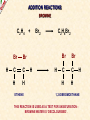* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download gorgpps.pps - Knockhardy
Survey
Document related concepts
Transcript
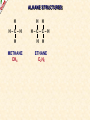
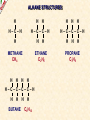
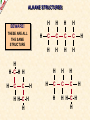
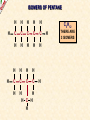
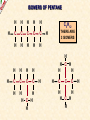
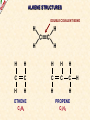
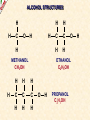
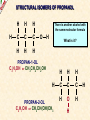
ORGANIC CHEMISTRY A guide for GCSE students 2010 KNOCKHARDY PUBLISHING SPECIFICATIONS ORGANIC CHEMISTRY INTRODUCTION This Powerpoint show is one of several produced to help students understand selected GCSE Chemistry topics. It is based on the requirements of the AQA specification but is suitable for other examination boards. Individual students may use the material at home for revision purposes and it can also prove useful for classroom teaching with an interactive white board. Accompanying notes on this, and the full range of AS and A2 Chemistry topics, are available from the KNOCKHARDY WEBSITE at... www.knockhardy.org.uk All diagrams, photographs and any animations in this Powerpoint are original and created by Jonathan Hopton. Permission must be obtained for their use in any work that is distributed for financial gain. ORGANIC CHEMISTRY CONTENTS • What is organic chemistry? • Hydrocarbons • Alkanes • Alkenes • Alcohols • Industrial preparation of alcohols • Carboxylic acids and esters • Questions ORGANIC CHEMISTRY This Powerpoint presentation does not cover… DISTILLATION OF CRUDE OIL THE PETROCHEMICAL INDUSTRY POLYMERS For further information on these topics, please see the other Powerpoints on the Knockhardy GCSE site. ORGANIC CHEMISTRY WHAT IS IT? ORGANIC CHEMISTRY WHAT IS IT? Organic chemistry is the study of carbon compounds. It is such a complex branch of chemistry because... ORGANIC CHEMISTRY WHAT IS IT? Organic chemistry is the study of carbon compounds. It is such a complex branch of chemistry because... • CARBON ATOMS FORM STRONG COVALENT BONDS TO EACH OTHER ORGANIC CHEMISTRY WHAT IS IT? Organic chemistry is the study of carbon compounds. It is such a complex branch of chemistry because... • CARBON ATOMS FORM STRONG COVALENT BONDS TO EACH OTHER • THE CARBON-CARBON BONDS CAN BE SINGLE, DOUBLE OR TRIPLE ORGANIC CHEMISTRY WHAT IS IT? Organic chemistry is the study of carbon compounds. It is such a complex branch of chemistry because... • CARBON ATOMS FORM STRONG COVALENT BONDS TO EACH OTHER • THE CARBON-CARBON BONDS CAN BE SINGLE, DOUBLE OR TRIPLE • CARBON ATOMS CAN BE ARRANGED IN and STRAIGHT CHAINS BRANCHED CHAINS RINGS ORGANIC CHEMISTRY WHAT IS IT? Organic chemistry is the study of carbon compounds. It is such a complex branch of chemistry because... • CARBON ATOMS FORM STRONG COVALENT BONDS TO EACH OTHER • THE CARBON-CARBON BONDS CAN BE SINGLE, DOUBLE OR TRIPLE • CARBON ATOMS CAN BE ARRANGED IN and STRAIGHT CHAINS BRANCHED CHAINS RINGS • OTHER ATOMS/GROUPS OF ATOMS ARE PLACED ON CARBON ATOMS ORGANIC CHEMISTRY WHAT IS IT? Organic chemistry is the study of