* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter 9 – Molecular Geometry and hybridization I. Molecular
Survey
Document related concepts
Transcript
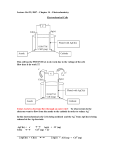
Chapter 9 – Molecular Geometry and hybridization I. Molecular Shapes II. Polarity III. sigma and pi bonds (sp hybridization) Chapter 14 - Kinetics I. Collision Theory II. Factors that affect reaction rates A. Potential energy graph B. Kinetic energy graph III. Instantaneous rate - determined from the slope of the curve (concentration vs. time) at a particular time IV. Average rate of reaction = ∆[Product]/∆time V. Reaction rates and Stoichiometry VI. Writing rate expression (rate law) Rate = k[A]m[B]n VII. Calculating rate constant with proper units VIII. Determining reaction orders from data and overall reaction order (m + n) A. Reaction order graphs IX. Reaction mechanisms A. Rate determining step B. Determining the rate law for a multistep mechanism X. Using Integrated first order rate law ln[A]t = -kt + ln[A]0 XI. Catalysts Chapter 15 – Equilibrium I. Dynamic equilibrium – characteristics of an equilibrium state II. Graphs: III. EQ constant (Kc = Keq) aA + bB Kc = [D]d [E]e [A]a [B]b IV. Magnitude of Keq dD + eE V. Le Chatelier’s Principle – predict shifts VI. EQ Law Qc = [D]d[E]e [A]a[B]b VII. “ICE” problems Chapter 16 – Acid-Base Equilibria I. II. Acid-Base Theories A. Arrhenius B. Bronsted-Lowry 1. Conjugate pairs C. Lewis Acid Reactions III. Net Ionic Equations IV. Strong & Weak Acids and Bases (Table/Figure 16.3 & Exercise 16.3) V. pH and pOH scales A. pH = -log [H+] B. [H+] = 10-pH C. pOH = -log [OH-] D. [OH-] = 10-pOH E. [H+] [OH-] = 1 x 10-14 VI. Kw : Ionization of Water A. [H+] [OH-] = Kw VII. Ka and Kb A. Ka = [H3O+] [A-] [HA] B. Kb = [BH-] [OH-] [B] Acid dissociation constant Base dissociation constant VIII. ICE problems with Ka and Kb IX. pKa and pKb relationships with strong and weak acids and bases Chapter 20 – Electrochemistry I. Assigning oxidation numbers II. Identifying oxidation and reduction III. Balancing redox half reactions IV. Activity Series Table V. Voltaic Cell 1. 2. 3. 4. 5. Spontaneous reaction where chemical energy is converted to electrical energy An Ox & a Big Red Cat – Anode is negative, Cathode is positive Metal at Anode is listed higher on Table J Electrons flow from Anode to Cathode Salt bridge – flow of ions – keeps charge from building up in one cell VI. Standard reduction potentials table and calculating voltage VII. Electrolytic Cell 1. Non-spontaneous reaction – needs outside source of energy (electrical energy is converted to chemical energy) 2. Anode is positive, Cathode is negative 3. Electrons flow from Anode to Cathode 4. Electroplating, electrolysis 5. Anode is the metal that is used for plating; Cathode is the object being plated 6. No salt bridge