* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download ATOMS
Photoelectric effect wikipedia , lookup
ATLAS experiment wikipedia , lookup
Identical particles wikipedia , lookup
Standard Model wikipedia , lookup
Grand Unified Theory wikipedia , lookup
Electric charge wikipedia , lookup
Nuclear structure wikipedia , lookup
Electron scattering wikipedia , lookup
Introduction to quantum mechanics wikipedia , lookup
Compact Muon Solenoid wikipedia , lookup
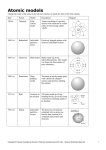
ATOMS! Unit 5 I. MODELS a. b. c. MODEL: a representation that shows the construction or appearance of something. As our technology has evolved our theories and models have also evolved. Models of the atom have changed through the years as we have discovered new things. II. Rutherford’s Model a. In 1911, Ernest Rutherford tested another scientist’s model (J.J. Thomson). b. Rutherford discovered that an atom is mostly made up of empty space. c. He said that the center of the atom is a small, dense nucleus that is positively charged. d. Rutherford said the negatively charged particles were attracted to the positively charged particles found in the nucleus. e. He said this attraction holds the negatively charged particles in the atom. f. He didn’t really address electrons. III. Bohr’s Model a. According to Bohr’s Model, there are 2 main pieces of an atom: 1. NUCLEUS 2. ELECTRONS The Bohr Model IV. What is True Today a. The nucleus is composed of 2 things: – PROTONS (positively charged particles) – NEUTRONS (neutral charged particles) b. Electrons are in a cloud – they are in orbitals, or regions of probable electron location. c. They DO move around the nucleus in different patterns. V. Wave-Mechanical Model Electrons are moving so fast that they look like a cloud. Orbitals are regions of most likely electron location. VI. Components of an Atom a. Atoms are the building blocks of EVERYTHING! b. Atoms are made up of subatomic particles • Sub means smaller or less than • Atomic means atom – So subatomic particles are the smaller particles that make up an atom. – The 3 subatomic particles are: • protons • electrons • neutrons VII. Meet the ATOM! Subatomic Particle Protons Neutrons Electrons Location in the Atom Center (in the nucleus) Center (in the nucleus) Around the nucleus in clouds or orbitals Charge Weight + (Positive) 1 amu* Neutral (no charge) - (Negative) 1 amu Almost none! It rounds to 0 amu *Amu means Atomic Mass Unit VIII. AMU a. AMU= Atomic Mass Unit b. This is the unit of measure scientist use to measure the mass of an atom. c. 1 amu = mass of 1 proton or 1 neutron. d. The mass of an electron = 1/1836 amu. PROTONS ELECTRONS • POSITIVE charges • NEGATIVE charges • Found in the • FLY around the CENTER of the nucleus in atom - aka nucleus “orbitals” or energy levels! • “HEAVY!” (1amu) • LIGHT! So light in fact, that they don’t factor in to the mass of an atom! IX. Back to the Atom…Why it Works a. Forces hold the atom together. b. Opposite charges attract and like repel! c. The negatively charged electrons are moving so fast that they stay in orbit around the positively charged nucleus. X. Remember Elements? • An element, remember, is a substance that cannot be chemically broken down into simpler things! • Elements are made up of ATOMS! • There are many of different elements - Elements are like classification of animals…. • There are about 118 known today. • They’re organized and arranged on the Periodic Table! XI. Ions a. If an atom gains or loses electrons it is called an ion. b. Gaining or losing an electron gives the atom a charge. – The charge could be positive (if the atom loses electrons) or negative (if it gains electrons). c. An atom is still the same element if it gains or loses electrons, it just now has a charge. d. We call an atom with a charge an ion. XII. Isotopes a. An isotope is when an atom is missing or has extra neutrons. b. Just like an ion, an isotope is still the same element it is just a little different from every other atom of the same element. c. For example-A carbon atom can have 12 neutrons or 14 neutrons. It is still carbon, it just has a slightly different mass. – Think of a pillow- some have more stuffing than others so their mass may be different but they are still all pillows.