* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download M elatonin: a novel protective agent against oxidative injury of the
Heart failure wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Saturated fat and cardiovascular disease wikipedia , lookup
Electrocardiography wikipedia , lookup
Cardiovascular disease wikipedia , lookup
Jatene procedure wikipedia , lookup
Remote ischemic conditioning wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Cardiac surgery wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
Coronary artery disease wikipedia , lookup
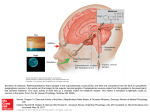
Cardiovascular Research 58 (2003) 10–19 www.elsevier.com / locate / cardiores Review Melatonin: a novel protective agent against oxidative injury of the ischemic / reperfused heart Russel J. Reiter*, Dun-Xian Tan Department of Cellular and Structural Biology, University of Texas Health Science Center, 7703 Floyd Curl Drive, Mail Code 7762, San Antonio, TX 78229 -3900, USA Received 3 September 2002; accepted 25 November 2002 Abstract Keywords: Cardiomyopathy; free radicals; Ischemia; Reperfusion 1. Introduction In the United States and other economically well-developed countries of the world, cardiovascular disease is a major cause of disability and mortality [1]. In these countries, conditions relating to the heart and adnexa account for almost half the deaths that occur each year. Furthermore, the costs of cardiovascular-related treatments including emergency room visits, by-pass surgery, cardiac catheterizations and stent placements have placed a major *Corresponding author. Tel.: 11-210-567-3859; fax: 11-210-5676948. E-mail address: [email protected] (R.J. Reiter). financial burden on the health care system and on many families. Ischemic heart disease is particularly debilitating with an estimated 1.5 million myocardial infarctions being reported annually in the US resulting in roughly 200 000 deaths. Unfortunately, significant reductions in these numbers seem unlikely considering the health status of many individuals. The American Heart Association estimates up to 50% of US inhabitants have elevated serum cholesterol with associated coronary artery disease [2] and in excess of 50% of the population is over weight, a factor that taxes the heart excessively. A reduction in coronary blood flow due to atherosTime for primary review 20 days. 0008-6363 / 03 / $ – see front matter 2003 European Society of Cardiology. Published by Elsevier Science B.V. All rights reserved. doi:10.1016 / S0008-6363(02)00827-1 Downloaded from by guest on October 12, 2016 This brief review summarizes the recently obtained evidence which illustrates the beneficial effects of the endogenously produced antioxidant, melatonin, in reducing tissue damage and reversing cardiac pathophysiology in models of experimental ischemia / reperfusion. The report also describes the actions of other antioxidants, especially vitamin E and antioxidative enzymes, in altering the degree of ischemia / reperfusion damage in the heart. Based on the data available, melatonin seems to have advantages over other antioxidants tested in terms of ameliorating the hypoxia and reoxygenation-induced damage. While the bulk of the studies that have used melatonin to overcome cardiac injury following transient arterial occlusion and subsequent reperfusion have used pharmacological doses to achieve protection, two recent reports have further shown that merely reducing endogenous circulating concentrations of melatonin (by surgical removal of a source of melatonin, i.e. the pineal gland) exaggerates the degree of injury and reduces survival of animals as a result of induced ischemia / reperfusion of the heart. These findings are consistent with observations in other organs where the loss of physiological concentrations of melatonin results in increased oxidative damage during hypoxia and reoxygenation. These findings have implications for the elderly since in the aged endogenous levels of melatonin are naturally reduced thereby possibly predisposing them to more severe cardiac damage during a heart attack. To date, the bulk of the studies relating to the protective actions of melatonin in reducing cardiac ischemia / reperfusion injury have used the rat as the experimental model. Considering the high efficacy of melatonin in limiting ischemia / reperfusion damage as well as melatonin’s low toxicity, the studies should be expanded to include other species and models of cardiac ischemia / reperfusion. The results of these investigations would help to clarify the potential importance of the use of melatonin in situations of oxidative damage to the heart in humans. 2003 European Society of Cardiology. Published by Elsevier Science B.V. All rights reserved. R. J. Reiter, D.-X. Tan / Cardiovascular Research 58 (2003) 10–19 clerotic plaques or vasospasm can be sufficiently prolonged to result in severe damage to the myocardium which leads to cellular injury and eventually to cellular death due to apoptosis and / or necrosis. Loss of this vital tissue ultimately leads to compromised cardiac function thereby jeopardizing the overall health of the individual and possibly leading to death. A number of treatment options have been exploited to reduce cardiac damage resulting from an ischemic episode. Some of these include the use of thrombolytic agents (e.g. tissue plasminogen activator and streptokinase) to dissolve clots and percutaneous transluminal balloon angioplasty; these procedures are essential to prevent long-term deprivation of oxygenated blood to the heart which would lead to tissue loss; however, the re-instatement of blood flow, i.e. reperfusion, is not without significant negative consequences and cellular damage [3,4]. Thus, when oxygenated blood reenters previously hypoxic cardiac tissue it initiates a cascade of events that, paradoxically, results in further cellular and tissue damage [5–8]. This tissue destruction is referred to as reperfusion injury. The combination of ischemia / reperfusion (I / R) causes 11 numerous and complex reactions in tissues (Fig. 1) which culminate in the mutilation of essential molecules and organelles in a number of cells including components of the coronary endothelium and myocardium with the recruitment of circulating blood elements, e.g. leukocytes and platelets. During the transient ischemia and the period of reperfusion many cells generate by-products of oxygen that are toxic to the heart. Indeed, the partially reducedoxygen metabolites, referred to as reactive oxygen species (ROS) are accepted as accounting for much of the cardiac damage that occurs during I / R injury [9,10]. 