* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download 04 Reactions in Aqueous Solution
Transition state theory wikipedia , lookup
Citric acid cycle wikipedia , lookup
Chemical bond wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Double layer forces wikipedia , lookup
Artificial photosynthesis wikipedia , lookup
Click chemistry wikipedia , lookup
Chemical equilibrium wikipedia , lookup
Stoichiometry wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Coordination complex wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Oxidation state wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Liquid–liquid extraction wikipedia , lookup
Water splitting wikipedia , lookup
History of electrochemistry wikipedia , lookup
Biochemistry wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Chemical reaction wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Equilibrium chemistry wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Electrolysis of water wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Acid strength wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Electrochemistry wikipedia , lookup
Metalloprotein wikipedia , lookup
Ionic compound wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
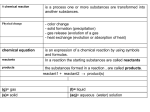
Lecture Presentation Chapter 4 Reactions in Aqueous Solution James F. Kirby Quinnipiac University Hamden, CT Aqueous Reactions © 2015 Pearson Education, Inc. LESSON 1 4-1 Aqueous Solution Properties Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. Chemical Reactions in Aqueous Solution Three Main Types of Chemical Reactions in Aqueous Solution 1. Precipitation reactions: Result in formation of an insoluble solid (precipitate) that separates from the solution. 2. Acid-base or neutralization reactions: Result in formation of water and an ionic compound (i.e., a salt) due to transfer of hydrogen ions (i.e., protons). 3. Oxidation-reduction (redox) reactions: Result in formation of new compounds due to transfer of electrons among reactants. Solution: a homogeneous mixture of 2 or more substances Aqueous solutions Solute: substance present in a smaller amount Solvent: substance present in a (much) larger amount A solution is a homogenous mixture of 2 or more substances The solute is(are) the substance(s) present in the smaller amount(s) The solvent is the substance present in the larger amount Solution Solvent Solute Soft drink (l) H2O Sugar, CO2 Air (g) N2 O2, Ar, CH4 Soft Solder (s) Pb Sn Solutions • Solutions are defined as homogeneous mixtures of two or more pure substances. • The solvent is present in greatest abundance. • All other substances are solutes. • When water is the solvent, the solution is called an aqueous solution. Aqueous Reactions © 2015 Pearson Education, Inc. Aqueous Solutions • Substances can dissolve in water by different ways: Ionic Compounds dissolve by dissociation, where water surrounds the separated ions. Molecular compounds interact with water, but most do NOT dissociate. Some molecular substances react with water when they dissolve. Aqueous Reactions © 2015 Pearson Education, Inc. NaCl (s) Na+ (aq) + Cl- (aq) H2O molecule Polar molecule Why is water such an effective solvent for ionic compounds? H d- Negative region (the O atom) d+ O Positive region (the H atom) H d+ Hydration is the process in which an ion is surrounded by water molecules arranged in a specific manner. d- d+ H2O H O O H H O H H Na+ H ClH O H Polar compounds: hydrogen bonding • Negative “pole” of water pointed toward H on -OH groups of polar molecule; positive “pole” of water pointed toward O on -OH group of polar molecule. • Clustering of H2O molecules around entire molecules keeps the molecules apart from each other. Ionic compounds: hydration • Negative “pole” of water pointed toward cation; positive “pole” of water pointed toward anion. • Clustering of H2O molecules around Na+ and Cl- ions keeps the ions apart from each other. H HO O CH2 H O H OH H H H OH O H O OH OH O H H Electrolytes and Nonelectrolytes • An electrolyte is a substance that dissociates into ions when dissolved in water. • A nonelectrolyte may dissolve in water, but it does not dissociate into ions when it does so. Aqueous Reactions © 2015 Pearson Education, Inc. Electrolytes • A strong electrolyte dissociates completely when dissolved in water. • A weak electrolyte only dissociates partially when dissolved in water. • A nonelectrolyte does NOT dissociate in water. Aqueous Reactions © 2015 Pearson Education, Inc. An electrolyte is a substance that, when dissolved in water, results in a solution that can conduct electricity. A nonelectrolyte is a substance that, when dissolved, results in a solution that does not conduct electricity. nonelectrolyte weak electrolyte strong electrolyte Substances that, when dissolved in H2O, produce solutions that can conduct electricity. A substance that, when dissolved in H2O, does not produces a solution that can conduct electricity. Conduct electricity in solution? Cations (+) and Anions (-) Strong Electrolyte – 100% dissociation NaCl (s) H 2O Na+ (aq) + Cl- (aq) Weak Electrolyte – not completely dissociated CH3COOH CH3COO- (aq) + H+ (aq) Strengths of Electrolytes: Complete, Partial, or No Ionization in Water STRONG ELECTROLYTES: Complete Ionization Hydrochloric acid + - H (aq) + Cl (aq) HCl (aq) NaC2H3O2 (aq) Sodium acetate (salts in general) + Na (aq) + C2H3O2 (aq) Strengths of Electrolytes: Complete, Partial, or No Ionization in Water WEAK ELECTROLYTES: Partial Ionization Acetic acid HC2H3O2 (aq) Reversible reaction: the reaction can proceed in both directions • Forward reaction occurs as fast as back reaction --- equilibrium NON-ELECTROLYTES: No Ionization Glucose C6H12O6 (aq) H+ (aq) + C2H3O2- (aq) Refer to Table 4.1, p. 107, Chang 