* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Dalton Model of the Atom - Teach-n-Learn-Chem
Organic chemistry wikipedia , lookup
Condensed matter physics wikipedia , lookup
Metallic bonding wikipedia , lookup
Nuclear transmutation wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Strengthening mechanisms of materials wikipedia , lookup
Electron configuration wikipedia , lookup
Periodic table wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Crystal structure of boron-rich metal borides wikipedia , lookup
Metalloprotein wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Stoichiometry wikipedia , lookup
Abundance of the chemical elements wikipedia , lookup
Isotopic labeling wikipedia , lookup
Chemical element wikipedia , lookup
Molecular dynamics wikipedia , lookup
Extended periodic table wikipedia , lookup
Chemical bond wikipedia , lookup
History of chemistry wikipedia , lookup
Chemistry: A Volatile History wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
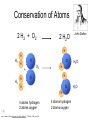
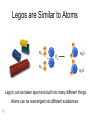
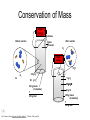
Foundations of Atomic Theory Law of Conservation of Mass Mass is neither destroyed nor created during ordinary chemical reactions. Law of Definite Proportions The fact that a chemical compound contains the same elements in exactly the same proportions by mass regardless of the size of the sample or source of the compound. Law of Multiple Proportions If two or more different compounds are composed of the same two elements, then the ratio of the masses of the second element combined with a certain mass of the first elements is always a ratio of small whole numbers. Conservation of Atoms 2 H2 + O2 2 H2O John Dalton H H H2 O H O2 + H2 H O H2O O H 2O H H O H H 4 atoms hydrogen 2 atoms oxygen Dorin, Demmin, Gabel, Chemistry The Study of Matter , 3rd Edition, 1990, page 204 4 atoms hydrogen 2 atoms oxygen Legos are Similar to Atoms H H2 H H O + H2 H H O2 H O H 2O H O O H H 2O Lego's can be taken apart and built into many different things. Atoms can be rearranged into different substances. Conservation of Mass High voltage electrodes Before reaction After reaction glass chamber O2 High voltage H2O O2 5.0 g H2 H2 80 0 g H2 g O2 300 g (mass of chamber) + 385 g total 45 ? g H2O 40 g O2 300 g (mass of chamber) + 385 g total Dorin, Demmin, Gabel, Chemistry The Study of Matter , 3rd Edition, 1990, page 204 Law of Definite Proportions Joseph Louis Proust (1754 – 1826) • Each compound has a specific ratio of elements • It is a ratio by mass • Water is always 8 grams of oxygen for every one gram of hydrogen The Law of Multiple Proportions • Dalton could not use his theory to determine the elemental compositions of chemical compounds because he had no reliable scale of atomic masses. • Dalton’s data led to a general statement known as the law of multiple proportions. • Law states that when two elements form a series of compounds, the ratios of the masses of the second element that are present per gram of the first element can almost always be expressed as the ratios of integers. Copyright © 2007 Pearson Benjamin Cummings. All rights reserved. Daltons Atomic Theory • Dalton stated that elements consisted of tiny particles called atoms • He also called the elements pure substances because all atoms of an element were identical and that in particular they had the same mass. Dalton’s Atomic Theory 1. All matter consists of tiny particles. Dalton, like the Greeks, called these particles “atoms”. 2. Atoms of one element can neither be subdivided nor changed into atoms of any other element. 3. Atoms can neither be created nor destroyed. 4. All atoms of the same element are identical in mass, size, and other properties. 5. Atoms of one element differ in mass and other properties from atoms of other elements. 6. In compounds, atoms of different elements combine in simple, whole number ratios. Dalton’s Symbols John Dalton 1808 Daltons’ Models of Atoms Carbon dioxide, CO2 Water, H2O Methane, CH4