carbon compounds. It is such a complex branch of chemistry because... • CARBON ATOMS FORM STRONG COVALENT BONDS TO EACH OTHER • THE CARBON-CARBON BONDS CAN BE SINGLE, DOUBLE OR TRIPLE • CARBON ATOMS CAN BE ARRANGED IN and STRAIGHT CHAINS BRANCHED CHAINS RINGS • OTHER ATOMS/GROUPS OF ATOMS ARE PLACED ON CARBON ATOMS ORGANIC CHEMISTRY CHAINS AND RINGS CARBON ATOMS CAN BE ARRANGED IN STRAIGHT CHAINS BRANCHED CHAINS and RINGS You can also get a combination of rings and chains ORGANIC CHEMISTRY SINGLE AND MULTIPLE BONDING CARBON-CARBON COVALENT BONDS CAN BE SINGLE, DOUBLE OR TRIPLE HYDROCARBONS Compounds containing only carbon and hydrogen atoms HOMOLOGOUS SERIES A FAMILY OF COMPOUNDS WHICH HAVE… ● A GENERAL FORMULA ● SIMILAR CHEMICAL PROPERTIES HOMOLOGOUS SERIES A FAMILY OF COMPOUNDS WHICH HAVE… ● A GENERAL FORMULA ● SIMILAR CHEMICAL PROPERTIES Examples:- HOMOLOGOUS SERIES A FAMILY OF COMPOUNDS WHICH HAVE… ● A GENERAL FORMULA ● SIMILAR CHEMICAL PROPERTIES Examples:ALKANES CnH2n+2 CH4 C 2H 6 HOMOLOGOUS SERIES A FAMILY OF COMPOUNDS WHICH HAVE… ● A GENERAL FORMULA ● SIMILAR CHEMICAL PROPERTIES Examples:ALKANES CnH2n+2 CH4 C 2H 6 ALKENES CnH2n C2H4 C 3H 6 HOMOLOGOUS SERIES WHAT ARE THE NEXT IN EACH SERIES? ALKANES ALKENES CH4 C2H6 C2H4 ? C3H6 ? ? ? ? HOMOLOGOUS SERIES WHAT ARE THE NEXT IN EACH SERIES? ALKANES ALKENES CH4 C2H6 C2H4 C3H8 C3H6 C4H10 C4H8 C5H12 C5H10 ALKANES SATURATED HYDROCARBONS ALKANES WHAT ARE THEY? ● HYDROCARBONS ● CARBON ATOMS ARE JOINED BY SINGLE COVALENT BONDS ● CONTAIN THE MAXIMUM NUMBER OF HYDROGEN ATOMS ● ARE SATURATED HYDROCARBONS ALKANES WHAT ARE THEY? ● HYDROCARBONS ● CARBON ATOMS ARE JOINED BY SINGLE COVALENT BONDS ● CONTAIN THE MAXIMUM NUMBER OF HYDROGEN ATOMS ● ARE SATURATED HYDROCARBONS ● EXAMPLES… METHANE CH4 ETHANE C 2H 6 PROPANE C 3H 8 BUTANE C4H10 GENERAL FORMULA IS… CnH2n+2 ALKANE STRUCTURES H H–C–H H METHANE CH4 ETHANE C2H6 PROPANE C3H8 ALKANE STRUCTURES H H–C–H H METHANE CH4 H H H–C–C–H H H ETHANE C2H6 PROPANE C3H8 ALKANE STRUCTURES H H H–C–H H H–C–C–H H METHANE CH4 H H ETHANE C2H6 H H H H–C–C–C–H H H H PROPANE C3H8 THESE ARE DISPLAYED STRUCTURES THEY SHOW ALL THE COVALENT BONDS IN THE MOLECULE Each covalent bond is represented by a line ALKANE STRUCTURES H H H–C–H H–C–C–H H H METHANE CH4 H H H H BUTANE H H ETHANE C2H6 H H–C–C–C–C–H H H H C4H10 H H H H–C–C–C–H H H H PROPANE C3H8 ALKANE STRUCTURES H H H–C–H H METHANE CH4 H H H BUTANE H H H H C4H10 H H H–C–C–C–H H ETHANE C2H6 H–C–C–C–C–H H H H–C–C–H H H H H H PROPANE C3H8 HOWEVER By the time you get to butane, there are two ways to arrange the carbon and hydrogen atoms… ALKANE STRUCTURES C4H10 H H–C–H H H H H H–C–C–C–C–H H H H BUTANE H H H H – C ––– C ––– C – H H H H METHYLPROPANE STRUCTURAL ISOMERS Compounds with the SAME MOLECULAR FORMULA but DIFFERENT STRUCTURAL FORMULA ALKANE STRUCTURES BEWARE! THESE ARE ALL THE SAME STRUCTURE H H H H H C C C C H H H H H H C H H H C C HH C H H H H H H H C C C H H H C H H H H ISOMERS OF PENTANE C5H12 HOW MANY ISOMERS ARE THERE? ISOMERS OF PENTANE C5H12 THERE ARE 3 ISOMERS ISOMERS OF PENTANE H H H H H H C C C C C H H H H H C5H12 H THERE ARE 3 ISOMERS