7th ed. For a partial list of electrolytes and nonelectrolytes. ....... Nonelectrolyte does not conduct electricity? No cations (+) and anions (-) in solution C6H12O6 (s) H 2O C6H12O6 (aq) Strong Electrolyte Weak Electrolyte Nonelectrolyte HCl CH3COOH (NH2)2CO HNO3 HF CH3OH HClO4 HNO2 C2H5OH NaOH H2O C12H22O11 Ionic Compounds © 2015 Pearson Education, Inc. Which solution, NaCl(aq) or CH3OH(aq), conducts electricity? a. NaCl(aq) b. CH3OH(aq) © 2015 Pearson Education, Inc. Which solution, NaCl(aq) or CH3OH(aq), conducts electricity? a. NaCl(aq) b. CH3OH(aq) © 2015 Pearson Education, Inc. Hwk: page 156-163: 15, 17, 19 Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. LESSON 2 4-2 Precipitation Reactions Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. Precipitation Reactions When two solutions containing soluble salts are mixed, sometimes an insoluble salt will be produced. A salt “falls” out of solution, like snow out of the sky. This solid is called a precipitate. Aqueous Reactions © 2015 Pearson Education, Inc. Precipitation reactions: AgNO3 (aq) + NaCl (aq) AgCl (s) + NaNO3 (aq) • Formation of an insoluble product, the precipitate • Usually involve ionic compounds + NaCl (s) NaCl (aq) AgCl (s), NaNO3 (aq) AgNO3 (aq) Precipitate: insoluble solid that separates from the solution AgCl (s) Precipitation reactions and 1. Can we predict if a precipitate will form if we mix 2 solutions or add a compound to a solution? 2. If the answer to No. 1 above is “YES”, how do we do it? SOLUBILITY The maximum amount of solute that will dissolve in a given quantity of solvent at a specific temperature. Solubility Rules for Common Ionic Compounds (applies to water at 25oC) Li+, Na+, K+, Rb+, Cs+, NH4+, NO3-, C2H3O2-, ClO4-, also bicarbonate (HCO3-) and chlorate (ClO3-) Ag+, Hg22+, Pb2+ Ca2+, Sr2+, Ba2+, Hg22+, Pb2+, also silver (Ag+) CO32-, PO43-, C2O42-, also chromates (CrO42-) and sulfites (SO32-) S2-, OH- Solubility of Ionic Compounds • Not all ionic compounds dissolve in water. • A list of solubility rules is used to decide what combination of ions will dissolve. Aqueous Reactions © 2015 Pearson Education, Inc. Solubility Rules for Common Ionic Compounds in Water (at 25oC) Soluble Compounds Exceptions All compounds containing alkali metal ions (Li+, Na+, K+, Rb+, Cs+) and ammonium ion (NH4+) Nitrates (NO3-), acetates (C2H3O2-), bicarbonates (HCO3-), chlorates (ClO3-), perchlorates (ClO4-) Halides (Cl-, Br-, I-) Halides of silver (Ag+), mercury(I) (Hg22+), lead(II) (Pb2+) Sulfates (SO42-) Sulfates of Group 2 metal ions (Be2+, Mg2+, Ca2+, Sr2+, Ba2+), silver (Ag+), mercury(I) (Hg22+), lead(II) (Pb2+) Insoluble Compounds Exceptions Sulfides (S2-) Sulfides of Groups 1 and 2 metal ions (Li+, Na+, K+, Rb+, Cs+, Be2+, Mg2+, Ca2+, Sr2+, Ba2+) and NH4+ Hydroxides (OH-) Hydroxides of Group 1 metal ions (Li+, Na+, K+, Rb+, Cs+), Ba2+, and NH4+ Carbonates (CO32-), oxalates (C2O42-), phosphates (PO43-), chromates (CrO42-), sulfites (SO32-) Those of Group 1 metal ions (Li+, Na+, K+, Rb+, Cs+) and NH4+ Solubility Rules for Common Ionic Compounds In water at 250C Soluble Compounds Exceptions Compounds containing alkali metal ions and NH4+ NO3-, HCO3-, ClO3- Cl-, Br-, ISO4 2- Halides of Ag+, Hg22+, Pb2+ Sulfates of Ag+, Ca2+, Sr2+, Ba2+, Hg2+, Pb2+ Insoluble Compounds Exceptions CO32-, PO43-, CrO42-, S2- Compounds containing alkali metal ions and NH4+ OH- Compounds containing alkali metal ions and Ba2+ Precipitation Reactions Precipitate – insoluble solid that separates from solution precipitate Pb(NO3)2 (aq) + 2NaI (aq) PbI2 (s) + 2NaNO3 (aq) molecular equation Pb2+ + 2NO3- + 2Na+ + 2I- PbI2 (s) + 2Na+ + 2NO3- ionic equation Pb2+ + 2IPbI2 PbI2 (s) net ionic equation Na+ and NO3- are spectator ions Ways to Write Metathesis Reactions 1) Molecular equation 2) Complete ionic equation 3) Net ionic equation Aqueous Reactions © 2015 Pearson Education, Inc. Molecular Equation The molecular equation lists the reactants and products without indicating the ionic nature of the compounds. AgNO3(aq) + KCl(aq) AgCl(s) + KNO3(aq) Aqueous Reactions © 2015 Pearson Education, Inc. Complete Ionic Equation • In the complete ionic equation all strong electrolytes (strong acids, strong bases, and soluble ionic salts) are dissociated into their ions. • This more accurately reflects the species that are found in the reaction mixture. Ag+(aq) + NO3−(aq) + K+(aq) + Cl−(aq) AgCl(s) + K+(aq) + NO3−(aq) Aqueous Reactions © 2015 Pearson Education, Inc. Net Ionic Equation • To form the net ionic equation, cross out anything that does not change from the left side of the equation to the right. • The ions crossed out are called spectator ions, K+ and NO3−, in this example. • The remaining ions are the reactants that form the product—an insoluble salt in a precipitation reaction, as in this example. Ag+(aq) + NO3−(aq) + K+(aq) + Cl−(aq) AgCl(s) + K+(aq) + NO3−(aq) Aqueous Reactions © 2015 Pearson Education, Inc. Writing Net Ionic Equations 1. Write a balanced molecular equation. 2. Dissociate all strong electrolytes. 3. Cross out anything that remains unchanged from the left side to the right side of the equation. 4. Write the net ionic equation with the species that remain. Aqueous Reactions © 2015 Pearson Education, Inc. Molecular and Ionic Equations Spectator ion Spectator ion Spectator ions: Ions that are not involved in the overall reaction Molecular and Ionic Equations Recap: 1. Molecular equation • Formulas of all compounds involved are written as though all species existed as whole units. • Doesn’t describe accurately what’s going on at the microscopic level. 2. (Complete)Ionic equation • Shows dissolved species as free ions. • Spectator ions--not involved in overall reaction. 3. Net ionic equation • Shows only the species that take part in the reaction. Molecular and Ionic Equations How to write ionic and net ionic equations: 1. Write a balanced molecular equation for the reaction. 2 AgNO3 (aq) + K2CrO4 (aq) Ag2CrO4 (s) + 2 KNO3 (aq) 2. Rewrite the equation to show the dissociated ions that form in solution. • Strong electrolytes ionize into cations and anions when dissolved in solution. 2 Ag+ (aq) + 2 NO3- (aq) + 2 K+ (aq) + CrO42- (aq) Ag2CrO4 (s) + 2 K+ (aq) + 2 NO3- (aq) 3. Identify and cancel spectator ions on both sides of the equation. Result: the net ionic equation. 2 Ag+ (aq) + CrO42- (aq) Ag2CrO4 (s) (net ionic equation) Molecular and Ionic Equations How to write ionic and net ionic equations: 1. Write a balanced molecular equation for the reaction. Pb(NO3)2 (aq) +2 NaI (aq) PbI2 (s) + 2 NaNO3 (aq) 2. Rewrite the equation to show the dissociated ions that form in solution. • Strong electrolytes ionize into cations and anions when dissolved in solution. Pb2+ (aq) + 2 NO3- (aq) + 2 Na+ (aq) + 2 I- (aq) PbI2 (s) + 2 Na+ (aq) + 2 NO3- (aq) 3. Identify and cancel spectator ions on both sides of the equation. Result: the net ionic equation. Pb2+ (aq) + 2 I- (aq) PbI2 (s) (net ionic equation) Molecular and Ionic Equations How to write ionic and net ionic equations: 1. Write a balanced molecular equation for the reaction. AgNO3 (aq) + KCl (aq) AgCl (s) + KNO3 (aq) 2. Rewrite the equation to show the dissociated ions that form in solution. • Strong electrolytes ionize into cations and anions when dissolved in solution. Ag+ (aq) + NO3- (aq) + K+ (aq) + Cl- (aq) AgCl (s) + K+ (aq) + NO3- (aq) 3. Identify and cancel spectator ions on both sides of the equation. Result: the net ionic equation. Ag+ (aq) + Cl- (aq) AgCl (s) (net ionic equation) Writing Net Ionic Equations 1. Write the balanced molecular equation. 2. Write the ionic equation showing the strong electrolytes 3. Determine precipitate from solubility rules 4. Cancel the spectator ions on both sides of the ionic equation © 2015 Pearson Education, Inc. Which ions remain in solution after PbI2 precipitation is complete? a. K+ and I– b. Pb2+ and I– c. K+ and NO3– d. Pb2+ and NO3– © 2015 Pearson Education, Inc. Which ions remain in solution after PbI2 precipitation is complete? a. K+ and I– b. Pb2+ and I– c. K+ and NO3– d. Pb2+ and NO3– © 2015 Pearson Education, Inc. Hwk: page 156-163: 21, 23, 25, 27, 29, 93, 107 Quiz to Follow Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. LESSON 3 4-3 Acid Base Reactions Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. Acids Have a sour taste. Vinegar owes its taste to acetic acid. Citrus fruits contain citric acid. React with certain metals to produce hydrogen gas. React with carbonates and bicarbonates to produce carbon dioxide gas Bases Have a bitter taste. Feel slippery. Many soaps contain bases. 4.3 General Properties of Acids and Bases Acids • Sour taste • Cause color changes in plant dyes (change litmus from blue to red) • React with certain metals (e.g., Zn, Mg, Fe) to produce hydrogen gas: Mg (s) + 2HCl (aq) MgCl2 (aq) + H2 (g) • React with carbonates (CO32-) & bicarbonates (HCO3-) to produce carbon dioxide (CO2) gas: 2HCl(aq) + CaCO3(s) CaCl2(aq) + H2O(l) + CO2(g) HCl(aq) + NaHCO3(s) NaCl(aq) + H2O(l) + CO2(g) • Aqueous acid solutions conduct electricity Bases • Bitter taste • Feel slippery • Cause color changes in plant dyes (change litmus from red to blue) • Aqueous base solutions conduct electricity Acids and Bases Around the House… Acids • Aspirin (acetylsalicylic acid) • Orange juice (citric acid) • Vinegar (dilute solution of acetic acid) • Muriatic acid (concentrated hydrochloric acid) Bases • Ammonia (concentrated ammonium hydroxide) • Washing soda (sodium carbonate) • Baking soda (sodium bicarbonate) • Drain cleaner (sodium & potassium hydroxides) Basic Compounds in Antacids 54 Acids and Metals Acids react with metals • such as K, Na, Ca, Mg, Al, Zn, Fe, and Sn • to produce hydrogen gas and the salt of the metal Molecular equations: 2K(s) + 2HCl(aq) 2KCl(aq) + H2(g) metal acid salt hydrogen gas Zn(s) + 2HCl(aq) ZnCl2(aq) + H2(g) metal acid salt hydrogen gas 55 Learning Check Write a balanced equation for the reaction of magnesium metal with HCl(aq). Label the metal, acid, and salt. 56 Learning Check Write a balanced equation for the reaction of magnesium metal with HCl(aq). Mg(s) + 2HCl(aq) MgCl2(aq) + H2(g) metal acid salt 57 Gas-Forming Reactions Some metathesis reactions do not give the product expected. When a carbonate or bicarbonate reacts with an acid, the products are a salt, carbon dioxide, and water. CaCO3(s) + 2 HCl(aq) CaCl2(aq) + CO2(g) + H2O(l) NaHCO3(aq) + HBr(aq) NaBr(aq) + CO2(g) + H2O(l) Aqueous Reactions © 2015 Pearson Education, Inc. Acids and Carbonates Acids react • with carbonates and hydrogen carbonates • to produce carbon dioxide gas, a salt, and water 2HCl(aq) + CaCO3(s) CO2(g) + CaCl2(aq) + H2O(l) acid carbonate carbon salt water dioxide HCl(aq) + NaHCO3(s) CO2(g) + NaCl (aq) + H2O(l) acid bicarbonate carbon salt water dioxide 59 Learning Check Write a balanced equation for the following reactions: A. MgCO3(s) + HBr(aq) B. HCl(aq) + NaHCO3(aq) 60 Solution Write a balanced equation for the following reactions: A. MgCO3(s) + 2HBr(aq) MgBr2(aq) + CO2(g) + H2O(l) carbonate acid salt carbon water dioxide B. HCl(aq) + NaHCO3(aq) NaCl(aq) + CO2(g) + H2O(l) acid bicarbonate salt carbon water dioxide 61 Neutralization Reactions In a neutralization reaction, • an acid reacts with a base to produce salt and water • the acid HCl reacts with NaOH to produce salt and water • the salt formed is the anion from the acid and cation of the base HCl(aq) + NaOH(aq) NaCl(aq) + H2O(l) acid base salt water 62 Neutralization Reactions In neutralization reactions, • if we write the strong acid and strong base