2. Use of melatonin to reduce cardiac I / R damage Several recent publications present evidence that a newly discovered antioxidant, melatonin, has significant protective actions against the cardiac damage and altered physiology that occurs during I / R injury. These actions were apparent even when melatonin was given as a single acute dose. In light of the observations that melatonin reduced molecular damage and cellular loss in other organs Downloaded from by guest on October 12, 2016 Fig. 1. As illustrated in this simplified figure, the events that lead to molecular damage and cell death during I / R injury are extremely complex. These processes clearly include free radical generation and ancillary events which eventually cause damage to key macromolecules. Considering the numerous intracellular actions of melatonin as a direct free radical scavenger, as an indirect antioxidant due to its ability to stimulate antioxidative enzymes, and its effect on mitochondrial electron transport and oxidative phosphorylation, this indole has proven effective in reducing molecular damage and cell death in models of rat I / R injury. 12 R. J. Reiter, D.-X. Tan / Cardiovascular Research 58 (2003) 10–19 with melatonin (1 or 10 mg / kg i.p.) or 5-MCA-NAT (10 mg / kg i.p.) 30 min before the hearts were perfused using the Langendorff method. In terms of both the physiological parameters and infarct size, the authors reported that melatonin or 5-MCA-NAT provided ‘spectacular protection’ against I / R injuries. Considering the large amount of experimental evidence implicating free radicals in cardiac arrhythmias during hypoxia / reoxygenation [14], Lagneux et al. [13] surmised that melatonin’s free radical scavenging activity accounted for its ability to restore cardiac function. However, since the role of free radicals in determining infarct size is somewhat less clear [15], the authors were more cautious in assuming that the reduction in the size of the lesion was exclusively related to the indole’s antioxidant properties. Additional confirmations of the ameliorative effects of melatonin on abnormal function and cardiac tissue destruction after I / R have been provided by Szarszoi et al. [16], Lee et al. [17] and Sahna and co-workers [18,19]. In the first of these reports, melatonin given in pharmacological doses (as in the studies reported above) prior to ischemia and / or during reperfusion reduced the incidence of reperfusion-induced ventricular fibrillation and decreased the arrhythmia score [16]. This protective action of melatonin was presumed to be related to its free radical scavenging activity. Lee et al. [17] performed the first in vivo study in which melatonin was tested for its ability to protect the heart when its blood supply is transiently interrupted. Using a bolus i.v. injection of either 0.5, 1.0 or 5.0 mg / kg before left coronary artery occlusion, melatonin markedly suppressed ventricular tachycardia and fibrillation and also reduced the total number of PVCs (Fig. 2). Additional studies showed that melatonin also significantly reduced O ?2 production and lowered myloperoxidase activity (an 2 index of neutrophil infiltration) during I / R. Finally, while eight of nine rats with an ischemic / reperfused heart died, none of the animals (10 of 10) treated with either 1 or 5 mg / kg melatonin succumbed. The authors of this report describe the actions of melatonin as being ‘pronounced’ and presumed its beneficial actions related to both its ability to reduce neutrophil infiltration into the ischemic / reperfused tissue and due to the antioxidant properties of the indole. Heretofore, the studies documenting the protective role of melatonin in the hypoxic / reperfused heart used pharmacological doses of the indole. Sahna and co-workers [18,19] were the first to examine whether endogenous, physiological concentrations of melatonin would change the outcome of such studies. In this case, rats were surgically pinealectomized to reduce endogenous levels of melatonin and, 2 months later, they, along with pinealintact controls, were used in studies of cardiac I / R injury. When the left coronary artery was occluded for 7 min followed by 7 min of reperfusion, the degree of cardiac arrhythmias was significantly greater in the heart of the Downloaded from by guest on October 12, 2016 during transient interruption of blood flow followed by reperfusion, Tan et al. [11] examined its effect on these responses at the level of the heart. Using a Langendorff rat heart preparation, a ligature was placed around the descending coronary artery for 10 min; this was estimated to have reduced total coronary flow by in excess of 25%. The ischemic episode was followed by a 10 min reperfusion period during which most hearts (80 and 90%, respectively) exhibited premature ventricular contractions (PVC) and / or ventricular fibrillation (VF). When melatonin was infused throughout the period of the occlusion and after reopening of the coronary artery, it highly significantly reduced both the PVC and VF. The arrhythmias in this study were defined in accordance with the guidelines of the Lamberth Conventions. The concentration of melatonin in the perfusate was either 