ISOMERS OF PENTANE H H H H H H H C C C C C H H H H H H H H H C C C C H H H H C H H H C5H12 H THERE ARE 3 ISOMERS ISOMERS OF PENTANE H H H H H H H C C C C C H H H H H H H H H C C C C H H H H C H H C5H12 THERE ARE 3 ISOMERS H H H C H H H H H C C C H H H C H H H SOME PROPERTIES OF ALKANES SOME PROPERTIES OF ALKANES Boiling point increases as they get more carbon atoms in the formula CH4 (-161°C) C2H6 (-88°C) C3H8 (-42°C) C4H10 (-0.5°C) SOME PROPERTIES OF ALKANES Boiling point increases as they get more carbon atoms in the formula CH4 (-161°C) C2H6 (-88°C) C3H8 (-42°C) C4H10 (-0.5°C) Viscosity Greater number of C atoms = greater viscosity Flammability Greater number of C atoms = less flammable SOME PROPERTIES OF ALKANES Boiling point increases as they get more carbon atoms in the formula CH4 (-161°C) C2H6 (-88°C) C3H8 (-42°C) C4H10 (-0.5°C) Viscosity Greater number of C atoms = greater viscosity Flammability Greater number of C atoms = less flammable Use as fuels Alkanes make useful fuels CH4 + 2O2 CO2 + 2H2O SOME PROPERTIES OF ALKANES Boiling point increases as they get more carbon atoms in the formula CH4 (-161°C) C2H6 (-88°C) C3H8 (-42°C) C4H10 (-0.5°C) Viscosity Greater number of C atoms = greater viscosity Flammability Greater number of C atoms = less flammable Use as fuels Alkanes make useful fuels CH4 + 2O2 CO2 + 2H2O The more carbon atoms they have the more oxygen they need C3H8 + 5O2 3CO2 + 4H2O ALKENES UNSATURATED HYDROCARBONS ALKENES WHAT ARE THEY? ● HYDROCARBONS ● CONTAIN A DOUBLE C=C COVALENT BOND ● ARE UNSATURATED HYDROCARBONS – THEY CAN HAVE ATOMS ADDED TO THEM ● ARE MORE REACTIVE THAN ALKANES ALKENES WHAT ARE THEY? ● HYDROCARBONS ● CONTAIN A DOUBLE C=C COVALENT BOND ● ARE UNSATURATED HYDROCARBONS – THEY CAN HAVE ATOMS ADDED TO THEM ● ARE MORE REACTIVE THAN ALKANES ● EXAMPLES… ETHENE C 2H 4 PROPENE C 3H 6 BUTENE C 4H 8 GENERAL FORMULA IS… CnH2n ALKENE STRUCTURES DOUBLE COVALENT BOND H H H H H C C C C C H H H ETHENE C2H4 H PROPENE C3H6 H ALKANES & ALKENES HOW CAN YOU TELL THEM APART? ADD BROMINE WATER; if the reddish-brown colour is removed the substance possesses a C=C bond. A PLACE A SOLUTION OF BROMINE IN A TEST TUBE B ADD THE HYDROCARBON TO BE TESTED AND SHAKE C IF THE BROWN COLOUR DISAPPEARS THEN THE HYDROCARBON IS AN ALKENE A B C ADDITION REACTIONS BROMINE C2H4 H Br Br C C H H ETHENE + H Br2 C2H4Br2 H Br Br C C H H H 1,2-DIBROMOETHANE THIS REACTION IS USED AS A TEST FOR UNSATURATION BROMINE WATER IS ‘DECOLOURISED’. ADDITION REACTIONS HYDROGEN C2H4 H H H C C H H ETHENE + H H2 CATALYST H C2H6 H H C C H H ETHANE VEGETABLE OILS CONTAINING UNSATURATED FATS ARE HARDENED TO FORM MARGARINE THIS WAY H POLYMERISATION For more detailed information on POLYMERS, please see the appropriate Powerpoint on the Knockhardy GCSE site. www.knockhardy.org.uk/gcse.htm POLYMERISATION • during polymerisation, alkenes undergo an addition reaction • all the atoms in the original alkenes are used to form the polymer • long hydrocarbon chains are formed POLYMERISATION • during polymerisation, alkenes undergo an addition reaction • all the atoms in the original alkenes are used