* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
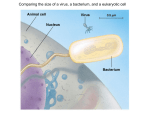
Download Virus Structure
Ebola virus disease wikipedia , lookup
Viral phylodynamics wikipedia , lookup
Social history of viruses wikipedia , lookup
Bacteriophage wikipedia , lookup
Endogenous retrovirus wikipedia , lookup
Oncolytic virus wikipedia , lookup
Introduction to viruses wikipedia , lookup
Virus quantification wikipedia , lookup
Henipavirus wikipedia , lookup
History of virology wikipedia , lookup
Plant virus wikipedia , lookup