as ions, we see that H+ reacts with OH− to form water, leaving the ions Na+ and Cl- in solution: H+(aq) + Cl-(aq) + Na+(aq) + OH-(aq) Na+(aq) + Cl-(aq) + H2O(l) • the overall reaction is H3O+ from the acid and OH- from the base form water: H+(aq) + OH-(aq) H2O(l) 63 Acid-base reactions: HCl (aq) + NaOH (aq) H2O (aq) + NaCl (aq) 0.2 M NaOH (aq) 0.2 M HCl (aq) H2O (l), NaCl (aq) Acid-base reactions: Neutralization reactions • Acid and base essentially cancel each other out (neutralize each other) to produce water and a salt: HCl (aq) + NaOH (aq) H2O (l) + NaCl (aq) (acid) (base) (water) (salt) NH4OH (aq) + HCl (aq) H2O (l) + NH4Cl (aq) (base) (acid) (water) (salt) Acid-Base Neutralization Reactions H2O acid + base salt + water An ionic compound made up of a cation other than H+ and an anion other than OHor O2- H+ donor H+ acceptor Acid-Base Reactions In an acid–base reaction, the acid (H2O above) donates a proton (H+) to the base (NH3 above). Reactions between an acid and a base are called neutralization reactions. When the base is a metal hydroxide, water and a salt (an ionic compound) are produced. Aqueous Reactions © 2015 Pearson Education, Inc. Neutralization Reactions When a strong acid (like HCl) reacts with a strong base (like NaOH), the net ionic equation is circled below: HCl(aq) + NaOH(aq) NaCl(aq) + H2O(l) H+(aq) + Cl−(aq) + Na+(aq) + OH−(aq) Na+(aq) + Cl−(aq) + H2O(l) H+(aq) + OH−(aq) H2O(l) Aqueous Reactions © 2015 Pearson Education, Inc. Guide for Balancing Neutralization Reactions 69 Balancing Neutralization Reactions Write the balanced equation for the neutralization of magnesium hydroxide and nitric acid. Step 1 Write the reactants and products. Mg(OH)2 + HNO3 Step 2 Balance the H+ in the acid with the OH- in the base. Mg(OH)2 + 2HNO3 Step 3 Balance the H2O with H+ and the OH-. Mg(OH)2 + 2HNO3 salt + 2H2O Step 4 Write the salt from the remaining ions. Mg(OH)2 + 2HNO3 Mg(NO3)2 + 2H2O 70 Learning Check Select the correct group of coefficients for each of the following neutralization equations. 1. HCl(aq) + Al(OH)3(aq) AlCl3(aq) + H2O(l) A. 1, 3, 3, 1 B. 3, 1, 1, 1 C. 3, 1, 1, 3 2. Ba(OH)2(aq) + H3PO4(aq) Ba3(PO4)2(s) + H2O(l) A. 3, 2, 2, 2 B. 3, 2, 1, 6 C. 2, 3, 1, 6 71 Solution 1. HCl(aq) + Al(OH)3(aq) AlCl3(aq) + H2O(l) Step 1 Write the reactants and products. HCl + Al(OH)3 Step 2 Balance the H+ in the acid with the OH- in the base. 3HCl + Al(OH)3 Step 3 Balance the H2O with H+ and the OH-. 3HCl + Al(OH)3 salt + 3H2O Step 4 Write the salt from the remaining ions. 3HCl(aq) + Al(OH)3(aq) AlCl3(aq) + 3H2O(l) The answer is C. 3, 1, 1, 3. 72 Solution 2. Ba(OH)2(aq) + H3PO4(aq) Ba3(PO4)2(s) + H2O(l) Step 1 Write the reactants and products. Ba(OH)2 + H3PO4 Step 2 Balance the H+ in the acid with the OH- in the base. 3Ba(OH)2 + 2H3PO4 Step 3 Balance the H2O with H+ and the OH-. 3Ba(OH)2 + 2H3PO4 salt + 6H2O Step 4 Write the salt from the remaining ions. 3Ba(OH)2(aq) + 2H3PO4(aq) Ba3(PO4)2(s) + 6H2O(l) The answer is B. 3, 2, 1, 6. 73 Learning Check Write the neutralization reactions for stomach acid, HCl, and the ingredients in Mylanta. Mylanta: Al(OH)3 and Mg(OH)2 74 Solution Write the neutralization reactions for stomach acid, HCl, and the ingredients in Mylanta. Mylanta: For Al(OH)3: Step 1 Write the reactants and products. Al(OH)3 + HCl Step 2 Balance the H+ in the acid with the OH- in the base. Al(OH)3 + 3HCl Step 3 Balance the H2O with H+ and the OH-. Al(OH)3 + 3HCl salt + 3H2O Step 4 Write the salt from the remaining ions. Al(OH)3(aq) + 3HCl(aq) AlCl3(aq) + 3H2O(l) 75 Solution Write the neutralization reactions for stomach acid, HCl, and the ingredients in Mylanta. Mylanta: For Mg(OH)2: Step 1 Write the reactants and products. Mg(OH)2 + HCl Step 2 Balance the H+ in the acid with the OH- in the base. Mg(OH)2 + 2HCl Step 3 Balance the H2O with H+ and the OH-. Mg(OH)2 + 2HCl salt + 2H2O Step 4 Write the salt from the remaining ions. 2HCl(aq) + Mg(OH)2(aq) MgCl2(aq) + 2H2O(l) 76 Sample Problem 8.8 Balancing Equations of Acids Write the balanced equation for the neutralization of HCl(aq) and Ba(OH)2(s). Solution Step 1 Write the reactants and products. HCl(aq) + Ba(OH)2(s) → salt + H2O(l) Step 2 Balance the H+ in the acid with the OH– in the base. Placing a coefficient of 2 in front of HCl provides 2H+ for the 2OH– in Ba(OH)2. 2HCl(aq) + Ba(OH)2(s) → salt + H2O(l) Step 3 Balance the H2O with the H+ and the OH–. Use a coefficient of 2 in front of H2O to balance 2H+ and 2OH–. 2HCl(aq) + Ba(OH)2(s) → salt + 2H2O(l) Step 4 Write the salt from the remaining ions. Use the ions Ba2+ and 2Cl– to write the formula of the BaCl2. salt, 2HCl(aq) + Ba(OH)2(s) → BaCl2(aq) + 2H2O(l) Study Check 8.8 Write the balanced equation for the reaction between H2SO4(aq) and NaHCO3(aq). Arrhenius acid is a substance that produces H+ (H3O+) in water Arrhenius base is a substance that produces OH- in water 4.3 Acid-Base Definitions: 1. Arrhenius acids & bases: Formulated by the Swedish chemist Svante Arrhenius (1859-1927) in the late 19th century, to classify substances whose properties in aqueous solution were well known. • Acids: Substances that ionize in water to produce H+ ions. HCl (aq) H+ (aq) + Cl- (aq) • Bases: Substances that ionize in water to produce OH- ions. NaOH (aq) Na+ (aq) + OH- (aq) A Brønsted acid is a proton donor A Brønsted base is a proton acceptor base acid acid base A Brønsted acid must contain at least one ionizable proton! 