* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Carbohydrates
Elias James Corey wikipedia , lookup
Discodermolide wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Wolff rearrangement wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Wolff–Kishner reduction wikipedia , lookup
Hydroformylation wikipedia , lookup
Asymmetric induction wikipedia , lookup
Petasis reaction wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
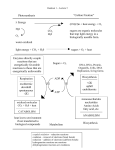
WADE CHAPTER TWENTY-SEVEN OUTLINE: CARBOHYDRATES Skip: 23-17:22 MONOSACCHARIDES 1. Ketoses (Sugars with Ketone groups) vs Aldoses (Sugars with Aldehyde groups) 2. Fischer projections a. Aldose D family: triose – tetrose - pentose – hexose (Figure 23-3) b. Erythro and Threo: History as hydroxyl groups on same or opposite side of Fischer projection in tetroses (Section 23-4) c. D and L :History as Degradation to (+) or (-) glyceraldehyde respectively (for glyceraldehyde: D is R and L is S for this case; In fischer projection D has OH of penultimate carbon on right wheras L has OH of penultimate carbon on left) 3. Epimers: Sugars that differ at just one carbon 4. Anomers: Sugars that differ at just C-1 5. Cyclic Structure: Equilbrium favors Hemiacetal (via intramolecular rxn) over open form (Section 23-6) a. Pyranose: Six membered cyclic hemiacetal b. Furanose: Five membered cyclic hemiacetal 6. Haworth projections 7. Mutarotation: Equilibration of Anomers 8. Glycosides: Acetals that do not undergo mutarotation 9. Reactions of monosaccharides a. Rxn under basic conditions o Formation of enols: Epimerization and Enediol rearrangement (Mechanisms 23-2 & 23-3) o Rxn with alcohols in basic medium (sugar as nucleophile at every OH) b. Oxidation o With Br2/H2O to form aldonic acids from aldoses o with Ag(I): “Reducing Sugars” (Section 23-10, 23-11) 1. A test for open form of hemiacetals 2. Glycosides: Sugars in the form of acetals c. Reduction of open form C=O with NaBH4 d. Rxn with alcohols in acid medium (sugar as electrophile at C1 only) to form -glycoside e. Reaction with electrophiles like CH3I or acetic anhydride (sugar as nucleophile at every OH) (Section 23-12) f. Ruff-Degradation (Section 23-14) o Br2/H2O to oxidize to carboxylic acid o Further oxidation to CO2 and shorter aldose with H2O2, Fe(III) g. Kiliani-Fischer Synthesis: Adding a carbon (Section 23-15) o Nucleophilic attack on aldose CHO with NaCN o reduction of nitrile to imine with H2 /Pd-BaSO4 o hydrolysis of imine to aldehyde LEARNING OUTCOMES: Identify reducing sugars. Draw glucose in Fischer, Haworth and chair perspectives. Draw Fischer projections for given monosaccharides. Recognize the stereochemical relationship between isomeric sugars. Predict the product of sugars with acetic anhydride, alcohols and amines, oxidizing and reducing agents. Predict the likelihood of a sugars’ mutarotation under given conditions. SAMPLE EXAM PROBLEMS: 1. Give the relationship between the two structures shown below as ANOMERS, ENANTIOMERS, DIASTEREOMERS or alternative representations of the SAME molecule 2. Fill in any missing atoms in the Fischer representation of the following sugar: 3. Draw the product(s) of the following reaction. Include stereochemistry where appropriate: