* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download SUMMARY: Electrochemical Reaction.
Survey
Document related concepts
Transcript
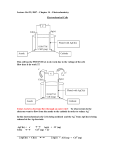
Learners must be able to define galvanic cell in terms of electrode reaction. e.g. salt bridge .(N.B. anode and cathode) Learners must be able to do the conversion of chemical energy to electrical energy. e.g. copper zinc cell. Learners must define electrolytic cell in terms of electrode reaction that are sustained by supply of electrical energy into chemical energy. e.g. porous membrane. (anode, cathode). Learners must be able to define oxidation reduction in terms of electron transfer. e.g. copper zinc cell. Anode Zn → Zn2+ + 2e- oxidation Cathode Cu2+ + 2e- → Cu reduction Relative of current and potential to rate and equilibrium Learners must be able to explain the relationship between current in an electrochemical cell and the rate of reaction. An experiment to perform using different electrodes. i.e. copper, zinc, magnesium and carbon and necessary solutions. State the pd cell related to spontaneous reaction at equilibrium. Use the qualitative relationship between v and the concentration of product and the reactants ions for the spontaneous reaction namely: v decreases while concentration of product ions increases, concentration of reactant ions decreases until equilibrium as reached at which v=o. Learners must understand the description of the movement of ions through solutions. Electron flow in the external circuit of the cell and their relation to the half reaction at the electrode . Function of the salt – bridge.