* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
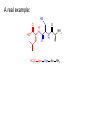
Download A real example: - McMaster University
List of types of proteins wikipedia , lookup
Nuclear magnetic resonance spectroscopy of proteins wikipedia , lookup
Protein structure prediction wikipedia , lookup
Homology modeling wikipedia , lookup
Alpha helix wikipedia , lookup
Degradomics wikipedia , lookup
Protein mass spectrometry wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
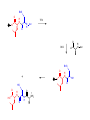
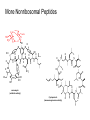
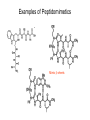
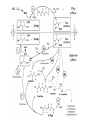
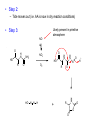
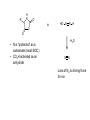
A real example: HS O HO O H N O HO2C Leu NH2 N H Cys Ala NH2 CH2Cl CH2 BnS O H N TFA N boc H O O O DCC -O BnS O + O O H N HO O NH O HS O H N N H + NH3 H N boc Natural Product Peptides, Peptidomimetics & Peptide Analogues • “Natural Product” Peptides (nonribosomal peptides) – Product of secondary metabolism – Synthesized on the NRPS – Numerous pharmaceutically relevant peptides: O O H2N O Ph O N H O O N H O HN H N H N O O N O O O N H NH O N N N H N O N N O O N O O N O Ph NH HN NH2 O O gramicidin S (antibiotic activity) N O Actinomycin C1 (apoptotic activity) O O More Nonribosomal Peptides HO HO OH HO H2N O O O O Cl O O HO O N H H O O O H N O N H OH O H N O N H H N O O N N N O O HN NH2 HO N O OH OH HO O HN O HN N OH O vancomycin (antibiotic activity) O N Cyclosporin A (immunosuppressive activity) N O HN N O O • Chemical synthesis demonstrated on solid support – – – – – Synthesis: weeks (soln) → days (solid) Employ more and/or different protecting groups Unusual functional groups Cyclization on resin? Other modifications (i.e. sugar moiety)? • Solid-supported synthesis has allowed the substitution and/or modification of AAs → analogues – – – – AA, functional groups, stereochemistry, substitution, etc Study structure-activity relationships Potential therapeutics Note: Industrial synthesis not performed on solid supported Peptide Analogues • Recently, there have been developments in the modification of peptides, particularly AMPs • AMPs = Antimicrobial Peptides – – – – – 15-30 AAs in length Produced by all animals (insects to frogs to humans) First line of defense against microbial organisms Answer to antibiotic resistance? Molecular diversity → dependent on structure AMP Structure • Large proportion of hydrophobic residues (~ 50 %) • Also contain varying amounts of Lys, Arg & His → +vely charged AAs – These AAs vary in their arrangement within the peptide • This arrangement of AAs allows disruption of bacterial membranes (anionic) “Teflon” Peptide: Fluorogainin-1 • Fluorous analogue of the AMP, magainin (isolated from the skin of frogs) – Replaced hydrophobic residues (i.e., Val, Leu,etc) with fluorinated versions → “Teflon like” – Resulted in more stable peptides: • More resistant to unfolding by chemical denaturants & heat • NMR also showed higher structural integrity – Results also indicated increased antimicrobial activity • Likely due to the increased hydrophobicity of peptide • This strong hydrophobic interaction may make the peptide less susceptible to proteases magainin series sites of fluorination: Leu 6, Ala 9, Gly 13, Val 17, and Ile 20 Other Analogues: NMR structure of magainin 2 Peptidomimetics • Peptide “mimics” – Contain non-natural peptidic structural elements (i.e. peptide bonds or unusual functional groups) – Molecules vary in size & structure – Commonly synthesized using Merrifield resin to study structureactivity relationships – Possible drug candidates Examples of Peptidomimetics Mimic -sheets Peptide Synthesis in the Prebiotic World Recall: • Murchison Meteorite – Possible source of AAs (via the Strecker mechanism) • Peptide (oligo) formation ? • Selection of an enantiomer – Selection by crystal faces – Circularly polarized light from stars • Enantioenrichment – Via Serine octamer – Enrichment by sublimation Peptide Synthesis in the Prebiotic World • Also recall: formation of peptides from monomers is energetically unfavorable (i.e., ΔG>0) – – – Modern world enzymes Chemical synthesis activation strategies Prebiotic world some energy input needed? Possibilities? 