1, 10 or 50 mM. Anticipating that melatonin’s effects in this study were likely related to its antioxidant action, as a positive control Tan et al. [11] infused another free radical scavenger, i.e. vitamin C, in a separate group of hearts. At a concentration of 500 mM (i.e. up to 500 times the concentration of melatonin), ascorbate was significantly less effective than melatonin in preventing the cardiac arrhythmias in this model of I / R. Additional investigations confirming the beneficial effects of melatonin on the physiology and morphology of the hypoxic / reoxygenated heart followed shortly. Kaneko et al. [12] also used isolated rat hearts that were subjected to 30 min ischemia and 30 min perfusion. The concentration of melatonin in the perfusion medium in this study was 100 mM. Again, the duration of ventricular tachycardia and VF were significantly reduced by melatonin relative to the durations of these arrhythmias in control hearts. Additionally, melatonin significantly restored the function of the left ventricule (LV), i.e. the LV developed pressure, 1dp / dt and 2dp / dt (63.0, 60.3 and 59.4%) compared to values of 30.2, 29.7 and 31.5%, respectively, in the diluent-infused hearts. Kaneko et al. [12] further extended their studies by measuring ?OH generation (by estimating levels of 2,3-dihydroxybenzoic acid after salicylate administration) and observed melatonin highly significantly reduced the amount of this metabolite in the perfusate. At the conclusion of the reperfusion period, the hearts were collected and the levels of lipid peroxidation products were measured in the tissues. For this endpoint, melatonin again highly significantly reduced the levels of oxidized lipids in the heart. These findings clearly documented the beneficial effects of melatonin when it is used during I / R of the heart and the results are consistent with melatonin’s protective actions as a free radical scavenger. Essentially simultaneously, Lagneux et al. [13] published their findings in which both melatonin and an analogue, 5-methoxy-carbonylamino-N-acetyl tryptamine (5-MCA-NAT), were compared in terms of their efficacies in reducing both the abnormal cardiac physiology and infarct volume after I / R. In this case, rats were treated R. J. Reiter, D.-X. Tan / Cardiovascular Research 58 (2003) 10–19 13 Downloaded from by guest on October 12, 2016 Fig. 2. Cardiac arrhythmias during induced ischemia (left) and reperfusion (right) of the rat heart and their reduction following the administration of melatonin. PVC, premature ventricular contraction; VT, ventricular tachycardia; VF, ventricular fibrillation; I / R, ischemia / reperfusion. * P,0.05 verses I / R only. pinealectomized rats compared to the controls. Even more importantly, the incidence of mortality was 63% in rats lacking their pineal gland compared to only 25% in the pineal-intact rats after I / R induction. These findings suggest that endogenous melatonin levels are protective of the heart during episodes of hypoxia and reoxygenation. A follow-up study by the same group [56] showed that infarct size was also exaggerated in the ischemia-reperfused heart when rats were deficient in physiological levels of melatonin due to pinealectomy. This is consistent with previously published findings in reference to the brain where a reduction in endogenous melatonin levels, due to pinealectomy, also exaggerated neural injury after an episode of I / R. The results showing increased I / R-induced cardiac damage after a reduction in physiological levels of melatonin have implications for the elderly inasmuch as in old individuals endogenous levels of melatonin may be significantly lower than in young individuals [20]. The findings illustrating a potential role for physiological levels of melatonin in abating cardiac damage during I / R also is interesting in light of several studies showing that humans with cardiovascular disease have noticeably lower circulating melatonin levels [21–24] than do age-matched subjects without significant cardiovascular deterioration. Similarly, patients suffering with cardiac syndrome X have an attenuated nocturnal rise in serum melatonin levels relative to that of age-matched individuals with no cardiac pathology [25]. It remains unknown, however, whether the reduced endogenous melatonin levels in patients with cardiovascular disease is a cause, an effect or even related to the compromised cardiovascular function. All of the studies summarized above in which melatonin, either physiologically or pharmacologically, was found to be protective of both the function and morphology of the I / R heart used the rat as the experimen- 14 R. J. Reiter, D.-X. Tan / Cardiovascular Research 58 (2003) 10–19 the cardiomyocytes. The reports that have investigated the efficacy of melatonin as a cardiac protector during I / R injury are summarized in Table 1. To further emphasize the efficacy of melatonin in reducing I / R damage, the reader is reminded that not only in the heart, but in other organs as well, melatonin has a highly favorable effect in reducing tissue damage that follows transient episodes of hypoxia and reoxygenation. Thus, in the in vivo brain (including the spinal cord) [29–32], liver [33], kidney [34], gastrointestinal tract [35], lung [36] and placenta [37] the amount of tissue damage and the severity of compromised function after I / R were shown to be reduced when melatonin was employed as an ameliorative agent. Furthermore, the damage inflicted by other agents that induce high oxidative stress is reduced when melatonin is exogenously administered [38–41]. Subsequent to the publication of the reports illustrating the potential utility of melatonin in the treatment of I / R, Duncker and Verdouw [27] posed the question as to whether melatonin has a future as a cardioprotective agent. The authors of the current review feel that this is a reasonable likelihood, not only in terms of