* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Phar 722 Pharmacy Practice III
Microbial metabolism wikipedia , lookup
Butyric acid wikipedia , lookup
Photosynthesis wikipedia , lookup
Metalloprotein wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
Biochemistry wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Biosynthesis wikipedia , lookup
Plant nutrition wikipedia , lookup
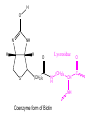
Phar 722 Pharmacy Practice III VitaminsBiotin Spring 2006 Biotin Study Guide • The applicable study guide items in the Vitamin Introduction • History • Structure of the vitamin and cofactor forms • Function of the cofactor including specific types of reactions catalyzed • Deficiency condition History • 1916 – It was recognized that rats fed a diet high in raw egg white exhibited muscle incoordination, dermatitis and loss of hair. • Cooked egg white was not toxic. – The toxicities from the raw egg white could be prevented by feeding liver or yeast to the animals. – Deficiencies can be induced by a diet restricted to raw egg white. • 1942 – The biotin structure was published. Biotin Chemistry • Biotin consists of two 5-membered rings cisfused to each other. • The d-isomer is the only active form. H O O N HN NH H H H Enolic form H H O O (CH2)4 S NH OH (CH2)4 S H Keto or ureido form OH Biotin Uptake and Metabolism • Little is known about biotin uptake. • There does not seem to be a cofactor form different from the basic structure. • The carboxyl chain forms an amide linkage with the ε-amino nitrogen of lysine which binds the vitamin to the enzyme's active site. • Biotin is required for the addition of carbon dioxide in many, but not all, carboxylation reactions. • There is some debate about the structure of the cofactor intermediate that transfers the carbon dioxide. See the next slide. H O N NH H H S Lys residue O (CH2)4 N H (CH2)4 CH NH Coenzyme form of Biotin O C Addition of CO2 to biotin for subsequent carboxylation reactions. O -O C O H Bicarbonate ATP H H ADP O O HO O P C Pi O H O C N C NH O O Phosphoric-carbonic acid anhydride S N -O H H - O O O NH H H O (CH2)4 Enz S O (CH2)4 Enz Biotin Function-1 • Methylmalonyl CoA from propionyl CoA Val, Ile, Met, Thr O CH3 CH2 CSCoA CO2 -O Propionyl CoA O CH3 O C CH C SCoA Methyl Malonyl CoA cobalamin O -O C O CH2 CH2 Succinyl CoA C SCoA Biotin Function-2 • Oxalacetate from pyruvate (gluconeogenesis; anapleurotic reactions) CH3 O O C C Pyruvate O CO2 O- -O C CH2 Oxalacetate O O C C O- Biotin Function-3 • Formation of carbamyl phosphate (urea cycle) O H2N C O Carbamyl Phosphate PO3 Biotin Function-4 • Malonyl CoA from acetyl CoA (fatty acid synthesis) CH3 C O CO2 O SCoA Acetyl CoA -O C O CH2 C Malonyl CoA SCoA Biotin Function-5 • Metabolism of leucine Biotin Deficiency • One of the ways to induce a deficiency in humans is to feed the volunteer a raw egg white diet. – Deficiency symptoms include hair loss, a rash around the nose and mouth, and conjunctivitis. • A good source of biotin is egg yolk. – Therefore, eating whole raw eggs will not cause a deficiency of this vitamin (although person could be at increased risk for salmonella poisoning). • What is the problem with raw egg white? – Egg white contains a basic protein known as avidin which forms salt linkages with the acidic biotin that might be in the intestinal tract (possibly produced by intestinal bacteria?). • This complex is not absorbed, but remains in the intestine eventually to be excreted. – Cooking the egg white ties up the avidin in the rest of the egg albumin preventing its binding biotin. Hypervitaminosis Biotin • None has been reported in humans. • There is no UL. Dosage Forms • The synthetic racemic mixture is used commercially. – As with racemic pantothenic acid and pantothenol, only half of the racemic biotin is active. • Biotin is considered one of the more expensive vitamins and can add to the cost of the final product. • Solubility 0.3 -0.4 mg/ml (1 gm/2,500-3,300 ml) DRIs • AI – – – – – – Infants Children (1 - 13 years) Adolescents (14 - 18 years) Adults Pregnancy Lactation • EAR – None reported • RDA – None reported • UL – None reported 5 - 6 μg/day 8 - 20 μg/day 25 μg/day 30 μg/day 30 μg/day 35 μg/day Sources • • • • Liver Kidney Yeast Animal and plant tissue in general; • Possibly the intestinal bacteria