to form the polymer • long hydrocarbon chains are formed POLYMERISATION • during polymerisation, alkenes undergo an addition reaction • all the atoms in the original alkenes are used to form the polymer • long hydrocarbon chains are formed • the equation shows… the original monomer and the repeating unit in the polymer ethene MONOMER poly(ethene) POLYMER POLYMERISATION • during polymerisation, alkenes undergo an addition reaction • all the atoms in the original alkenes are used to form the polymer • long hydrocarbon chains are formed • the equation shows… the original monomer and the repeating unit in the polymer n represents a large number ethene MONOMER poly(ethene) POLYMER the number of repeating units is the same as the number of original molecules POLYMERISATION The animation shows the monomers turning into the polymer ALCOHOLS ALCOHOLS WHAT ARE THEY? ● NOT HYDROCARBONS ● CONTAIN THE –OH FUNCTIONAL GROUP ALCOHOLS WHAT ARE THEY? ● NOT HYDROCARBONS ● CONTAIN THE –OH FUNCTIONAL GROUP ● EXAMPLES… METHANOL CH3OH ETHANOL C2H5OH PROPANOL C3H7OH GENERAL FORMULA IS… CnH2n+1OH ALCOHOL STRUCTURES H H C O H H H METHANOL CH3OH H H H C C H H O ETHANOL C2H5OH H H H C C C H H H O H PROPANOL C3H7OH H STRUCTURAL ISOMERS OF PROPANOL H H H H C C C H H H There is another alcohol with the same molecular formula O PROPANOL C3H7OH or CH3CH2CH2OH H What is it? STRUCTURAL ISOMERS OF PROPANOL H H H H C C C H H H There is another alcohol with the same molecular formula O H What is it? PROPAN-1-OL C3H7OH or CH3CH2CH2OH H PROPAN-2-OL C3H7OH or CH3CH(OH)CH3 H H H C C C H O H H H REACTIONS OF ALCOHOLS COMBUSTION • ALCOHOLS MAKE USEFUL FUELS • ETHANOL IS A CLEAN FUEL - DOESN’T FORM POLLUTANTS • ETHANOL CAN BE MADE BY FERMENTATION (RENEWABLE) C2H5OH ETHANOL + 3O2 OXYGEN 2CO2 + CARBON DIOXIDE 3H2O WATER REACTIONS OF ALCOHOLS OXIDATION • ALCOHOLS CAN BE OXIDISED TO CARBOXYLIC ACIDS • ETHANOL IS OXIDISED TO ETHANOIC ACID (ACETIC ACID) • THIS IS WHY WINE GOES SOUR WHEN LEFT TOO LONG Air ETHANOL Wine ETHANOIC ACID Vinegar REACTIONS OF ALCOHOLS ESTERIFICATION • ALCOHOLS REACT WITH CARBOXYLIC ACIDS • THE REACTION IS REVERSIBLE • COMPOUNDS CALLED ESTERS ARE FORMED • ESTERS HAVE CHARACTERISTIC SMELLS CH3COOH + C2H5OH ETHANOIC ACID ETHANOL CH3COOC2H5 + H2O ETHYL ETHANOATE WATER REACTIONS OF ALCOHOLS REACTION WITH SODIUM • ALCOHOLS REACT WITH SODIUM • HYDROGEN GAS IS PRODUCED 2Na SODIUM + 2C2H5OH 2C2H5ONa ETHANOL SODIUM ETHOXIDE + H2 HYDROGEN INDUSTRIAL PREPARATION OF ALCOHOLS INDUSTRIAL PREPARATION OF ALCOHOLS 1. FERMENTATION Reagent(s) GLUCOSE - produced by the hydrolysis of starch Conditions yeast warm, but no higher than 37°C Equation C6H12O6 SUGAR (GLUCOSE) ——> 2 C2H5OH ETHANOL + 2 CO2 CARBON DIOXIDE INDUSTRIAL PREPARATION OF ALCOHOLS 1. FERMENTATION Reagent(s) GLUCOSE - produced by the hydrolysis of starch Conditions yeast warm, but no higher than 37°C Equation C6H12O6 Advantages LOW ENERGY PROCESS USES RENEWABLE RESOURCES - PLANT MATERIAL SIMPLE EQUIPMENT ——> 2 C2H5OH Disadvantages SLOW PRODUCES IMPURE ETHANOL BATCH PROCESS + 2 CO2 INDUSTRIAL PREPARATION OF ALCOHOLS 2. HYDRATION OF ETHENE