protecting against I / R injury but also against cardiotoxicity induced by a variety of drugs [42]. For example, there is a significant amount of literature regarding the beneficial actions of melatonin at the level of heart in animals treated with chemotherapeutic drugs which often have significant toxicity in cardiac tissue which results in serious cardiomyopathy often leading to congestive heart failure [42– 45]. The data that have accumulated to date surely justify more extensive studies using melatonin in other models (and species) of cardiac I / R injury and, if successful, tests in humans as well. Melatonin is an uncommonly safe, stable and inexpensive molecule. For example, numerous studies have shown that melatonin, given to humans in huge pharmacological doses, e.g. 1 g daily for a month [46] or in smaller amounts for varying treatment periods [47–50], are without measurable side effects. The same holds for animal studies. Thus, giving female rats 200 mg melatonin per kg body weight (the equivalent of an average-weight human consuming 14 g melatonin daily) throughout pregnancy caused no maternal or fetal toxicity [51]. 3. Mechanisms of melatonin’s protective actions in the heart It is well accepted that O 2 -derived reactants can cause contractile dysfunction of the myocardium and certainly reperfusion of the heart with oxygenated blood after a period of ischemia causes oxidative damage in the reperfused tissue [52]. These toxic O 2 -based reactants along with RNS (Fig. 3) are widely believed to play a significant role in cardiopathophysiology after I / R [9,10]. Downloaded from by guest on October 12, 2016 tal model. The only other species that has been tested in similar studies is the rabbit. In this report [26], rabbits underwent coronary artery occlusion and reperfusion and, when melatonin was given at a dose of either 10 or 50 mg / kg i.v., it did not change the hemodynamic parameters of the I / R heart nor did it alter infarct volume. Melatonin also did not aggravate any of the parameters tested and the authors concluded that it was a safe drug with no apparent side effects on the cardiovascular system. Considering the numerous reports documenting the high efficacy of melatonin in reducing abnormal cardiac physiology, infarct volume and mortality in rats subjected to cardiac I / R, the results obtained in this rabbit study would seem to be unusual. In evaluating this study, Duncker and Verdouw [27] point out that the rabbit heart lacks xanthine oxidase which may account for the failure of the antioxidant melatonin to reduce cardiac malfunction during I / R in this species. It would seem judicious that melatonin administration be reassessed in the rabbit I / R model and certainly in other species that are considered surrogates for human heart attack. This seems especially important given melatonin’s highly protective effects in models of cardiac I / R in the rat. The doses of melatonin used in the aforementioned studies certainly caused blood concentrations of the indole to exceed the physiological levels that normally occur in the species used; therefore, the doses used were unquestionably pharmacological. On the other hand, pharmacological doses of any effective antioxidant would have to be given to reduce the massive cardiac damage that occurs during ischemia / reperfusion injury. Indeed, the reason the oxidative damage occurs in these situations is that the physiological levels of all antioxidants combined, i.e. glutathione, vitamins C and E, uric acid, etc., are insufficient to prevent the tissue destruction associated with such an extreme oxidative stress. A thorough in vitro analysis of melatonin’s actions at the level of the hypoxic / reoxygenated cardiomyocytes has been performed by Salie et al. [28]. For this study, adult rat cardiomyocytes were isolated and grown in culture and chemical hypoxia was induced by the addition of 1.5 mM KCN and 20 mM deoxyglucose to the superfusion fluid. Cells were preloaded with either tetramethylrhodamine (TMRM) in combination with either dichlorodihydrofluorescein (DCDHF), dihydrorhodamine 123 (DHR) or fluo 3 to determine the effects of melatonin, using confocal laser scanning microscopy, on H 2 O 2 levels, ROS concentrations and Ca 21 levels, respectively. Chemical hypoxia caused marked morphological changes of the cardiomyocytes and significantly increased H 2 O 2 , ROS and [Ca 21 ] i accumulation. Melatonin, at a concentration of either 50 or 100 mM, significantly reduced these changes (except for DCDHF fluorescence). These findings, as with the in vivo results summarized above, strongly implicate the antioxidant properties of melatonin in its protective effects against hypoxia / reoxygenation injury at the level of R. J. Reiter, D.-X. Tan / Cardiovascular Research 58 (2003) 10–19 15 Table 1 Publications in which pharmacological or physiological levels of melatonin have been used to protect the heart from ischemia / reperfusion (I / R) injury Duration of I / R (min) Actions of melatonin Reference Langendorff model (rat) 10 / 10 Reduced premature ventricular contraction and fibrillation [11] Langendorff model (rat) 30 / 30 Reduced ventricular tachycardia and fibrillation; restored left ventricular function; reduced lipid peroxidation, lowered ?OH generation [12] Langendorff model (rat) 5 / 30 Reduced reperfusion arrhythmias, reduced infarct volume [13] Langendorff model (rat) 20 / 40 Reduced ventricular fibrillation, improved arrhythmia score [16] In situ heart (rat) 45 / 60 Reduced ventricular tachycardia and fibrillation, lowered total ventricular contractions, reduced O ?2 and MPO activity, lowered 2 infarct volume, increased survival [17] In situ heart (rat) 7/7 Reduced ventricular fibrillation, lowered mortality [18] a In situ heart (rat) 3 / 120 Reduced infarct volume [19] a In situ heart (rabbit) 30 / 180 No improvement of cardiac function, no reduction in infarct volume [26] b Cardiomyocytes in culture (rat) 12.5 or 27.5 / 1.5 Reduced morphological damage, lowered reactive oxygen generation, reduced [Ca 21 ] i [28] 21 ?OH, hydroxyl radical; O ?2 ] i , intracellular calcium levels. 