzes!! Both backbone and side chain groups participate in important intra- and intermolecular binding interactions - some are responsible for folding protein into correct 2D and 3D shape - others bind to small molecules like enzyme cofactors & substrates Features of protein secondary structure (commonly encountered folding patterns) like b-sheets are due to repeated H-bonding between backbone groups of a protein