* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download document
Polysubstance dependence wikipedia , lookup
Psychedelic therapy wikipedia , lookup
Orphan drug wikipedia , lookup
Compounding wikipedia , lookup
Drug design wikipedia , lookup
Drug discovery wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Theralizumab wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Pharmacognosy wikipedia , lookup
Neuropharmacology wikipedia , lookup
Prescription costs wikipedia , lookup
Psychopharmacology wikipedia , lookup
Adverse Drug Reactions
Objectives
To make health
professionals and
public more aware of
adverse drug
reactions.
To explore the
importance of
monitoring and
reporting ADRs.
2
Topics
• Introduction & definitions of adverse
drug reactions (ADR )
• Most common reasons of ADR
• Prevention & Risk factors for ADR
• General Classification of ADR
• Examples of ADRs
• Conclusion
• Resources
3
Introduction
The safety of medicines is an essential
part of patient safety.
√Definition ( WHO) of Adverse drug
reaction (ADR) : Any noxious*,
unintended, undesired effect of a drug
.
which occurs at doses used for
prophylaxis, diagnosis, or therapy.
pharmacovigilance : Preventing and detecting
adverse effects from medicines يقظة, حذر,احتراس
4
Many drugs has been withdrawn
• Fenfluramine anti-obesity medication
• introduced in 1973 ; withdrawn in 1997
• Why ? reports of heart valve disease, and pulmonary
hypertension, including a condition known as cardiac
fibrosis.
• cerivastatin. Treatment of hypelipdeimeia
• Introduced in 1990s, withdrawn in 2001
• Why ? , 52 deaths were reported from
rhabdomyolysis and its resultant renal failure
• The story of thalidomide is will known
5
• Rofecoxib {Vioxx }
• (NSAID) approved for tttt of
osteoarthritis, acute pain conditions, and
dysmenorrhoea.
• Introduced in 1999, withdrawn in 2004
• Why ? because of concerns about
increased risk of heart attack and stroke
associated with long-term, high-dosage
use.
• More and more are left for you to explore ???
6
Serious ADR
?
The American FDA defines a serious adverse event
as that which can cause one or more of the
following :
•
•
•
•
•
•
Death
Life-Threatening event.
Hospitalization
Disability -.
Congenital Anomaly
Requires Intervention to Prevent
Permanent Impairment or Damage
7
Some common causes of
ADR√√
1. Wrong diagnosis;
2. In appropriate dosage regimen:.
3. Poor assessment of the patients .
4. Non compliance
5.
6.
7.
8.
Drug -drug interaction
Drug food or herbal medicine interaction
Self-medication
counterfeit medicines :
9.
expired medication
8
Risk factors for ADR
• Drug-related factors
–
–
–
–
Nature of the drug
Degree of exposure (dose, duration, frequency)
Route of administration
Cross-sensitization
• Host-related factors
– Age : (elderly & neonates)
– Sex : ( pregnancy )
– Genetic factors (HLA type, Acetylator status)
– Concurrent medical illness (e.g. viral infection, bronchial
asthma )
– Previous drug reaction
– Multiple allergy syndrome
9
Host realted factors e.g Viral infections
Ceratin veial infections make the patiants more
predisposed for ADR examples are :
• Acute EBV infections:
make the patients predisposed for maculopapular exanthem
with aminopenicillins
• HIV infections:
– Sulfonamides: MPE, SJS/TEN, DRESS
– SJS/TEN to various drugs is 500 fold more frequent
– Nevirapine and abacavir: frequent side effects
10
Host related factors
•
•
•
•
•
Renal impairment – failing excretion of drugs/active metabolites
Liver disease – failing drug metabolism
Some specific diseases – e.g Bronchial asthma
Neonates – drug metabolizing systems are not fully developed
History of allergies to some drugs –
Health staff efforts to minimize
ADR
[1] Avoid inappropriate drugs in the context of clinical condition
[2] Use right dose, route, frequency based on patient variables
[3] Elicit medication history; consider untoward incidents
[4] Elicit history of allergies [identify in patients with allergic diseases]
[5] Rule out drug interactions
[6] Adopt right technique: Eg slow iv injection of aminophylline
[7] Carry out appropriate monitoring [Eg PT with warfarin; Li levels]
8) Monitor , occurred & report ADR
12
√√√Classification of Adverse
effects of drugs
Type A: Augmented pharmacologic effects ,Dose related ,
Type B: Bizarre effects, non-dose related, unpredictable
Type C: Chronic effects : dose related & time-related
Type D: Delayed effects , e.g., time –related
Type E: End-of-use or withdrawal effects,
Type F : Failure of therapy
Type A effects (‘Augmented pharmacological actions’):
Dose-related
Related to a
pharmacological effect
Predictable.
Very Common .