4.3 Acid-Base Definitions: 2. Bronsted-Lowry acids & bases: Formulated simultaneously by the Danish chemist Johannes Bronsted (1879-1947), and the British chemist Thomas Lowry (1874-1936), in 1923 to cover substances dissolved in water and other solvents (e.g., alcohols). • Acids: Substances that donate H+ ions (a.k.a. protons). HC2H3O2 (aq) = H+ (aq) + C2H3O2- (aq) (acetic acid) (acetate anion) • Bases: Substances that accept H+ ions. C2H3O2- (aq) + H2O (l) = HC2H3O2 (aq) + OH- (aq) (acetate anion) (acetic acid) Acids • The Swedish physicist and chemist S. A. Arrhenius defined acids as substances that increase the concentration of H+ when dissolved in water. • Both the Danish chemist J. N. Brønsted and the British chemist T. M. Lowry defined them as proton donors. Aqueous Reactions © 2015 Pearson Education, Inc. Bases • Arrhenius defined bases as substances that increase the concentration of OH− when dissolved in water. • Brønsted and Lowry defined them as proton acceptors. Aqueous Reactions © 2015 Pearson Education, Inc. Bronsted-Lowry Acids & Bases HC2H3O2 (aq) = H+ (aq) + C2H3O2- (aq) (acetic acid) (proton) should be written as… (acetate anion) Can’t exist alone in solution due to its strong attraction to the negative pole (the O atom) in H2O. Consequently, the proton is coordinated to H2O molecules (i.e., hydrated) in aqueous solution. HC2H3O2(aq) + H2O(l) = H3O+(aq) + C2H3O2-(aq) (acetic acid) (hydronium ion) Chemists write the hydrogen ion as “H+” in acid-base reactions as a shorthand, in which the presence of water as solvent is understood. (acetate anion) Bronsted-Lowry Acids & Bases Monoprotic Acids: Those acids which, upon ionization in H2O, donate only one proton. Examples: HCl (aq) H+ (aq) + Cl- (aq) HCl is a strong electrolyte--ionizes completely in H2O. HC2H3O2 (aq) = H+ (aq) + C2H3O2- (aq) HC2H3O2 is a weak electrolyte--ionizes partially in H2O. Bronsted-Lowry Acids & Bases Polyprotic Acids: Those acids which, upon ionization in H2O, donate more than one proton. Example: Sulfuric acid (H2SO4) -- a diprotic acid (donates 2 H+ upon ionization in H2O) H2SO4 (aq) H+ (aq) + HSO4- (aq) HSO4- (aq) = H+ (aq) + SO42- (aq) H2SO4 is a strong electrolyte, ionizes completely into H+ and HSO4-, as represented by the single arrow. HSO4- is a weak electrolyte, ionizes partially into H+ and SO42-, as represented by the double arrow. Example: Phosphoric acid (H3PO4) -- a triprotic acid (donates 3 H+ upon ionization in H2O) H3PO4 (aq) = H+ (aq) + H2PO4- (aq) H2PO4- (aq) = H+ (aq) + HPO42- (aq) HPO42- (aq) = H+ (aq) + PO43- (aq) Monoprotic acids HCl H+ + Cl- HNO3 H+ + NO3H+ + CH3COO- CH3COOH Strong electrolyte, strong acid Strong electrolyte, strong acid Weak electrolyte, weak acid Diprotic acids H2SO4 H+ + HSO4- Strong electrolyte, strong acid HSO4- H+ + SO42- Weak electrolyte, weak acid Triprotic acids H3PO4 H2PO4HPO42- H+ + H2PO4H+ + HPO42H+ + PO43- Weak electrolyte, weak acid Weak electrolyte, weak acid Weak electrolyte, weak acid 4.3 STRONG ACIDS Ionize completely…see list HCl H+ + Cl- Strong or Weak? • Strong acids completely dissociate in water; weak acids only partially dissociate. • Strong bases dissociate to metal cations and hydroxide anions in water; weak bases only partially react to produce hydroxide anions. Aqueous Reactions © 2015 Pearson Education, Inc. Weak Acid => an acid that only partially dissociates to H+ ions in water. ex) HF(aq) ↔ H+ (aq) + F- (aq) note the double arrow indicates that the reaction does not go to completion Bronsted-Lowry Acids & Bases Monobases: Those bases which, in H2O, can accept only one proton. Example: Hydroxide ion (OH-) H+ (aq) + OH- (aq) H2O (l) Example: Ammonia (NH3) NH3 (aq) + H+ (aq) = NH4+ (aq) Polybases: Those bases which, in H2O, can accept more than one proton. Example: Phosphate anion (PO43- ) -- a tribase (can accept 3 H+) PO43- (aq) + H+ (aq) = HPO42- (aq) HPO42- (aq) + H+ (aq) = H2PO4- (aq) H2PO4- (aq) + H+ (aq) = H3PO4 (aq) In H2O, weak Bronsted bases undergo a hydrolysis reaction. This term means that the weak Bronsted base reacts with H2O to form an acid and hydroxide ion (OH-). NH3 (aq) + H2O (l) = NH4+ (aq) + OH- (aq) PO43- (aq) + H2O (l) = HPO42- (aq) + OH- (aq) HPO42- (aq) + H2O (l) = H2PO4- (aq) + OH- (aq) H2PO4- (aq) + H2O (l) = H3PO4 (aq) + OH- (aq) Strong Bronsted bases ionize completely in H2O. NaOH (aq) Na+ (aq) + OH- (aq) Ba(OH)2 (aq) Ba2+ (aq) + 2OH- (aq) Weak Bases: a base that is only partially dissociated to form OH- ions in water. ex) NH3(aq) + H2O(l) ↔ NH4 + (aq) + OH- Neutralization Reaction 1) Strong Acid – Strong Base Reaction acid + base salt + water All salts are strong electrolytes. HCl (aq) + NaOH (aq) H+ + Cl- + Na+ + OH- H+ + OH- NaCl (aq) + H2O Na+ + Cl- + H2O H2O 2) Weak Acid – Strong Base Two Step Reaction (1) HB(aq) ↔ H+ (aq) + B- (aq) (2) H+ (aq) + OH- (aq) → HOH (3) Net Ionic: HB(aq) + OH- (aq) → B- (aq) + HOH ex) HF(aq) + OH- (aq) → HOH + F- (aq) 3) Strong Acid – Weak Base Two Step Reaction (1) B + HOH → BH+ (aq) + OH- (aq) (2) H+ (aq) + OH- (aq) → HOH (3) Net Ionic: H+ (aq) + B(aq) → ex) H+ (aq) + NH3(aq) → NH4 + (aq) BH + (aq) Hwk: page 156-163: 31, 33, 35, 37, 39, 41 Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. LESSON 4 4-4 Oxidation-Reduction Reactions Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. Oxidation-reduction (redox) reactions: H2O2 (aq) + 2KI (aq) + 2HCl (aq) I2 (aq) + 2KCl (aq) I2 (aq) + 2Na2S2O3 (aq) 2NaI (aq) + Na2S4O6 (aq) + + Na2S2O3 (aq) + KI (aq) 1M HCl (aq) 0.1 M H2 O2 (aq) I2 (aq), KCl (aq), NaI (aq), Na2S4O6 (aq) Oxidation-Reduction Reactions 2 Na0 2 Na+ + 2 eLoss of electrons from Na metal to form Na+ cations OXIDATION - 2 e- Cl2 (g) + 2 Na (s) 2 NaCl (s) + 2 e- Cl20 + 2 e- 2 ClGain of electrons from Cl2 gas to form Cl- anions REDUCTION Oxidation-Reduction Reactions 2 Na0 2 Na+ + 2 e- OXIDATION HALF-REACTION Cl20 + 2 e- 2 Cl- REDUCTION HALF-REACTION 0 Cl2 + 2 Na0 2 Na+ and Clcombine to form NaCl Na+ + 2 Cl- Add the half-reactions… Electrons cancel each other Cl2 (g) + 2 Na (s) 2 NaCl (s) OVERALL EQUATION: SUM OF THE HALF-REACTIONS O I L xidation s oss (of electrons) R I G eduction s ain (of electrons) Oxidation-Reduction Reactions • • • • Loss of electrons is oxidation. Gain of electrons is reduction. One cannot occur without the other. The reactions are often called redox reactions. How many electrons does each oxygen atom gain during the course of this reaction? a. None b. One c. Two d. Four How many electrons does each oxygen atom gain during the course of this reaction? a. None b. One c. Two d. Four Identifying Redox Reactions Reducing Agent • • • • Causes reduction Loses one or more electrons Undergoes oxidation Oxidation number of atom increases Oxidizing Agent • • • • Causes oxidation Gains one or more electrons Undergoes reduction Oxidation number of atom decreases e- eCl Cl Na OXIDIZING AGENT Accepts e- from Na, causes Na to be oxidized to Na+ + e- + e- Na REDUCING AGENT Donates e- to Cl2, causes Cl to be reduced to Cl- -e- - e- Na+ Cl- Cl- Na+ Oxidation Number • a.k.a. oxidation state --- the number of charges an atom would have in a molecule or an ionic compound if electrons were transferred completely. oxidation numbers 0 Formation of an ionic compound: 0 +1 -1 Cl2 (g) + 2 Na (s) 2 NaCl (s) oxidation numbers Formation of a molecular compound: 0 0 +5 -2 P4 (s) + 5 O2 (g) P4O10 (s) Oxidation Number Seven rules for assigning oxidation numbers: 1. In FREE elements (uncombined e.g. H2, P4, S8, Ag, Au, Fe), each atom has an oxidation number of ZERO. 2. For ions containing only one atom (monatomic ions), the oxidation number is equal to the charge on the ion. Examples familiar to you: Alkali metals (Group 1) – ox. # is +1. Alkaline earth metals (Group 2) – ox. # is +2. Aluminum ion – ox. # is +3 (all its compounds) 3. The oxidation number of oxygen in most compounds (e.g. MgO and H2O) is –2. However, in H2O2 (hydrogen peroxide) and O22- (peroxide anion), the ox. # is –1. 4. The oxidation number of hydrogen is +1, EXCEPT when it is bonded to metals in binary compounds (e.g. LiH and CaH2). Oxidation Number Seven rules for assigning oxidation numbers: 5. Fluorine has an oxidation number of –1 in ALL its compounds. Other halogens (Cl, Br, I) have NEGATIVE oxidation numbers when they occur as HALIDE ions in their compounds. When combined with oxygen (e.g. in ClO4-, HClO4), the halogens have POSITIVE oxidation numbers. 6. In a NEUTRAL molecule or ionic compound, the SUM of the oxidation numbers of ALL the atoms must equal ZERO. In a POLYATOMIC ION, the sum of oxidation numbers of all the elements in the ion must equal the NET CHARGE ON THE ION. 7. Oxidation numbers do not have to be integers. Example: ox. # of O in the superoxide ion, O2-,is –1/2. IT’S A WISE IDEA TO REFER TO THE OXIDATION TABLE I GAVE YOU AND BECOME FAMILIAR WITH THE OXIDATION NUMBERS OF THE VARIOUS ELEMENTS, ESPECIALLY THE COMMON OXIDATION NUMBERS. Oxidation Number Additional information: 1. Metallic elements have only positive oxidation numbers. Nonmetallic elements, however, may have either positive or negative oxidation numbers. 2. The HIGHEST oxidation number an element in Groups 1A through 7A can have is its group number. The halogens, for example, can possess an oxidation number of +7. 3. The transition metals (Groups 1B through 8B) can possess several oxidation numbers. Oxidation Numbers To determine if an oxidation–reduction reaction has occurred, we assign an oxidation number to each element in a neutral compound or charged entity. Rules to Assign Oxidation Numbers • Elements in their elemental form have an oxidation number of zero. • The oxidation number of a monatomic ion is the same as its charge. Rules to Assign Oxidation Numbers • Nonmetals tend to have negative oxidation numbers, although some are positive in certain compounds or ions. – Oxygen has an oxidation number of −2, except in the peroxide ion, in which it has an oxidation number of −1. – Hydrogen is −1 when bonded to a metal, +1 when bonded to a nonmetal. Rules to Assign Oxidation Numbers • Nonmetals tend to have negative oxidation numbers, although some are positive in certain compounds or ions. – Fluorine always has an oxidation number of −1. – The other halogens have an oxidation number of −1 when they are negative; they can have positive oxidation numbers, most notably in oxyanions. Rules to Assign Oxidation Numbers • The sum of the oxidation numbers in a neutral compound is zero. • The sum of the oxidation numbers in a polyatomic ion is the charge on the ion. Oxide vs Peroxide Oxidation Number Assign oxidation numbers to all the elements in the following compounds and ions: S4O62- KMnO4 Fe (IO4)2 Fe(CN)64- PtCl62- NF3 Oxidation Number Consider the reaction of aqueous copper (II) sulfate with zinc metal: CuSO4 (aq) + Zn (s) Cu (s) + ZnSO4 (aq) The products are Cu metal and zinc sulfate. Which reactant is oxidized? Zn0 Zn2+ + 2e- Which reactant is reduced? Cu2+ + 2e- Cu0 Oxidation number of Cu2+ ions is +2 Overall Reaction: Oxidation number of Cu metal is 0 Cu2+ (aq) + Zn0 (s) Cu0 (s) + Zn2+ (aq) Identifying Redox Reactions Oxidizing Agent reduction 0 4Fe(s) + 3O2(g) 0 Reducing Agent Copyright © 2008 Pearson Prentice Hall, 2Fe2 O3 (s) +3 oxidation Chapter 4/142 -2 Identifying Redox Reactions Oxidizing Agent reduction +3 0 2Fe2O3 (s) + 3 C (s) 4Fe(s) + 3 C O2 (g) 0 Reducing Agent Copyright © 2008 Pearson Prentice Hall, +4 oxidation Chapter 4/143 Ex) Zn + 2 HCl ZnCl2 + H2 Identify what species is oxidized and reduced? Identify oxidizing and reducing agents. Ex) 2Al + 3Cl2 2AlCl3 Identify what species is oxidized and reduced? Identify oxidizing and reducing agents. Ex) CH4 + 2O2 CO2 + 2H2O Identify what species is oxidized and reduced? Identify oxidizing and reducing agents. Ex) PbO + CO Pb + CO2 Identify what species is oxidized and reduced? Identify oxidizing and reducing agents. Displacement Reactions In displacement reactions, ions oxidize an element. In this reaction, silver