* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 2.3: Carbon Compounds
Microbial metabolism wikipedia , lookup
Drug discovery wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Isotopic labeling wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Proteolysis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Photosynthesis wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Metalloprotein wikipedia , lookup
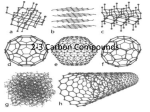
Chapter 2.3: Carbon Compounds Chemistry is. . . 1. What life is made of Example: Macromolecules 2. What life does a. b. c. d. Growth Reproduction Movement Interaction with the environment Chemistry of Carbon 1. Carbon can form four covalent bonds. 2. Carbon can bond with carbon, hydrogen, oxygen, nitrogen, phosphorus, and sulfur 6 protons 6 neutrons 6 electrons first shell- 2 second shell- 4 Bonding Symbols • A single bond is signified by a solid line between symbols; shares 2 electrons • A double bond is signified by two solid lines between symbols; shares 4 electrons • A triple bond is signified by three solid lines between symbols; shares 6 electrons Macromolecules 1. Macromolecules are “Giant molecules” 2. Consist of monomers (smaller units) that join together to form polymers. a. This process is called polymerization. Macromolecules • Synthesizing covalent bonds between the monomers involves losing a water molecule- dehydration, or condensation reaction Macromolecules • Breaking the covalent bond within the polymer to break off a monomer involves breaking a water molecule and inserting its pieces – hydrolysis Types of macromolecules 1. 2. 3. 4. Carbohydrates Lipids Nucleic Acids Proteins Carbohydrates • • • 1. Composition: made of C, H and O atoms *** monomer- monosaccharide 2. Uses: Main source of energy for organisms, structural purpose in cell membrane, and exoskeleton of insects. Carbohydrates 4. Examples a. Sugars 1. Monosaccharide = single sugar molecule 2. Polysaccharide = polymer of monosaccharides bonded together with a glycosidic linkage Carbohydrates 4. Examples b. Starch: Complex carbohydrates (polysaccharides) that store extra sugar 1. In animals, starch is called glycogen, structural is called chitin 2. In plants, “plant starch”, structural is called cellulose Lipids • • • • • 1.Composition: C, H, O atoms *** subunits are glycerol and fatty acids 2. Not soluble in water, hydrophobic 3. Function: energy storage, main part of cell membrane, hormones 4. Examples: fats, oils, waxes, steroids, cholesterol Lipids • • • • • Types of fatty acids: a. Unsaturated fatty acids are found in lipids that are liquid at room temperature, C=C bonds Example: Olive oil b. Saturated fatty acids are found in lipids that are solids at room temperature, no C=C bonds Example: Shortening, butter Types of Lipids Saturated = only single bonds, maximum number of H atoms Unsaturated = at least on C = C double bond lipid lipid Glycerol + fatty acid lipids + water Proteins 1. Contain nitrogen, carbon, hydrogen, oxygen 2. Uses: 1. 2. 3. 4. 5. 6. Structure Storage, transport of other substances, movement, Immunity Catalyze reactions (make them happen) Proteins 1. Made of monomers called amino acids a. 20 types of amino acids b. Same general structure, but different R group We will draw the 4 levels of Protein structure on the board… Amino group Carboxyl group General structure Alanine Serine Nucleic Acids 1. Contain hydrogen, oxygen, nitrogen, carbon, phosphorus 2. Store and transmit genetic information 3. Two types a. DNA (deoxyribonucleic acid) b. RNA (ribonucleic acid) Nucleotide 4. Made of monomers called nucleotides. A nucleotide has three parts a. 5-carbon sugar (ribose) b. Phosphate group c. Nitrogenous base Carbon Compounds include Carbohydrates Lipids Nucleic acids Proteins that consist of that consist of that consist of that consist of Glycerol and Fatty acids Nucleotides Amino Acids Monosaccharides which contain which contain Carbon, hydrogen, oxygen Carbon, hydrogen, oxygen Sugars and starches Fats, oils, waxes, steroids which contain which contain Carbon,hydrogen, oxygen, nitrogen, phosphorus Carbon, hydrogen,oxygen, nitrogen DNA or RNA Actin, enzymes, hemoglobin Chapter 2.4: Chemical Reactions and Enzymes Chemical Reactions Chemical Reaction: A change of one set of chemicals into another 1. Can be slow or fast 2. Chemical reactions require collisions between molecules 3. Involves changes in chemical bonds A+BC+D Chemical Reactions 1. Involves changes in chemical bonds a. Reactants are elements or compounds that enter into a chemical reaction. Bonds of reactants are broken in a chemical reaction. b. Products are elements or compounds that are produced in a chemical reaction. Bonds of products are formed in a chemical reaction Chemical Reactions 1. Involves changes in chemical bonds A+BC+D Which are the reactants? Which are the products? Chemical Reaction Example: CO2 in the body 1. Cells produce CO2,then blood carries CO2 from cells to lungs (exhale) Problem: CO2 is not soluble (dissolvable) in water Solution: A chemical reaction converts CO2 to a soluble compound In blood, CO2 converted to soluble compound: CO2 + H2O → H2CO3 In the lungs, reaction is reverse to exhale CO2 H2CO3 → CO2 + H2O Chemical reactions involve energy 1. Breaking and forming chemical bonds requires energy release or absorption 2. Reactions that release energy can occur spontaneously (but not all do) a. Energy is released as heat 3. Reactions that absorb energy will not occur without an energy source 4. What is activation energy? The energy needed to get a reaction started 5. Some chemical reactions are really slow or require lots of energy and cannot occur on their own Endergonic/ Endothermic Energy-Absorbing Reaction Exergonic/ Exothermic Energy-Releasing Reaction Activation energy Products Activation energy Reactants Reactants Products Enzymes are catalysts. 1. A catalyst is a substance that speeds up the rate of a chemical reaction by lowering the activation energy of the reaction. a. An enzyme is a protein that act as biological catalyst b. Enzymes speed up reactions that take place in cells Enzymes are catalysts. c. Enzymes provides a site (called the active site) where reactants can be brought together to react. This decreases the activation energy d. In a reaction involving an enzyme, the reactants are called substrates e. Enzymes can be reused, but can only have one type of substrate An enzymatic reaction: Enzymes are catalysts. f. The enzyme-substrate relationship is like that of a “lock and key” g. Enzymes can have an allosteric inhibitor in which another molecule can turn the enzyme on or off by binding to it (make the “lock” work or not) http://www.execulink.com/~ekimmel/mixed_flash.htm Enzymes are catalysts. h. Enzymes can be affected by: 1. pH 2. Temperature 3. Other proteins CO2 + H2O → H2CO3 requires an enzyme called carbonic anhydrase glucose + ATP → glucose-6-phosphate + ADP Enzyme (hexokinase) Glucose Substrates Products ADP Glucose-6phosphate Products are released ATP Active site Enzyme-substrate complex Substrates are converted into products Substrates bind to enzyme