Reagent(s) ETHENE - from cracking of fractions from crude oil Conditions catalyst - phosphoric acid high temperature and pressure Equation C2H4 + ETHENE H2O STEAM ——> 2 C2H5OH ETHANOL INDUSTRIAL PREPARATION OF ALCOHOLS 2. HYDRATION OF ETHENE Reagent(s) ETHENE - from cracking of fractions from crude oil Conditions catalyst - phosphoric acid high temperature and pressure Equation C2H4 + Advantages FAST PURE ETHANOL PRODUCED CONTINUOUS PROCESS H2O ——> 2 C2H5OH Disadvantages HIGH ENERGY PROCESS EXPENSIVE PLANT REQUIRED USES NON-RENEWABLE FOSSIL FUELS TO MAKE ETHENE INDUSTRIAL PREPARATION OF ALCOHOLS USES OF ETHANOL • ALCOHOLIC DRINKS • SOLVENT • FUEL - industrial alcohol / methylated spirits - petrol substitute in countries with limited oil reserves CARBOXYLIC ACIDS CARBOXYLIC ACIDS WHAT ARE THEY? ● NOT HYDROCARBONS ● CONTAIN THE –COOH FUNCTIONAL GROUP CARBOXYLIC ACIDS WHAT ARE THEY? ● NOT HYDROCARBONS ● CONTAIN THE –COOH FUNCTIONAL GROUP ● EXAMPLES… METHANOIC ACID HCOOH ETHANOIC ACID CH3COOH PROPANOIC ACID C2H5COOH CARBOXYLIC ACID STRUCTURES O H C O H H H O C C O H METHANOIC ACID HCOOH ETHANOIC ACID CH3COOH H CARBOXYLIC ACIDS USES ● VINEGAR CONTAINS ETHANOIC ACID - ethanoic acid is used in the manufacture of rayon ● ORANGES AND LEMONS CONTAIN CITRIC ACID ● ASPIRIN IS A CARBOXYLIC ACID - it is used for pain relief and prevention heart attacks ● VITAMIN C CONTAINS ASCORBIC ACID - it is used for pain relief and prevention heart attacks CARBOXYLIC ACIDS REACTIONS ● WEAK ACIDS - neutralised by alkalis to form salts - react with carbonates making salts, carbon dioxide and water ● REACT WITH ALCOHOLS TO PRODUCE ESTERS ESTERS ESTERS WHAT ARE THEY? ● NOT HYDROCARBONS ● CONTAIN THE –COOC FUNCTIONAL GROUP ESTERS WHAT ARE THEY? ● NOT HYDROCARBONS ● CONTAIN THE –COOC FUNCTIONAL GROUP ● FORMATION… Formed by replacing the H on the COOH of a carboxylic acid by a carbon atom group METHANOIC (HCOOH) ACID METHYL METHANOATE (HCOOCH3) ETHANOIC (CH3COOH) ACID METHYL ETHANOATE (CH3COOCH3) ESTERS WHAT ARE THEY? ● NOT HYDROCARBONS ● CONTAIN THE –COOC FUNCTIONAL GROUP ● FORMATION… Formed by replacing the H on the COOH of a carboxylic acid by a carbon atom group METHANOIC (HCOOH) ACID METHYL METHANOATE (HCOOCH3) ETHANOIC (CH3COOH) ACID METHYL ETHANOATE (CH3COOCH3) ● USE… Flavourings – many have fruity odours QUESTIONS QUESTIONS WHICH FORMULAE REPRESENT ALKANES? C10H22 C 3H 6 C 2H 6 C 2H 6O C6H12O6 C3H7COOH QUESTIONS WHICH FORMULAE REPRESENT ALKANES? C10H22 C 3H 6 C 2H 6 C 2H 6O C6H12O6 C3H7COOH QUESTIONS CARBOXYLIC ACID OR ESTER? HCOOC2H5 C3H7COOH C6H6COOCH3 C2H5COOC3H7 CH3COOH QUESTIONS CARBOXYLIC ACID OR ESTER? HCOOC2H5 ESTER C3H7COOH CARBOXYLIC ACID C6H6COOCH3 ESTER C2H5COOC3H7 ESTER CH3COOH CARBOXYLIC ACID QUESTIONS WHAT TYPES OF ORGANIC COMPOUND ARE THESE? H H H–C–C–C–H H H H H H O C C C H H H H O H H H H C C H H O H H H H O C C C H H H O C H H QUESTIONS WHAT TYPES OF ORGANIC COMPOUND ARE THESE? H H H–C–C–C–H H H H O C C C H H H O H H ALKANE H H H H H C C H H O H ALCOHOL H CARBOXYLIC ACID H H O C C C H H H O C H ESTER H ORGANIC CHEMISTRY THE END ©2011 JONATHAN HOPTON & KNOCKHARDY PUBLISHING