ions oxidize copper metal: Cu(s) + 2 Ag+(aq) Cu2+(aq) + 2 Ag(s) The reverse reaction does NOT occur. Why not? Why does the solution in Fig. 4.14 turn blue? a. Cu2+ (aq) ions are blue. b. NO3– (aq) ions are blue. c. Ag(s) is blue. d. H2O is blue. Why does the solution in Fig. 4.14 turn blue? a. Cu2+ (aq) ions are blue. b. NO3– (aq) ions are blue. c. Ag(s) is blue. d. H2O is blue. Activity Series • Elements higher on the activity series are more reactive. • They are more likely to exist as ions. Metal/Acid Displacement Reactions • The elements above hydrogen will react with acids to produce hydrogen gas. • The metal is oxidized to a cation. How many moles of hydrogen gas would be produced for every mole of magnesium added into the HCl solution? a. None b. One c. Two d. Four How many moles of hydrogen gas would be produced for every mole of magnesium added into the HCl solution? a. None b. One c. Two d. Four Hwk: page 156-163: 45, 49, 51, 53 Quiz to Follow Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. LESSON 5 4-5 Concentration of Solutions Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. grams of solute = M . V . (molar mass) Molarity • The quantity of solute in a solution can matter to a chemist. • We call the amount dissolved its concentration. • Molarity is one way to measure the concentration of a solution: Molarity (M) = moles of solute volume of solution in liters Aqueous Reactions © 2015 Pearson Education, Inc. Mixing a Solution • To create a solution of a known molarity, weigh out a known mass (and, therefore, number of moles) of the solute. • Then add solute to a volumetric flask, and add solvent to the line on the neck of the flask. Aqueous Reactions © 2015 Pearson Education, Inc. Dilution Formula M1. V1 = M2 . V2 moles of solute before dilution moles of solute after dilution Dilution • One can also dilute a more concentrated solution by – using a pipet to deliver a volume of the solution to a new volumetric flask, and – adding solvent to the line on the neck of the new flask. Aqueous Reactions © 2015 Pearson Education, Inc. Dilution The molarity of the new solution can be determined from the equation Mc Vc = Md Vd, where Mc and Md are the molarity of the concentrated and dilute solutions, respectively, and Vc and Vd are the volumes of the two solutions. Aqueous Reactions © 2015 Pearson Education, Inc. What is the concentration of a solution made by dissolving 0.2500 g of K2CrO4 (potassium chromate) in 500.0 mL of distilled H2O? What is the concentration of a new solution made by dilution of 10.00 mL of the above K2CrO4 solution to 1000.0 mL with distilled H2O? How many grams of K2CrO4 are contained in the new solution? Hwk: page 156-163: 60, 62, 63, 65, 66, 67, 69, 73, 75, 77 Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. LESSON 6 4-6 Solution Stoichiometry Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. Using Molarities in Stoichiometric Calculations Aqueous Reactions © 2015 Pearson Education, Inc. Titration A titration is an analytical technique in which one can calculate the concentration of a solute in a solution. Aqueous Reactions © 2015 Pearson Education, Inc. © 2015 Pearson Education, Inc. How would the volume of standard solution added change if that solution were Ba(OH)2(aq) instead of NaOH(aq)? a. Increase by one-half the volume used for titration with NaOH. b. Increase by two the volume used for titration with NaOH. c. Decrease by two the volume used for titration with NaOH. d. Decrease by one-half the volume used for titration with NaOH. © 2015 Pearson Education, Inc. How would the volume of standard solution added change if that solution were Ba(OH)2(aq) instead of NaOH(aq)? a. Increase by one-half the volume used for titration with NaOH. b. Increase by two the volume used for titration with NaOH. c. Decrease by two the volume used for titration with NaOH. d. Decrease by one-half the volume used for titration with NaOH. © 2015 Pearson Education, Inc. Titration • A solution of known concentration, called a standard solution, is used to determine the unknown concentration of another solution. • The reaction is complete at the equivalence Aqueous point. Reactions © 2015 Pearson Education, Inc. Titration of 0.200 M NaOH with 0.200 M HCL Start: 5.00 mL of 0.200 M NaOH diluted to 50.0 mL with distilled H2 O Indicator: Bromothymol blue G B Y pH 6.0 pH 7.6 pH 7.0 pH 12.30 0.0 mL HCl added pH 7.00 5.0 mL HCl added pH 3.14 5.2 mL HCl added See handout Hwk: page 156-163: 79, 81, 83, 85, 87, Atoms, Molecules, and Ions © 2015 Pearson Education, Inc. Review Questions Chapter 4 Stoichiometry © 2015 Pearson Education, Inc. A homogeneous mixture of two or more components is referred to as a. b. c. d. a solute. a solution. an electrolyte. a mess. © 2015 Pearson Education, Inc. A homogeneous mixture of two or more components is referred to as a. b. c. d. a solute. a solution. an electrolyte. a mess. © 2015 Pearson Education, Inc. The solvent in a sample of soda pop is a. b. c. d. sugar. carbon dioxide. water. air. © 2015 Pearson Education, Inc. The solvent in a sample of soda pop is a. b. c. d. sugar. carbon dioxide. water. air. © 2015 Pearson Education, Inc. The gaseous solute in a sample of soda pop is a. b. c. d. sugar. carbon dioxide. water. air. © 2015 Pearson Education, Inc. The gaseous solute in a sample of soda pop is a. b. c. d. sugar. carbon dioxide. water. air. © 2015 Pearson Education, Inc. Gatorade® and other sports drinks conduct electricity because they contain a. b. c. d. water. sugar. air. electrolytes. © 2015 Pearson Education, Inc. Gatorade® and other sports drinks conduct electricity because they contain a. b. c. d. water. sugar. air. electrolytes. © 2015 Pearson Education, Inc. When Fe(NO3)2 dissolves in water, the particles in solution are