Usually Low mortality
Can be minimized by
appropriate dosing
Include toxicity & Side
effects:
14
Type A effects (more details ’):
A- Mediated by same receptors & same sites as for
beneficial effects
hypoglycemia caused by insulin (anti-diabetic)
bleeding caused by warfarin (anti-coagulant)
• hypnotic effect caused by H2 R antagonist : antihistaminics
B-Mediated by same receptors but at different sites:
e.g., prazosin controls hypertension via blocking α1 receptors in
the peripheral blood vessels, but cause pupil constriction by
action on α1 receptors in the radial muscle of iris
C-Mediated by different types of receptors at different
sites:
e.g., propranolol relieves angina (β1 receptors in
heart), but can cause bronchoconstriction ( β2
receptors in the bronchi)
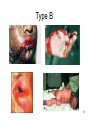
Type B: (‘Bizarre effects’):
Uncommon ,
Not related to pharmacological
action of the drug
Unpredictable
High mortality
No simple dose-response
relationship:
Bizzare = strange
Type B : more details
• I. Allergic reactions:
• Abnormal responses related to immune system
• Type I: Immediate (anaphylactic reaction with
penicillins)
• Type II: Cytolytic reactions ( haemolysis with αmethyldopa)
• Type III: Arthus reactions (serum sickness with
streptokinase)
• Type IV: Delayed allergic reaction (contact
dermatitis with penicillins)*
17
Idiosyncratic adverse drug
reactions
Idiosyncratic reactions:
May be related to genetic abnormality, e.g.
oHemolysis by primaquine in G6PD
deficiency
oApnea caused by succinylcholine in
patients with low plasma cholinestrase
oIncreased peripheral neuritis in slow
acetylators during isoniazid therapy
1I-
oMore examples
•Acute porphyria
•Malignant hyperthermia
•More examples
18
Type B
19
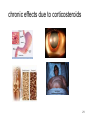
Type C: Chronic effect
Dose-related and time-related
Related to the cumulative dose
Uncommon
Typical example :
Hypothalamic-pituitary-adrenal axis suppression by
corticosteroids
We may also include :
•
Some Drug induced diseases, (Iatrogenic diseases )
a) peptic ulcer caused by NSAIDs;
b)
c)
d)
e)
Nephrotoxicity caused by ciclosporin , aminoglycosides
Hepatoxicity caused by valproic acid
Neurotoxicity caused by isoniazid
Reproductive toxicity: decreased sperm count in male and an-ovulation
in female caused by anti cancer drugs (alkylating agents)
f) Drug Dependence & addiction ?? .
chronic effects due to corticosteroids
21
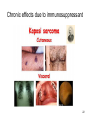
Chronic effects due to immunosuppressant
22
Type D: Delayed effect
Occurs or becomes apparent some time after the
use of the drug ( even after several years )
• Uncommon ●
• Typical example : , diethylstilbestrol increase incidence
of vaginal carcinoma in offspring ●
We may also consider the following as delayed drug
effect :
Carcinogenesis
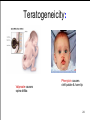
Teratogeneicity:
Teratogeneicity:
Valproate causes
spina-bifida
Phenytoin causes
cleft palate & hare-lip
24
Type E: Ending of use or
withdrawal effects
• Occurs soon when a drug is stopped abruptly
• Uncommon
• Typical examples :
– Opiate withdrawal syndrome
– Myocardial ischaemia (-blocker withdrawal)
– Acute adrenal insufficiency sudden stop of
corticosteroid
Failure of therapy
• Unexpected failure of therapy
• ● Dose-related particularly when used
Often caused by drug interactions
• Typical example :
– failure of contraceptive pills due to
concomitant use of strong enzyme inducer
such as anticonvulsant drugs.
• SDL : add more examples
26
Be also aware of ADR due to herbal
medicine
• OTC products & Herbal and traditional
medicines also have safety problems
allergic reactions, bronchospasm, dyspnoea,
urticaria, angina due to Echinacea
purpurea –
Australian Adverse Drug Reaction Bulletin, v.18, No.1,
1999:
27
Reporting & monitoring ADR
• Health professionals (physicians,
pharmacists, nurses, dentists and others)
are in the best position to report suspected
ADRs as part of their daily patient care.
• Health professionals should report ADRs
even if they are doubtful about the precise
relationship between the given medicine
and reaction
28
Conclusion
• ADR are common and can be dangerous
• Many ADR can be predicated and avoided .
• Special attention should be given to elderly,
neonates , pregnant women those suffering from chronic
disorders.
• Self medication and OTC, polypharmacy ,
noncompliance increase the potential of
incidence of ADR.
• Risk factors include : drug related &patient
related
29
More examples of ADR try to classify ??
• Abortion, miscarriage or uterine hemorrhage associated
with misoprostol (Cytotec),
• Addiction to many sedatives and analgesics such as
diazepam, morphine, etc.
• Birth defects associated with and Accutane.
• Bleeding of the intestine associated with aspirin therapy
• Deafness and kidney failure associated with gentamicin
• Liver damage from paracetamol overdose
• Melasma and thrombosis associated with use of
estrogen-containing hormonal contraception such as the
combined oral contraceptive pill
30
• Rhabdomyolysis associated with statins (anti-cholesterol
drugs)
• Seizures caused by withdrawal from benzodiazepine
• Drowsiness or increase in appetite due to antihistamine
use..
• Stroke or heart attack associated with sildenafil (Viagra)
when used with nitroglycerine
• Suicide, increased tendency associated to the use of
fluoxetine and other SSRI antidepressants
• Tardive dyskinesia associated with long-term use of
metoclopramide and many antipsychotic
31
Where to find more information
• http://www.smso.net/encyclolpedia :
•
•
•
•
•
•
•
Adverse_effects_of_drugs
http://www.who.int
Drug Safety Update –
FDA: http://www.fda.gov/
EMEA: http://www.emea.europa.eu/
SIDC: http://www.sukl.sk
http://www.liv.ac.uk/~druginfo/csm/adr%20presentation.htm
:
Clarifying Adverse Drug Events: A Clinician's Guide to Terminology, Documentation,
and Reporting Jonathan R. Nebeker, MS, MD; Paul Barach, MD, MPH; and Matthew H.
Samore, MD 18 May 2004 | Volume 140 Issue 10 | Pages 795-801