2 , superoxide anion radical; MPO, myloperoxidase activity; [Ca a In these reports, previously pinealectomized rats exhibited greater cardiac malfunction and damage as a result of I / R injury; these results suggest that physiological (endogenous) levels of melatonin protect the heart during I / R. b This is the only study on I / R and the potential of melatonin as a pharmacological protector that did not use the rat heart. That melatonin directly detoxifies O 2 -based free radicals and related reactants is well documented [53–56]. For example, melatonin is highly effective scavenger of the ?OH as shown using a variety of techniques by many different authors [57–59]; this is important because of the highly toxic nature of this reactant. Furthermore, some products of melatonin’s interaction with O 2 -based reactants have been identified and two of them, i.e. cyclic 3-hydroxymelatonin [60] and N-acetyl-5-methoxykynuramine (AFMK) [61,62], are also effective free radical scavengers [55,56]. This cascade of reactions greatly increases the effective concentrations of melatonin and may account, in part, for the high efficacy of this antioxidant in protecting against I / R injury at the level of the heart. Furthermore, the chief hepatic metabolite of melatonin, namely, 6-hydroxymelatonin, is also reportedly an effective free radial scavenger [63]. Thus, even when melatonin itself is metabolically converted to 6-hydroxymelatonin before it can function in the direct detoxification of a radical(s), its major hepatic metabolic product may be capable of doing so. Besides O 2 -based reactants, nitrogen-derived species (Fig. 3), most noteably the ONOO 2 and peroxynitrous acid (ONOOH) are likely involved in cardiomyocyte damage during I / R injury. Melatonin also has been shown to neutralize these non-radical, but nevertheless toxic, species [64,65] and, additionally, melatonin probably reduces ONOO 2 [which occurs when nitric oxide (NO?) couples with O ?2 2 ] by directly scavenging NO? [66,67] and / or by inhibiting its formation by suppressing the activity of its rate limiting enzyme, nitric oxide synthase. In I / R, NO? is generally considered to contribute to the resulting molecular damage, but under some other conditions inhibition of nitric oxide synthase may result in detrimental effects. While melatonin’s (and its metabolites) functions as a free radical scavenger, including its ability to apparently directly detoxify H 2 O 2 [68], may be important relative to its protective effects against injury due to hypoxia / reoxygenation, it is unlikely that this is the sole means of its beneficial effects. Not specifically during cardiac I / R injury but in other cases of high oxidative stress, melatonin has been shown to promote the metabolism of O 2 -based reactants to innocuous or less toxic substances. For example, melatonin stimulates gene expression and activities of the superoxide dismutases, important antioxidative en- Downloaded from by guest on October 12, 2016 Model 16 R. J. Reiter, D.-X. Tan / Cardiovascular Research 58 (2003) 10–19 zymes which dismutase O ?2 to H 2 O 2 (Fig. 3) [69]. 2 Thereafter, the two enzymes assigned the function of metabolizing H 2 O 2 , i.e. catalase and glutathione peroxidase (GPx), and thereby reducing the formation of the devastatingly toxic ?OH, have been shown to be stimulated by melatonin as well [70,71]. These indirect antioxidative actions of melatonin could certainly magnify its protective actions in the heart under conditions of high oxidative stress. Also of significance is a report by Urata et al. [72] who have shown that melatonin increases the levels of glutathione, an important intracellular antioxidant, by stimulating its rate-limiting enzyme, gammaglutamylcysteine synthase. Besides directly neutralizing and indirectly detoxifying oxygen and nitrogen-based reactants, melatonin may also act at the level of the mitochondrial respiratory chain to reduce electron leakage and, therefore, free radical generation [73,74]. While melatonin’s actions at the level of the mitochondria is a rather recent area of investigative interest, the ability of the indole to reduce electron leakage is inferred from its stimulatory action on the activities of complex I and complex IV, its promotion of ATP production [73], and its action in reducing oxidative damage to mitochondrial DNA. It is anticipated that research related to the mitochondrial actions of melatonin will flourish in the next several years and the information garnered will clarify how the functions of melatonin at the mitochondrial level may be beneficial to the heart generally and during I / R injury specifically. Another important component of I / R injury is the recruitment of activated leukocytes to the injured tissue (Fig. 1) [75]. Once at their target, these cells generate inflammatory mediators, e.g. leukotriene B 4 , platelet-activating factor, etc., and they also lead to the activation of oxidant-sensitive nuclear transcription factors, in particular nuclear factor kappa B (NFkB); these agents, combined with the generation of free radicals by the leukocytes, induce inflammatory mediators by endothelial cells and / or mast cells which cause the mobilization of adhesion molecules (selectins and ICAM). As a consequence, leukocytes adhere to the endothelium which leads to a cascade of events that contribute to cardiac damage. Melatonin, due to its ability to reduce the expression of the adhesion molecules, P-selectin and ICAM [76], as well as the suppression of NFkB translocation into the nucleus [77] may be helpful in reducing cardiac injury during I / R. Although the specific details of the mechanisms involved in the melatonin’s ability to reduce oxidative