* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download naming-and-formulas-chem-1-ab
Bremsstrahlung wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Rigid rotor wikipedia , lookup
Host–guest chemistry wikipedia , lookup
List of phenyltropanes wikipedia , lookup
Hypervalent molecule wikipedia , lookup
History of molecular theory wikipedia , lookup
History of chemistry wikipedia , lookup
Oxidation state wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Extended periodic table wikipedia , lookup
Organic chemistry wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
Molecular dynamics wikipedia , lookup
Drug discovery wikipedia , lookup
Atomic theory wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Metalloprotein wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Ionic compound wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Homoaromaticity wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
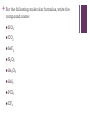
+ Nomenclature Chemistry 1 + The Language of Chemistry Subscripts indicate the number of atoms of an element in a molecule. C8H18 How or MgCl2 many atoms of each element are in the following? CH4 SO2 O2 P4O10 Pb(NO3)2 + Review What is a cation? What is an anion? What group of elements typically form +1 cations? Why? What group of elements typically form +2 cations? Why? What group of elements typically form -1 anions? Why? What group of elements typically form -2 anions? Why? + Ions and Compounds Monatomic ions (N3-) Ions formed from a single atom Polyatomic ions (SO42-) Ions formed from more than one atom See hand-out (purple) Binary Compounds Made of two elements Ternary Compounds Made of three or more elements (contains polyatomic ion) **When writing chemical formulas for ionic compounds, the overall charge of the compound must be ZERO/NEUTRAL!** + Formula of Ionic Compounds 1) List the cation (positively charged ion) chemical symbol first. 2) List the anion (negatively charged ion) chemical symbol after the cation. 3) If the charges are not the same (sum of the oxidation numbers must equal zero), subscripts must be added to indicate a ratio of positive and negative ions. -The polyatomic ion acts as an individual ion. -Because polyatomic ions act as a unit, NEVER CHANGE SUBSCRIPTS OF ANY ATOM WITHIN THE ION. -Transition metals with more than one oxidation number… the charge is noted using roman numerals! + Let’s Practice Writing Formulas Q: What is the chemical formula for aluminum fluoride? 1. Symbol for aluminum ion = Al3+ Symbol for fluoride ion = F- 2. Al3+F3. Least common multiple is 4. AlF3 5. Orbital Notation Verification . + Practice Writing Formulas Write the correct formula for the following ionic compounds. 1) Potassium and Iodide 2) Magnesium and Chloride 3) Aluminum and Bromide 4) Cesium and Nitride 5) Barium and Sulfide + Practice Writing Formulas With Polyatomic Ions 1) Sodium and Nitrate 2) Calcium and Chlorate 3) Aluminum and Carbonate 4) Potassium and Chromate 5) Magnesium and Carbonate + Naming Ionic Compounds List the name of the cation (watch for transition metals). List the name of the anion, changing the ending to “ide” If you have a polyatomic anion, just list its name after the cation. CaCl2 Na2S = calcium chloride = sodium sulfide + Naming Ionic Compounds With A Transition Metal Work backwards to determine the charge on the transition metal…. Cr2O3 FeO NiCl2 Mn3(PO4)2 Rules To Name Compound: 1. Name the cation 2. Place the charge of the transition metal as a roman numeral 3. Name the anion by changing the ending to “ide” + Review – Binary Compounds Write the correct chemical formula for each of the following compounds. potassium bromide magnesium selenide sodium phosphide tin(IV) chloride aluminum oxide scandium(III) sulfide Give the correct names for each of the following compounds listed below. KF SnI4 SiF4 AlBr3 FeCl3 ZnO + Review Ternary Compounds Write the chemical formulas for the following names: Iron (III) acetate Gold(III) sulfate Ammonium phosphate Calcium nitrate Write the chemical names for the following chemical formulas: FeC2O4 Pb(CO3)2 Al(MnO4)3 Ba(OH)2 + Entrance Ticket Write the formula for the following compounds. Hint-look up the charges for each element/ion and then balance the charges Calcium Nitride Rubidium Sulfide Lead (II) Nitrate Iron (III) Oxide Manganese (III) Chlorate Write the name for the following compounds. Cr3P2 Al2 (SO4) 3 BaO CoN Cu3 (PO4 ) 2 + Molecular Compounds Nomenclature (Covalent) When naming compounds consisting of two nonmetals, the names of the elements are written in the same order as they appear in the formula. (The more metallic element is written first.) The second element uses the suffix “-ide”. Prefixes are added to the name of each element to