* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Redox reactions
George S. Hammond wikipedia , lookup
Kinetic resolution wikipedia , lookup
Asymmetric induction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Baylis–Hillman reaction wikipedia , lookup
Petasis reaction wikipedia , lookup
Hydroformylation wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
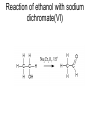
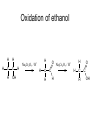
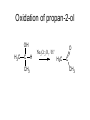
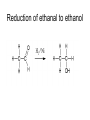
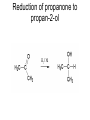
Redox reactions Redox reactions of primary alcohols • Primary alcohols react with oxidising agents such as potassium manganate(VII) or sodium dichromate(VI), forming the corresponding aldehyde • For example, ethanol reacts forming ethanal • Ethanal is also formed in the metabolism of ethanol in the human body Reaction of ethanol with sodium dichromate(VI) Reaction of ethanol with sodium dichromate(VI) • This reaction is used in the preparation of ethanal • Reaction conditions: heat, excess ethanol, acidified sodium dichromate(VI) solution • The aldehyde is distilled off as it is formed in order to prevent further oxidation to ethanoic acid Preparation of ethanal Oxidation of primary alcohols • Primary alcohols such as ethanol are oxidised to the corresponding aldehydes, which can be further oxidised to the corresponding carboxylic acids. Oxidation of ethanol H H H C C H OH H + H Na2Cr2O7 / H H C H O C H Na2Cr2O7 / H+ H H C H O C OH Reaction of ethanol with sodium dichromate(VI) • This reaction is used in the preparation of ethanoic acid • Reaction conditions: heat, excess acidified sodium dichromate(VI) solution • The reaction mixture is refluxed in order to bring about oxidation to ethanoic acid Preparation of ethanoic acid Reflux followed by Distillation Oxidation of secondary alcohols • Secondary alcohols such as propan-2-ol are oxidised to the corresponding ketones, such as propanone • Unlike aldehydes, ketones are not easily oxidised, and so no further oxidation takes place Oxidation of propan-2-ol OH O + H3C C CH3 H Na2Cr2O7 / H H3C C CH3 Combustion of organic compounds • Most organic compounds burn in air, forming carbon dioxide and water • The structure of the compounds’ molecules is completely destroyed, with the carbon and hydrogen atoms in each molecule being oxidised • Combustion is exothermic, and ethanol is used as a fuel where it can be produced cheaply Non-flammable organic compounds • Fully halogenated alkanes such as bromochlorodifluoromethane are nonflammable • Because of this they can be used in fire extinguishers and as flame retardants • For environmental reasons, the use of many of these substances is being phased out Reduction of aldehydes and ketones • Aldehydes and ketones can be reduced to the corresponding alcohols, using hydrogen passed over the heated surface of a nickel catalyst • For example, ethanal is reduced to ethanol Reduction of ethanal to ethanol Reduction of propanone to propan-2-ol