damage at the level of the heart (and other tissues) during hypoxia and especially during reperfusion remain to be clarified, studies to date almost uniformly confirm the beneficial effects of the indole under these conditions. Clearly, melatonin has at its disposal numerous means by which it may limit cardiac damage during I / R (Table 2), but which of these functions plays the dominant role in its protective actions is uncertain at this time. Beyond the actions of melatonin described, Duncker and Verdouw [27] summarize still other effects of melatonin which may be beneficial to cardiovascular physiology in general and cardiac function in particular. Downloaded from by guest on October 12, 2016 Fig. 3. Oxygen and nitrogen-based reactants which are generated in abundance during ischemia / reperfusion inflict massive damage to molecules and cellular organelles. SOD, superoxide dismutase; GPx, glutathione peroxidase; GRd, glutathione reductase; NOS, nitric oxide synthase. R. J. Reiter, D.-X. Tan / Cardiovascular Research 58 (2003) 10–19 Table 2 Effects of melatonin that potentially contribute to its ability of protect the cardiovascular system from oxidative injury Actions References Scavengers of oxygen-based reactants Scavenges nitrogen-based reactants Cascade actions whereby melatonin metabolites also function of scavengers Stimulates antioxidative enzymes Inhibits a proxidative enzyme Increases glutathione levels Stabilizes cellular membranes Increases the efficiency of oxidative phosphorylation Reduces leukocyte recruitment and adhesion molecule expression Reduces homocysteine-induced damage [53–58] [64,65,78] [55,61,63] [70,79] [71] [72] [80,81] [73,74] [35,76] [82,83] 4. Concluding remarks References [1] Keys A. Coronary artery disease in seven countries. Circulation 1970;41(Suppl 1):1–8. [2] Kannel WB, Casletti WP, Gordon T. Cholesterol in the prediction of atherosclerotic disease: New perspectives based on the Framingham study. Ann Intern Med 1979;90:85–91. [3] Koren G, Weiss AT, Hasin Y et al. Prevention of myocardial damage by early treatment with intravenous streptokinase. New Engl J Med 1985;313:1384–1389. [4] Braunwald E, Kloner RA. Myocardial reperfusion: A double-edged sword? Circulation 1985;76:1713–1719. [5] Simpson PJ, Michelson JK, Lucchesi BR. Free radical scavengers in myocardial ischemia. Fed Proc 1987;46:2413–2421. [6] Tsao PS, Aoki N, Lefer DJ, Johnson G, Lefer AM. Time course of endothelial dysfunction during myocardial ischemia and reperfusion in the cat. Circulation 1990;82:1402–1412. [7] Ma LX, Weyrich AS, Lefer DJ, Lefer AM. Diminished basal nitric oxide release after myocardial ischemia and reperfusion promotes neutrophil adherence to coronary endothelium. Circ Res 1993;72:403–412. [8] Hansen PR. Role of neutrophils in myocardial ischemia and reperfusion. Circulation 1995;91:1872–1885. [9] Lefer DJ, Granger DN. Oxidative stress and cardiac disease. Am J Med 2000;109:315–323. ˜ G, Elhafidi M, Franco M. Oxidative stress and cardiovascular [10] Banos physiopathology. Curr Topics Pharmacol 2000;5:1–17. [11] Tan DX, Manchester LC, Reiter RJ et al. Ischemia / reperfusioninduced arrhythmias in the isolated rat heart: Prevention by melatonin. J Pineal Res 1998;25:184–191. [12] Kaneko S, Okumura K, Numaguchi Y et al. Melatonin scavenges hydroxyl radical and protects isolated rat hearts from ischemic reperfusion injury. Life Sci 2000;67:101–112. [13] Lagneux C, Joyeux M, Demenge P, Ribant C, Godin-Ribuot D. Protective effect of melatonin against ischemia–reperfusion injury in the isolated rat heart. Life Sci 2000;66:503–509. [14] Hearse DJ. Stunning: A radical re-review. Cardiovasc Drugs Ther 1991;5:225–236. [15] Jeroudi DO, Hartley CJ, Bolli R. Myocardial reperfusion injury: Role of oxygen radicals and potential therapy with antioxidants. Am J Physiol 1994;72:2B–7B. [16] Szarszoi O, Asemu G, Vanecek J, Ostadal B, Kolar F. Effects of melatonin on ischemia and reperfusion injury of the rat heart. Cardiovasc Drugs Ther 2001;5:251–257. [17] Lee YM, Chen HR, Hsiao G et al. Protective effects of melatonin on myocardial ischemia / reperfusion injury in vivo. J Pineal Res 2002;33:72–80. [18] Sahna E, Olmez E, Acet A. Effects of physiological and pharmacological concentrations of melatonin on ischemia–reperfusion arrhythmias in rats: Can the incidence of complete sudden cardiac death be reduced? J Pineal Res 2002;32:194–198. [19] Sahna E, Acet A, Ozer MK, Olmez E. Myocardial ischemia– reperfusion in rats: Reduction of infarct size by either supplemental physiological or pharmacological doses of melatonin. J Pineal Res 2003: in press. [20] Reiter RJ. The aging pineal gland and its physiological consequences. BioEssays 1992;14:169–175. [21] Brugger P, Marktl W, Herold M. Impaired nocturnal secretion of melatonin in coronary heart disease. Lancet 1995;345:1408. [22] Brugger P, Herold M. Human melatonin and cortisol circadian rhythms in patients with coronary heart disease. Biol Rhythm Res 1998;29:121–128. [23] Sakotnik A, Liebmann PM, Stoschitsky K et al. Decreased melatonin synthesis in patients with coronary artery disease. Eur Heart J 1999;20:1314–1317. [24] Dominguez-Rodriquez A, Abreu-Gonzalez P, Garcia MJ, Sanchez J, Manero F, de Armas-Trujillo D. Decreased nocturnal melatonin levels during acute myocardial infarction. J Pineal Res 2003: in press. [25] Altun A, Yaprak M, Aktoz M et al. Impaired nocturnal synthesis of melatonin in patients with cardiac syndrome X. Neurosci Lett 2002;327:143–145. [26] Dave RH, Hole SL, Kloner RA. The effect of melatonin on hemodynamics, blood flow and myocardial infarct size in a rabbit Downloaded from by guest on October 12, 2016 In this brief review we have summarized the data describing the protective actions of an endogenously produced molecule, melatonin (and its metabolites), on myocardial damage that accompanies a transient interruption and re-establishment of blood flow to the heart. While