indicate the NUMBER of atoms of the element in the molecule. (If the first element’s prefix is mono-, it will be dropped. For example, monocarbon dioxide (CO2) is simply called carbon dioxide.) **Note: the “o” or “a” at the end of a prefix is usually dropped when the word following the prefix begins with another vowel…e.g. monoxide or pentoxide. + Prefixes –Polyatomic Handout Prefix Number of atoms Mono- 1 Di- 2 Tri- 3 Tetra- 4 Penta- 5 Hexa- 6 Hepta- 7 Octa- 8 Nona- 9 Deca- 10 + For the following molecular compounds, write the molecular formula: Carbon Sulfur mononitride tribromide Phosphorus Nitrogen Carbon dioxide tetrachloride Dihydrogen Sulfur triflouride monoxide trioxide Dinitrogen hexoxide + For the following molecular formulas, write the compound name: SiO2 CO2 SeF6 N2O5 As2O5 AsI3 PCl5 CF4 + Flow Chart Challenge Create a flow chart to use while NAMING Ionic and Covalent Compounds Hints: How do you distinguish between an Ionic and Covalent Compound? Which one uses prefixes? When do you use Roman Numerals in the name? What do you do for naming polyatomic ions? + Chemical Formulas Chemistry 1 + Oxidation Numbers “oxidation The states” number of electrons that must be added to or removed from an atom in a combined state to convert the atom into the elemental form. + Assigning Oxidation States Rules for Assigning Oxidation States The Oxidation State of… An atom in an element is zero A monatomic ion is the same as its charge HF, PF3 Oxygen is usually -2 in its compounds Na+, Cl- Fluorine is -1 in its compounds Na(s), O2(g), Hg(l) H2O, CO2 Exception: Peroxides (containing O22-) in which oxygen is -1 Hydrogen is +1 in its covalent compounds H2O, HCl, NH3 + Oxidation Numbers - Model Assign oxidation numbers to the atoms of each element in the following compounds. PI3 HF CS2 As2S3 NO2- + Oxidation Numbers Practice Assign oxidation numbers to the atoms of each element in the following compounds. CI4 HCl SO3 IO3 H2CO3 SO42- + Percent Composition The percentage by mass of each element in a compound. massofelementincompound x100% = %ElementinCompound massofcompound +1. Using the periodic table determine the molar mass of the compound. 2. Take each mass for each element (molar mass times number of atoms) and divide it by the total mass of the compound. 3. Multiply this value by 100 to obtain a percentage. 4. Percentages should add up to equal 100% Steps For Calculations + Percent Composition Determine the percent composition in the following compounds: PbBr4 Ba(OH)2 + Percent Composition Determine the percent composition in the following compounds: NaOH (NH4)3PO4 CCl2F2 Pb(NO3)2 + Bite the Bubble Lab See handout + Empirical Formula: The formula with the smallest whole number ratio of the elements in the compound (simplest formula) 1. Note: May not always be the same as the molecular formula. To calculate, convert masses of elements to moles. If a question gives the percentages of each compound, assume you have 100 g of the sample and then each percentage becomes the mass of the element. 2. Next, divide each mole by the smallest one. 3. This gives the ratio of atoms, which you will use to write the formula. Empirical Formula + Empirical Formulas The percent composition of a compound was found to be 63.5% silver, 8.2% nitrogen, and 28.3% oxygen. Determine this compound’s empirical formula. Name the compound. + Empirical Formulas A 170.00g sample of a compound contains 29.84 g sodium, 67.49 g chromium and 72.67 g oxygen. What is the empirical formula? Name the compound. + Empirical Formula 200.00 grams of an organic compound is known to contain 83.884 grams of carbon, 10.486 grams of hydrogen, 18.640 grams of oxygen and the rest is nitrogen. What is the empirical formula of the compound? + Molecular Formulas Molecular Formulas: A formula that specifies the actual number of atoms of each element in one molecule of the substance. -Molecular formula is a multiple of the empirical formula. Steps for Solving: Determine the Empirical Formula first! Use the formula : molecularweight N= empiricalformulaweight N= The number we will multiply the subscripts in the empirical formula by to obtain the new molecular formula. + Empirical and Molecular Formulas A compound of boron and hydrogen has a percent composition of 78.14% boron and 21.86% hydrogen. If the molar mass is 27.6, what is the empirical and molecular formula? + Empirical and Molecular Formulas A compound with the formula C2H5O is found to have a molar mass of 90g. What is the molecular formula of the compound? + Test will cover… New Ionic, Molecular, and Acid Nomenclature Oxidation Numbers and Percent Composition Empirical and Molecular Formulas Chemical Bonding Periodic Table Trends, Electron Configuration and Periodic Table History Chapter Review