* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Tamoxifen
Pharmacokinetics wikipedia , lookup
Discovery and development of antiandrogens wikipedia , lookup
Discovery and development of direct thrombin inhibitors wikipedia , lookup
Prescription costs wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Psychopharmacology wikipedia , lookup
Psychedelic therapy wikipedia , lookup
Neuropharmacology wikipedia , lookup
Drug interaction wikipedia , lookup
Toxicodynamics wikipedia , lookup
Pharmacogenomics wikipedia , lookup
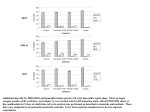
Tamoxifen NMe2 O Trade Names Nolvadex Classification Antiestrogen Category Hormonal agent Drug Manufacturer AstraZeneca Mechanism of Action Nonsteroidal antiestrogen with weak estrogen agonist effects. Competes with estrogen for binding to ERs. Binding of tamoxifen to ER leads to ER dimerization. The tamoxifen-bound ER dimer is transported to the nucleus, where it binds to DNA sequences referred to as ER elements. This interaction results in inhibition of critical transcriptional processes and signal transduction pathways that are required for cellular growth and proliferation. Cell cycle–specific agent that blocks cells in the mid-G1 phase of the cell cycle. Effect may be mediated by cyclin D. Stimulates the secretion of transforming growth factor- (TGF-) which then acts to inhibit the expression and/or activity of TGF-_ and IGF-1, two genes that are involved in cell growth and proliferation. Mechanism of Resistance Decreased expression of ER. Mutations in the ER leading to decreased binding affinity to tamoxifen. Overexpression of growth factor receptors, such as TGF-_ or IGF-1, that counteract the inhibitory effects of tamoxifen. Absorption Rapidly and completely absorbed in the GI tract. Peak plasma levels are achieved within 4–6 hours after oral administration. Distribution Distributes to most body tissues, especially in those expressing estrogen receptors. Present in very low concentrations in CSF. Nearly all of drug is bound to plasma proteins. Metabolism Extensively metabolized by liver cytochrome P450 enzymes after oral administration. The main metabolite, N-desmethyl tamoxifen, has biologic activity similar to that of the parent drug. Both tamoxifen and its metabolites are excreted primarily (75%) in feces with minimal clearance in urine. The terminal half-lives of tamoxifen and its metabolites are relatively long, approaching 7–14 days. Indications Adjuvant therapy in axillary node-negative breast cancer following surgical resection. Adjuvant therapy in axillary node-positive breast cancer in postmenopausal women following surgical resection. Adjuvant therapy in women with ductal carcinoma in situ (DCIS) after surgical resection and radiation therapy. Metastatic breast cancer in women and men. Approved as a chemopreventive agent for women at high risk for breast cancer. “High-risk” women are defined as women _35 years and with a 5-year predicted risk of breast cancer _1.67%, according to the Gail model. Endometrial cancer. Dosage Range Recommended dose for treatment of breast cancer patients is 20 mg PO every day. Drug Interaction 1 Warfarin—Tamoxifen can inhibit metabolism of warfarin by the liver P450 system leading to increased anticoagulant effect. Coagulation parameters, including PT and INR, must be closely monitored, and dose adjustments may be required. Drug Interaction 2 Drugs activated by liver P450 system—Tamoxifen and its metabolites are potent inhibitors of hepatic P450 enzymes and may inhibit the metabolic activation of drugs utilizing this pathway, including cyclophosphamide. Drug Interaction 3 Drugs metabolized by liver P450 system—Tamoxifen and its metabolites are potent inhibitors of hepatic P450 enzymes and may inhibit the metabolism of various drugs, including erythromycin, calcium channel blockers, and cyclosporine. Special Considerations Instruct patients to notify physician about menstrual irregularities, abnormal vaginal bleeding, and pelvic pain and/or discomfort while on therapy. Patients should have routine follow-up with a gynecologist as tamoxifen therapy is associated with an increased risk of endometrial hyperplasia, polyps, and endometrial cancer. Use with caution in patients with abnormal liver function as there may be an increased risk of drug accumulation resulting in toxicity. Use with caution in patients with either personal history or family history of thromboembolic disease or hypercoagulable states as tamoxifen is associated with an increased risk of thromboembolic events. Tamoxifen therapy is associated with antithrombin III deficiency. Initiation of treatment with tamoxifen may induce a transient tumor flare. Tamoxifen should not be given in patients with impending ureteral obstruction, spinal cord compression, or in those with extensive painful bone metastases. Premenopausal patients should be warned about the possibility of developing menopausal symptoms with tamoxifen therapy. Monitor CBC during therapy with tamoxifen as myelosuppression can occur, albeit rarely. Pregnancy category D. Toxicity 1 Menopausal symptoms, including hot flashes, nausea, vomiting, vaginal bleeding, and menstrual irregularities. Vaginal discharge and vaginal dryness also observed. Toxicity 2 Fluid retention and peripheral edema observed in about 30% of patients. Toxicity 3 Tumor flare usually occurs within the first 2 weeks of starting therapy. May observe increased bone pain, urinary retention, back pain with spinal cord compression, and/or hypercalcemia. Toxicity 4 Headache, lethargy, dizziness occur rarely. Visual disturbances, including cataracts, retinopathy, and decreased visual acuity, have been described. Toxicity 5 Skin rash, pruritus, hair thinning, and/or partial hair loss. Toxicity 6 Myelosuppression is rare, with transient thrombocytopenia and leukopenia. Usually resolves after the first week of treatment. Toxicity 7 Thromboembolic complications, including deep vein thrombosis, pulmonary embolism, and superficial phlebitis. Incidence of thromboembolic events may be increased when tamoxifen is given concomitantly with chemotherapy. Toxicity 8 Elevations in serum triglycerides. Toxicity 9 Increased incidence of endometrial hyperplasia, polyps, and endometrial cancer.