* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download LIPIDS
Survey
Document related concepts
Transcript
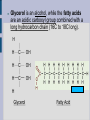
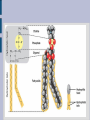
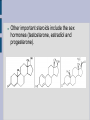
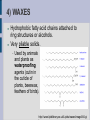
LIPIDS Hydrophobic, non-polar molecules Used for energy storage, building membranes and chemical signalling Four main types: fats, waxes, steroids and phospholipids 1) FATS Store more energy per gram than carbohydrates, BUT their energy is not as easily accessible. Used for: insulation, cushioning internal organs... Most common fats in plants and animals are the triacylglycerols (aka triglycerides) which consist of a glycerol backbone and 3 fatty acids. Glycerol is an alcohol, while the fatty acids are an acidic carboxyl group combined with a long hydrocarbon chain (16C to 18C long). H H C OH H C OH H C OH H Glycerol Fatty Acid Saturated fatty acids have no double bonds in the hydrocarbon chain, solid at room temperature (animal fats, butter, lard). Unsaturated fatty acids do have double bonds in the hydrocarbon chain, liquid at room temperature, known as oils (plant fats). HYDROGENATION ...is the process of adding hydrogen molecules to a plant oil in order to make it saturated and therefore solid (margarine /shortening). The process is done using an enzyme and hydrogen gas. Can lead to TRANS FATS ESTERIFICATION When glycerol combines with each fatty acid a condensation reaction (dehydration synthesis) occurs. An acid reacting with an alcohol produces an ester linkage, hence this process is called esterification. 2) PHOSPHOLIPIDS Make up the majority of the cell membrane. Contain a glycerol molecule combined with two fatty acids and a highly polar phosphate group. Phosphate head is hydrophillic while the fatty acid tail is hydrophobic. In a cell membrane the phospholipid bilayer makes it impossible for polar molecules and charged molecules to pass due to the hydrophobic center. That is why there are protein channels in the cell membrane to allow these types of molecules to pass through. http://academic.brooklyn.cuny.edu/biology/bio4fv/page/phoph.gif http://www.smartdraw.com/examples/content/Examples/Heal thcare/Microbiology_&_Biochemistry/Phospholipid_BiLayer_L.jpg 3) STEROLS (Steroids) Highly hydrophobic Four fused hydrocarbon rings with many different functional groups Cholesterol is a steroid, which is important in cell membranes to maintain their fluidity. It can also be dangerous as ingested cholesterol can lead to blocking of the arteries. Other important steroids include the sex hormones (testosterone, estradiol and progesterone). 4) WAXES Hydrophobic fatty acid chains attached to ring structures or alcohols. Very pliable solids. Used by animals and plants as waterproofing agents (cutin in the cuticle of plants, beeswax, feathers of birds). http://www.lipidlibrary.co.uk/Lipids/waxes/image004.gi