* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Slide 1
Static electricity wikipedia , lookup
History of electrochemistry wikipedia , lookup
Electrical resistance and conductance wikipedia , lookup
Alternating current wikipedia , lookup
Insulator (electricity) wikipedia , lookup
Stray voltage wikipedia , lookup
Supercapacitor wikipedia , lookup
Mains electricity wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Electromotive force wikipedia , lookup
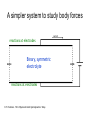
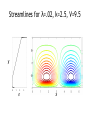
The role of Faradaic reactions in microchannel flows David A. Boy Brian D. Storey Franklin W. Olin College of Engineering Needham, MA Sponsor: NSF CTS, Research in Undergraduate Institutions. Motivation: ACEO & ICEO Electric Field Positive Ions Flow ++++++++++++++++++++++++ Negative Ions ----------------------------------- Negative Electrode Positive Electrode Advantages over DC • Low voltage, portable (~1 – 10 volts) • Good flow rates (~mm/s) Soni, Squires, Meinhart, BC00004 Swaminathan , Hu FC00003 Yossifon, Frankel, Miloh, GC00007 Green et al PRE 2000, 2002 Ajdari PRE 2000 Brown PRE 2000 Bazant & Squires JFM 2004 Olesen et al PRE 2005 Experimental observations (reactions have been proposed as possible mechanism for each of these) • Reversal of net pumping in ACEO is observed at high frequency. • Most flow stops at ~ 10 mM in ACEO & ICEO • Typically, only qualitative flow is predicted. Our goals • Understand the general coupling between reactions and flow. • Account for non-linear effects – Surface conduction – Mass transfer: concentrations at electrodes are not the same as the bulk. – Body forces outside of EDL. Olesen et al PRE 2005 A simpler system to study body forces current reactions at electrodes Binary, symmetric electrolyte reactions at electrodes R. F. Probstein. 1994. Physicochemical Hydrodynamics. Wiley. Bulk equations (symmetric, binary, dilute electrolyte): v 1 : electric potential v v p 2 v E E t Re : electrical conductivi ty E : charge density v 0 E 1 v E E t Pe 1 v E t Pe 2 1 E 2 E : electric field : dimensionl ess Debye length Pe : Peclet number Re : Reynolds number V : Dimensionl ess applied voltage K : Dimensionl ess reaction rates Voltage scaled thermal voltage (25 mV) λ = 0.1 to 0.0001 Pe = 100 to 1,000,000 Small device Dilute Large device High Concentration Boundary conditions boundary conditions at electrodes: - fixed voltage difference - No slip - reactions periodic boundary conditions in x f 0, y f 2 , y y 1 : v0 S n V E n R E n R Butler-Volmer reaction kinetics: R C exp C exp KH D : voltage drop across Stern layer 1D Solutions λ=0.01 K. T. Chu and M. Z. Bazant. 2005. SIAM J. Appl. Math. 65, 1485-1505. 1D Voltage-Current Behavior (fixed geometry & fluid properties) unstable Dilute K. T. Chu and M. Z. Bazant. 2005. SIAM J. Appl. Math. 65, 1485-1505. Rubinstein & Zaltzman PRE (2000, 2003, 2005 ) Fixed Debeye length 0.1 unstable Stable Streamlines for λ=.02, k=2.5, V=9.5 y 0 1 2 3 x Unsteady flow at high voltages Voltage-Current behavior ACEO Pumping Geometry Time averaged flow Electrode Electrode AC When reactions occur: •Flow occurs for all voltages •Flow occurs in AC and DC case •Flow is not symmetric even when electrodes are ACEO: Symmetric Electrodes (DC, λ=0.01, Pe=1000, V=10) Potential Charge Density Streamlines ACEO: Typical Streamlines (DC, λ=0.01, Pe=1000) V=1 Neg. V=5 Pos. Neg. V=20 V=10 Neg. Pos. Pos. Neg. Pos. Reverse the sign on the electrodes (DC, λ=0.01, Pe=1000, V=5) Pos. Neg. Neg. Pos. Frequency response (AC, λ=0.05 Pe=1000) Olesen et al. PRE 2005. Future work • Complete the parameter study of ACEO geometry. Can body forces destabilize the flow? • Compare ACEO flow computed with our “full” simulation to simpler models (i.e. Olesen et al. PRE 2005). • Use realistic reactions and electrolyte parameters as opposed to model binary, symmetric electrolyte. • Incorporate non-dilute effects. All applications well exceed kT/e = 25 mV. Conclusions • Body force in extended charge region can induce instability in parallel electrode geometry. • Instability occurs in parameter range found in microfluidic applications. • Thus far, we have not flow instability due to body forces in ACEO applications. Apparently, steady flow overwhelms the instability. (Note: our study is currently incomplete).