a. b. c. d. Fe+ and (NO3)2–. Fe2+ and 2 NO3–. Fe and 2 NO3. Fe and N2 and 3 O2. © 2015 Pearson Education, Inc. When Fe(NO3)2 dissolves in water, the particles in solution are a. b. c. d. Fe+ and (NO3)2–. Fe2+ and 2 NO3–. Fe and 2 NO3. Fe and N2 and 3 O2. © 2015 Pearson Education, Inc. Which set includes only substances that produce electrolytes in water? a. b. c. d. NaBr, KCl, MgSO4 C6H12O6, CH3OH, C6H6 HCl, NH3, Cl2, N2 SiO2, CaCO3, H2SO4 © 2015 Pearson Education, Inc. Which set includes only substances that produce electrolytes in water? a. b. c. d. NaBr, KCl, MgSO4 C6H12O6, CH3OH, C6H6 HCl, NH3, Cl2, N2 SiO2, CaCO3, H2SO4 © 2015 Pearson Education, Inc. Which compound below is not soluble in water? a. b. c. d. NaBr KNO3 MgSO4 ZnS © 2015 Pearson Education, Inc. Which compound below is not soluble in water? a. b. c. d. NaBr KNO3 MgSO4 ZnS © 2015 Pearson Education, Inc. Pb(NO3)2 + 2 KI PbI2 + 2 KNO3 The physical evidence that the above reaction occurs is a. b. c. d. an explosion. the formation of a gas. that the solution boils. the formation of a precipitate. © 2015 Pearson Education, Inc. Pb(NO3)2 + 2 KI PbI2 + 2 KNO3 The physical evidence that the above reaction occurs is a. b. c. d. an explosion. the formation of a gas. that the solution boils. the formation of a precipitate. © 2015 Pearson Education, Inc. Which pair of compounds will produce a precipitate if solutions of appropriate concentrations are mixed together? a. b. c. d. H2SO4 and NaOH HNO3 and CaCl2 Ba(NO3)2 and Na3PO4 LiCl and SrI2 © 2015 Pearson Education, Inc. Which pair of compounds will produce a precipitate if solutions of appropriate concentrations are mixed together? a. b. c. d. H2SO4 and NaOH HNO3 and CaCl2 Ba(NO3)2 and Na3PO4 LiCl and SrI2 © 2015 Pearson Education, Inc. When an acid reacts with a base, the result is a. b. c. d. cancellation. elimination. neutralization. adduct formation. © 2015 Pearson Education, Inc. When an acid reacts with a base, the result is a. b. c. d. cancellation. elimination. neutralization. adduct formation. © 2015 Pearson Education, Inc. When nitric acid is neutralized by potassium hydroxide, the spectator ions are a. b. c. d. K+ and NO3–. H+ and OH–. H+ and NO3–. K+ and OH–. © 2015 Pearson Education, Inc. When nitric acid is neutralized by potassium hydroxide, the spectator ions are a. b. c. d. K+ and NO3–. H+ and OH–. H+ and NO3–. K+ and OH–. © 2015 Pearson Education, Inc. Which compound below is not a strong acid? a. b. c. d. HC2H3O2 H2SO4 HNO3 HBr © 2015 Pearson Education, Inc. Which compound below is not a strong acid? a. b. c. d. HC2H3O2 H2SO4 HNO3 HBr © 2015 Pearson Education, Inc. When an atom undergoes oxidation, it ______ electrons. a. b. c. d. gains loses retains balances © 2015 Pearson Education, Inc. When an atom undergoes oxidation, it ______ electrons. a. b. c. d. gains loses retains balances © 2015 Pearson Education, Inc. When an atom undergoes reduction, it ______ electrons. a. b. c. d. gains loses retains balances © 2015 Pearson Education, Inc. When an atom undergoes reduction, it ______ electrons. a. b. c. d. gains loses retains balances © 2015 Pearson Education, Inc. When Zn(s) reacts with HCl(aq) to produce H2(g) and ZnCl2(aq), the zinc is _____ because it _____ electrons. a. b. c. d. reduced; gains reduced; loses oxidized; gains oxidized; loses © 2015 Pearson Education, Inc. When Zn(s) reacts with HCl(aq) to produce H2(g) and ZnCl2(aq), the zinc is _____ because it _____ electrons. a. b. c. d. reduced; gains reduced; loses oxidized; gains oxidized; loses © 2015 Pearson Education, Inc. Al + H+ Al3+ + H2 When the oxidation–reduction reaction above is correctly balanced, the coefficients are a. b. c. d. 1, 2 1, 1. 1, 3 1, 2. 2, 3 2, 3. 2, 6 2, 3. © 2015 Pearson Education, Inc. Al + H+ Al3+ + H2 When the oxidation–reduction reaction above is correctly balanced, the coefficients are a. b. c. d. 1, 2 1, 1. 1, 3 1, 2. 2, 3 2, 3. 2, 6 2, 3. © 2015 Pearson Education, Inc. In the list shown, the metal that is most easily oxidized is ___. a. b. c. d. Li Ca Fe Cu © 2015 Pearson Education, Inc. In the list shown, the metal that is most easily oxidized is ___. a. b. c. d. Li Ca Fe Cu © 2015 Pearson Education, Inc. A solution is prepared by dissolving 35.0 g of NaCl in water to make 500 mL of solution. What is the molarity? a. b. c. d. 7.00 M 3.04 M 1.97 M 1.20 M © 2015 Pearson Education, Inc. A solution is prepared by dissolving 35.0 g of NaCl in water to make 500 mL of solution. What is the molarity? a. b. c. d. 7.00 M 3.04 M 1.97 M 1.20 M © 2015 Pearson Education, Inc. 250.0 mL of 0.100 M AgNO3 solution contains _____ g of silver nitrate. a. b. c. d. 4.25 8.50 17.0 34.0 © 2015 Pearson Education, Inc. 250.0 mL of 0.100 M AgNO3 solution contains _____ g of silver nitrate. a. b. c. d. 4.25 8.50 17.0 34.0 © 2015 Pearson Education, Inc. To make 250.0 mL of 0.500 M KI solution, _____ mL of 6.00 M KI must be used. a. b. c. d. 20.8 41.7 500.0 3000.0 © 2015 Pearson Education, Inc. To make 250.0 mL of 0.500 M KI solution, _____ mL of 6.00 M KI must be used. a. b. c. d. 20.8 41.7 500.0 3000.0 © 2015 Pearson Education, Inc. HCl + KOH KCl + H2O 25.00 mL of HCl was titrated using 18.50 mL of 0.1554 M NaOH. The concentration of HCl is a. b. c. d. 0.0777 M. 0.1150 M. 0.2100 M. 0.2875 M. © 2015 Pearson Education, Inc. HCl + KOH KCl + H2O 25.00 mL of HCl was titrated using 18.50 mL of 0.1554 M NaOH. The concentration of HCl is a. b. c. d. 0.0777 M. 0.1150 M. 0.2100 M. 0.2875 M. © 2015 Pearson Education, Inc. HX + NaOH NaX + H2O 229 mg of HX was titrated using 29.33 mL of 0.0965 M NaOH. What is element X? a. b. c. d. F Cl Br I © 2015 Pearson Education, Inc. HX + NaOH NaX + H2O 229 mg of HX was titrated using 29.33 mL of 0.0965 M NaOH. What is element X? a. b. c. d. F Cl Br I © 2015 Pearson Education, Inc.