melatonin’s beneficial actions are thus far convincing, by far the bulk of the studies have been directed to a single species, the rat. Extension of these findings to other commonly used experimental models of heart attack is essential for advancement of this potentially important molecule as a cardioprotective agent. The importance of these studies is emphasized by the fact that melatonin, given either acutely or chronically at pharmacological doses, is virtually without toxicity [46,49–51]. Additionally, when given orally it is rapidly absorbed and it is also safe and easy to administer intravenously. Furthermore, exogenously administered melatonin does not interfere with its endogenous production. It is known that melatonin levels in the serum correlate with the total antioxidant status of this fluid in both the rat and human [79,84,85]. Its ability to protect the heart does not only derive from studies documenting its beneficial actions during I / R injury, but also the data showing it is equally protective of the heart against drugs, especially cancer chemotherapeutic agents, that normally cause cardiomyopathy [41–44]. In view of the large amount of positive data that has already accumulated, additional studies in this field should be of high priority. 17 18 [27] [28] [29] [30] [31] [32] [33] [34] [36] [37] [38] [39] [40] [41] [42] [43] [44] [45] [46] [47] model of ischemia–reperfusion. J Cardiovasc Pharmacol Ther 1998;3:153–160. Duncker DJ, Verdouw PD. Has melatonin a future as a cardioprotective agent? Cardiovasc Drugs Ther 2001;15:205–207. Salie R, Harper I, Allie C et al. Melatonin protects against ischemic–reperfusion myocardial damage. J Mol Cell Cardiol 2001;33:343–357. ¨ Kilic E, Ozdemir Y, Bolay H, Kelestimur H, Dalkara T. Pinealectomy aggravates and melatonin administration attenuates brain damage in focal ischemia. J Cereb Blood Flow Metab 1999;19:511– 516. Wakatsuki A, Okatani Y, Shinohara K, Ikenoue N, Fukaya T. Melatonin protects against ischemia / reperfusion-induced oxidative damage to mitochondria in fetal rat brain. J Pineal Res 2001;31:167–172. Pei Z, Pang SF, Cheung RTF. Pretreatment with melatonin reduces volume of cerebral infarction in a rat middle cerebral artery occlusion stroke model. J Pineal Res 2002;32:168–172. Sun FY, Lin X, Mao LZ et al. Neuroprotection by melatonin against ischemia neuronal injury associated with modulation of DNA damage and repair in rat following a transient cerebral ischemia. J Pineal Res 2002;33:57–60. Rodriquez-Reynoso S, Leal C, Portilla E, Olivaries N, Munoz J. Effect of melatonin on hepatic energetic status during ischemia / reperfusion: Possible role of tumor necrosis factor-a and nitric oxide. J Surg Res 2001;100:141–149. Sener G, Schirli AD, Keyer-Uysal M et al. The protective effect of melatonin on renal ischemia–reperfusion injury in the rat. J Pineal Res 2002;32:120–126. Cuzzocrea S, Costantino G, Mazzon R et al. Beneficial effects of melatonin in a model of splanchnic artery occlusion and reperfusion. J Pineal Res 2000;28:52–63. Inci I, Inci D, Dutly A, Boehler A, Weder W. Melatonin attenuates posttransplant lung ischemia–reperfusion injury. Ann Thorac Surg 2002;73:220–225. Okatani Y, Wakatsuki A, Shinohara K, Taniguchi K, Fukaya T. Melatonin protects against oxidative mitochondrial damage induced in rat placenta by ischemia and reperfusion. J Pineal Res 2001;31:173–178. Hara M, Yoshida M, Nishijima H et al. Melatonin, a pineal secretory product with antioxidant properties, protects against cisplatin-induced nephrotoxicity in rats. J Pineal Res 2001;30:129–138. Tesoriere L, Allegra M, D’Arpa D, Butera D, Livrea MA. Reaction of melatonin with hemoglobin-derived oxoferryl radicals and inhibition of the hydroperoxide-induced hemoglobin denaturation in red blood cells. J Pineal Res 2001;31:114–119. Wu CC, Chiao CW, Hsias G, Chen A, Yen MH. Melatonin prevents endotoxin-induced circulatory failure in rats. J Pineal Res 2001;30:147–156. Reiter RJ, Tan DX, Sainz RM, Mayo JC. Melatonin protects the heart against both ischemia / reperfusion injury and chemotherapeutic drugs. Cardiovasc Drugs Ther 2002;16:5–6. Reiter RJ, Tan DX, Sainz RM, Mayo JC, Lopez-Burillo S. Melatonin: Reducing the toxicity and increasing the efficacy of drugs. J Pharm Pharmacol 2002;54:1299–1321. Agapito MT, Antolin Y, del Brio MT et al. Protective effect of melatonin against adriamycin toxicity in the rat. J Pineal Res 2001;31:23–30. Xu MF, Ho S, Qian ZM, Tan PL. Melatonin protects against cardiac toxicity of doxorubicin in rat. J Pineal Res 2001;31:301–307. Granzotto M, Rapozzi V, Decorti G, Giraldi T. Effects of melatonin on doxorubicin cytotoxicity in sensitive and pleotropically resistant tumor cells. J Pineal Res 2001;31:206–213. Norlund JJ, Lerner AB. The effects of melatonin on skin color and on the release of pituitary hormones. J Clin Endocrinol Metab 1997;45:768–774. Molina-Carballo A, Munoz-Hoyos A, Reiter RJ et al. Utility of high [48] [49] [50] [51] [52] [53] [54] [55] [56] [57] [58] [59] [60] [61] [62] [63] [64] [65] [66] [67] [68] doses of melatonin adjunctive therapy in a child with severe myoclonic epilepsy: Two years’ experience. J Pineal Res 1997;23:97–105. Brusco LI, Marquez M, Cardinali DP. Monozygotic twins with Alzheimer’s disease treated with melatonin: Case report. J Pineal Res 1998;25:260–263. Seabra MDLV, Bignotto M, Pinto Jr. LR, Tufik S. Randomized, double-blind clinical trial, controlled with placebo, of the toxicology of chronic melatonin treatment. J Pineal Res 2000;29:193–200. Jan JE, Hamilton D, Seward N et al. Clinical trials of control released melatonin in children with sleep–wake cycle disorders. J Pineal Res 2000;29:34–39. Jahnke G, Marr M, Myers R et al. Maternal and developmental toxicity evaluation of melatonin administration orally to pregnant Sprague–Dawley rats. Toxicol Res 1999;50:271–279. McCord JM. Free radicals and myocardial ischemia: Overview and outlook. Free