1) Synthesis with minerals! • Clay has been shown to catalyze the condensation of Gly to peptides up to (Gly)6 The experiment: • Uses SFM (scanning force microscopy) Faults (cracks) Apply gly to surface (at STP) Hectorite (layered silicate) containing Mg2+, Li+ & Cu2+ • No visible change in faults or layers • HPLC showed no gly peptides Experiment (con’t): Apply gly to surface Alternate cycles of heating to 90 °C + ddH2O Small glycine peptides (oligomers) HPLC Gly peptides of up to 6 AAs in length Other Similar Experiments: Gly montmorillonite or hectorite + Tyr Tyr-Tyr Gly-Gly 60 - 90 oC Gly-Gly-Tyr Varying the mineral can give different peptides! ...etc • Another experiment: – Mixed NaCl + Clay (mineral) + heat • NaCl alone gave only short peptides • When clay was added, longer peptides were produced! Hadean Beach – “the primary pump” 2) • This resembles many of the features of chemical peptide synthesis: • Step 1: In aqueous phase (i.e., ocean), 25 °C O O NH2 HO R • • H N C O + H H N HO R NH2 O Similar to Wohler synthesis of urea Amino group is now less reactive (amide-like) • Step 2: – Tide moves out (i.e. AA is now in dry reaction conditions) • Step 3: Likely present in primitive atmosphere NO O H N HO NH2 O NOx H N HO R O2 O HO N N H R + H N O N O H N R O O O + H H N R O O + HO N N H O • N is “protected” as a carbamate (recall BOC) • CO2H activated as an anhydride -H2O N N Loss of N2 is driving force for rxn • Step 4 & 5: Condensation O HN O R2 O R + H2N + H2N CO2H R2 R N H CO2H CO2 O Drives rxn • Experimentally, this system generates oligo-peptides with diastereoselection & preferred sequences (?) • May have given rise to earliest protein catalysts 3) Nucleic acid templated peptide synthesis: • • Template-- Model for the transfer of RNA world into the protein world? Basic idea: • Modify DNA strands with activated amino acids (i.e., DNAlinked substrate) • These DNA strands are specific in sequence in order to “tune” their hybridization abilities • DNA acts a template for further reactions, such as peptide bond formation • Reactions performed as “one pot” Nucleic Acid Template Synthesis • Step 1: – Templates are loaded with an AA – Attached to DNA as an N-hydroxysuccinimidyl ester (recall lab 6 → NHS & DCC) – Each AA (i.e. R1) has a unique DNA sequence associated with it • Step 2: – Masking of portion of template (i.e., “protect”) – Add other DNA-substrate molecules to the “pot” • Step 3: – Mixture is cooled to 4 °C (for 20 mins) & R1 template selectively hybridizes – Amine and activated carboxylate are now in close proximity & can undergo “intramolecular” peptide bond formation • Step 4: – Temperature raised, causing dissociation of template – DNA-R2 template hybridizes & peptide bond formation occurs • Cycle repeats for the third AA (R3) until tripeptide is obtained • Model demonstrates that DNA can resemble an enzyme (i.e., ribozyme) – Promotes coupling of 2 AAs through non-covalent interactions – Specificity (template sequence → one AA selected → tRNA like) • Could a similar model or sequence have given rise to peptides in the prebiotic world? • So far, we have looked at both amino acids & peptides (peptide bond formation) in the prebiotic & modern world • Common themes were: – Selectivity • Regioselectivity • Stereoselectivity • Protecting groups – Overcoming ΔG • Activation of carboxylate to make a peptide bond ( E of starting material) • Stabilization of TS ( E) (i.e., Lewis acid) – What about an active site? • Peptide → active site? • Peptides may fold and/or associate to produce a simple “active site” • Proteins/peptides have specific conformations due to intramolecular non-covalent forces: – – – – – H-bonding salt bridge Ionic Dipole-dipole Van der Waals • The sum of many weak forces → strong total binding force to restrict the conformation – Folding has a –ve ΔS, but a +ve ΔH • Also have covalent bonding: disulphide bridge