* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download PWE 16-5: Determining Charge-to
Introduction to gauge theory wikipedia , lookup
Field (physics) wikipedia , lookup
Jerk (physics) wikipedia , lookup
Magnetic monopole wikipedia , lookup
Lagrangian mechanics wikipedia , lookup
Anti-gravity wikipedia , lookup
Modified Newtonian dynamics wikipedia , lookup
Path integral formulation wikipedia , lookup
Relational approach to quantum physics wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Renormalization wikipedia , lookup
Nuclear physics wikipedia , lookup
Newton's laws of motion wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Fundamental interaction wikipedia , lookup
Electrostatics wikipedia , lookup
Electric charge wikipedia , lookup
A Brief History of Time wikipedia , lookup
Standard Model wikipedia , lookup
Lorentz force wikipedia , lookup
Equations of motion wikipedia , lookup
Chien-Shiung Wu wikipedia , lookup
Classical mechanics wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Centripetal force wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Work (physics) wikipedia , lookup
Elementary particle wikipedia , lookup
Newton's theorem of revolving orbits wikipedia , lookup
History of subatomic physics wikipedia , lookup
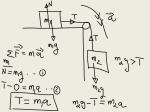
Example 16-5 Determining Charge-to-Mass Ratio When released from rest in a uniform electric field of magnitude 1.00 * 104 N>C, a certain charged particle travels 2.00 cm in 2.88 * 1027 s in the direction of the field. You can ignore any nonelectric forces acting on the particle. (a) What is the charge-to-mass ratio of this particle (that is, the ratio of its charge q to its mass m)? (b) Other experiments show that the mass of this particle is 6.64 * 10227 kg. What is the charge of this particle? Set Up The only force that acts on the particle is the electric force given by Equation 16-2. Since the particle accelerates in the direction of the s, the force on the particle must electric field E s. So the charge on also be in the direction of E s is uniform the particle must be positive. Since E (it has the same value at all points), the force on the particle will be constant. Its acceleration ax will be constant as well, so we can use one of the constant-acceleration equations from Chapter 2 to determine ax. We’ll use this with Newton’s second law and Equation 16-2 to learn what we can about this particle. Solve (a) We are given that the particle travels 2.00 cm = 2.00 * 1022 m in 2.88 * 1027 s. Use this to determine the particle’s constant acceleration. Relate the acceleration to the net external (electric) force on the particle and solve for the charge-to-mass ratio. Electric field and electric force: s (16-2) Fs = qE Straight-line motion with constant acceleration: 1 x = x0 + v0x t + axt 2 2 Newton’s second law: s s a Fext = ma q>0 a E x (2-9) (4-2) Take the positive x direction to be the direction in which the particle moves. The particle begins at rest, so v0x = 0. If we take the initial position of the particle to be x0 = 0, then Equation 2-9 becomes 1 x = ax t 2 2 Solve for the acceleration: 212.00 * 10-2 m2 2x = 4.82 * 1011 m>s 2 ax = 2 = t 12.88 * 10-7 s2 2 The net force on the particle of charge q is the electric force in the x direction, which from Newton’s second law is equal to the mass m of the particle multiplied by the acceleration ax: qEx = max This says that the acceleration of a particle in an electric field depends on the particle’s charge-to-mass ratio: q E ax = m x In this example we know both ax and Ex, so the charge-to-mass ratio is 4.82 * 1011 m>s 2 q ax = = m Ex 1.00 * 104 N>C Since 1 N = 1 kg # m>s 2, this is q = 4.82 * 107 C>kg m (b) Given the charge-to-mass ratio and the mass of the particle, determine the charge q. Reflect The charge of the particle is q q = ma b = 16.64 * 10-27 kg2 14.82 * 107 C>kg2 m = 3.20 * 10219 C The charge on a proton is e = 1.60 * 10219 C; the charge on this particle is 2e. The particle in this example has about four times the mass of a proton but only double the charge of a proton. For historical reasons it’s known as an alpha particle; in fact, it’s the nucleus of a helium atom, which contains two protons (each with charge e) and two neutrons (each with nearly the same mass as a proton but with zero charge).