Radic Biol Med 1988;4:9–14. Hardeland R, Balzer I, Poeggeler B et al. On the primary function of melatonin in evolution: Mediation of photoperiodic signals in a unicells, photoxidation and scavenging of free radicals. J Pineal Res 1995;18:104–111. Reiter RJ, Tan DX, Manchester LC, Qi W. Biochemical reactivity of melatonin with reactive oxygen and nitrogen species: A review of the evidence. Cell Biochem Biophys 2001;34:237–256. Tan DX, Reiter RJ, Manchester LC et al. Chemical and physical properties and potential mechanisms: Melatonin as a broad spectrum antioxidant and free radical scavenger. Curr Topics Med Chem 2002;2:181–198. Allegra M, Reiter RJ, Tan DX, Gentile C, Tesoriere L, Livrea MA. The chemistry of melatonin’s interaction with reactive species. J Pineal Res 2003;34:1–10. Tan DX, Chen LD, Poeggeler B, Manchester LC, Reiter RJ. Melatonin: A potent, endogenous hydroxyl radical scavenger. Endocr J 1993;1:57–60. Matuszek Z, Reszka K, Chignell CF. Reaction of melatonin and related indoles with hydroxyl radicals: ESR and spin trapping investigations. Free Radic Biol Med 1997;23:347–372. Reiter RJ. Melatonin: Lowering the high price of free radicals. News Physiol Sci 2000;15:246–250. Tan DX, Manchester LC, Reiter RJ et al. A novel melatonin metabolite, cyclic 3-hydroxymelatonin: A biomarker of melatonin interaction with hydroxyl radicals. Biochem Biophys Res Commun 1998;253:614–620. Tan DX, Manchester LC, Burkhardt S et al. N 1 -acetyl-N 2 -formyl-5methoxykynuramine, a biogenic amine and melatonin metabolite, functions as a potent antioxidant. FASEB J 2001;15:2294–2296. Hardeland R, Poeggeler B, Niebergali R, Zelosko V. Oxidation of melatonin by carbonate radicals and chemiluminescence emitted during pyrrole ring cleavage. J Pineal Res 2003: in press. Maharaj DS, Anoopkumar-Dukie S, Glass BD et al. The identification of the UV degradants of melatonin and their ability to scavenge radicals. J Pineal Res 2002;32:257–261. Dugo L, Serriano I, Fulia F et al. Effect of melatonin on cellular energy depletion mediated by peroxynitrite and poly (ADP-ribose) synthetase activation in an acute model of inflammation. J Pineal Res 2001;31:76–84. Zhang H, Squadrito GL, Pryor WA. The reaction of melatonin with peroxynitrite: Formation of melatonin radical cation and absence of stable nitrated products. Biochem Biophys Res Commun 1998;251:83–87. Blanchard B, Pompon D, Ducroq C. Nitrosation of melatonin by nitric oxide and peroxynitrite. J Pineal Res 2000;29:184–192. Turjanski AG, Leonik F, Rosenstein RF, Estrin DA, Doctorovich F. Scavenging of NO by melatonin. J Am Chem Soc 2000;122:10468– 10469. Tan DX, Manchester LC, Reiter RJ et al. Melatonin directly scavenges hydrogen peroxide: A potentially new metabolic pathway Downloaded from by guest on October 12, 2016 [35] R. J. Reiter, D.-X. Tan / Cardiovascular Research 58 (2003) 10–19 R. J. Reiter, D.-X. Tan / Cardiovascular Research 58 (2003) 10–19 [69] [70] [71] [72] [73] [74] [75] [76] [77] [78] Cuzzocrea S, Reiter RJ. Pharmacological actions of melatonin in acute and chronic inflammation. Curr Topics Med Chem 2002;2:153–165. [79] Albarran MT, Lopez-Burillo S, Pablos MI, Reiter RJ, Agapito MT. Endogenous rhythms of melatonin, total antioxidant status and superoxide dismutase activity in several tissues of chick and their inhibition by light. J Pineal Res 2001;30:227–233. [80] Garcia JJ, Reiter RJ, Guerrero JM et al. Melatonin prevents changes in microsomal membrane fluidity during induced lipid peroxidation. FEBS Lett 1997;408:297–300. [81] Garcia JJ, Reiter RJ, Ortiz GG et al. Melatonin enhances tamoxifen’s ability to prevent the reduction in microsomal membrane fluidity induced by lipid peroxidation. J Membr Biol 1998;162:59– 65. [82] Okatani Y, Wakatsuki A, Reiter RJ. Melatonin suppresses homocysteine enhancement of serotonin-induced vasoconstriction of the human umbilical artery. J Pineal Res 2001;31:242–247. [83] Osuna C, Reiter RJ, Garcia JJ et al. Inhibitory effect of melatonin on homocysteine-induced lipid peroxidation in rat brain homogenates. Pharmacol Toxicol 2002;90:32–37. [84] Benot S, Molinero P, Soutto M, Goberna R, Guerrero JM. Circadian variations in the rat serum total antioxidant status: Correlation with melatonin levels. J Pineal Res 1998;25:1–4. [85] Benot S, Goberna R, Reiter RJ et al. Physiological levels of melatonin contribute to the antioxidant capacity of human serum. J Pineal Res 1999;27:59–64. Downloaded from by guest on October 12, 2016 of melatonin biotransformation. Free Radic Biol Med 2000;29:1177–1185. Antolin I, Rodriquez C, Sainz RM et al. Neurohormone melatonin prevents cell damage: Effect on gene expression for antioxidant enzymes. FASEB J 1996;10:882–890. Okatani Y, Wakatsuki A, Shinohara K, Kaneda C, Fukaya T. Melatonin stimulates glutathione peroxidase activity in human chorion. J Pineal Res 2001;30:199–205. Reiter RJ, Tan DX, Osuna C, Gitto L. Actions of melatonin in the reduction of oxidative stress. J Biomed Sci 2001;7:444–458. Urata Y, Honma S, Goto S et al. Melatonin induces gglutamylcysteine synthetase mediated by activator protein-1 in human vascular endothelial cells. Free Radic Biol Med 1999;27:838–847. ˜ Acuna-Castroviejo D, Martin M, Macias M et al. Melatonin, mitochondria and cellular bioenergetics. J Pineal Res 2001;30:65– 74. Reiter RJ, Tan DX, Manchester LC, El-Sawi MR. Melatonin reduces oxidant damage and promotes mitochondrial respiration: Implications for aging. Ann NY Acad Sci 2002;959:238–250. Granger DN. I / R injury: Role of leukocyte–endothelial adhesion. News Physiol Sci 1997;12:142–143. Cuzzocrea S, Mazzon E, Serriano I et al. Melatonin reduces dinitrobenzene sulfonic acid-induced colitis. J Pineal Res 2001;30:1–12. Lezovalc’h F, Szaropani M, Behl C. N-acetyl-serotonin (normelatonin) and melatonin protect neurons against oxidative challenges and suppresses the activity of the transcription factor NF kappa B activation. FASEB J 1998;12:685–693. 19