* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Methods and strategies of peptide ligation
Fatty acid synthesis wikipedia , lookup
Western blot wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Molecular Inversion Probe wikipedia , lookup
Genetic code wikipedia , lookup
Oligonucleotide synthesis wikipedia , lookup
Catalytic triad wikipedia , lookup
Protein structure prediction wikipedia , lookup
Metalloprotein wikipedia , lookup
Artificial gene synthesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Biosynthesis wikipedia , lookup
Biochemistry wikipedia , lookup
Proteolysis wikipedia , lookup
Ribosomally synthesized and post-translationally modified peptides wikipedia , lookup
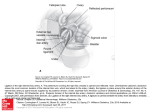
James P. Tam Jiaxi Xu Khee Dong Eom Department of Microbiology and Immunology, Vanderbilt University, A5119 MCN, Nashville, TN 37232 Methods and Strategies of Peptide Ligation* Abstract: This review focuses on the concept, methods, and strategies of orthogonal peptide ligation. It updates our previous review in 1999 on the same subject matter in Biopolymers (Peptide Science, 1999, Vol. 51, p. 311). Orthogonal peptide ligation is an amino terminal specific method to couple chemically unprotected peptides or proteins derived from synthetic or biosynthetic sources. Unlike conventional chemical methods, peptide ligation methods do not require coupling reagents or protection schemes, but are achieved through a variable chemoselective capture step and then an invariable intramolecular acyl transfer reaction. It is also a convergent method with the fewest steps. More than a dozen orthogonal ligation methods have been developed based on captures by either imine or thioester chemistries to afford native and unusual amino acids at ligation sites of linear, branched, or cyclic peptides. The ligation strategies for multiple segments including sequential and tandem ligations are also discussed. © 2001 John Wiley & Sons, Inc. Biopolymers (Pept Sci) 60: 194 –205, 2001 Keywords: imine capture; peptide ligation; peptide synthesis; protein synthesis; sequential ligation; tandem ligation; thioester capture INTRODUCTION Over the past forty years, chemical synthesis of proteins has been a major incentive in the development and refinement of solid phase peptide synthesis by Bruce Merrifield.1,2 Indeed, this goal has long been realized in Merrifield’s own laboratory and demonstrated repeatedly by many laboratories.2 During the past ten years, our laboratory has exploited the efficiency of stepwise solid phase synthesis to prepare unprotected peptides as building blocks for a convergent synthesis known as peptide ligation.3 This method couples unprotected peptide or protein seg* This paper is dedicated to the 80th birthday of Professor Bruce Merrifield Correspondence to: James P. Tam; email: tamjp@ctrvax. vanderbilt.edu Biopolymers (Peptide Science), Vol. 60, 194 –205 (2001) © 2001 John Wiley & Sons, Inc. 194 ments through an amide bond, usually under an aqueous buffer condition (Figure 1).3– 6 Conceptually, peptide ligation is based on the Prior Thiol Capture developed by Kemp et al.7 It is different from the conventional synthetic or semisynthetic methods because no enthalpic agents, enzymes, biologicals or protecting groups are used.3 Furthermore, this ligation method represents a significant methodological advance because it is orthogonal, forming an amide bond regiospecific to the desired N-terminal (NT)-amine among other free NT-amines of unprotected peptide segments present in the reaction mixture. Although orthogonal peptide ligation has also Methods and Strategies of Peptide Ligation FIGURE 1 A schematic representation of orthogonal ligation of two unprotected peptides or proteins. X and Y represent a pair of nucleophile and electrophile. been called chemoselective, capture-activated,8 native,9 intramolecular,10 or biomimetic ligation11 in the literature because it contains attributes of all or part of these descriptions, we have referred to this NT-specific coupling method as the orthogonal peptide ligation in accordance with other orthogonal concepts in chemistry, including orthogonal protection schemes12 and orthogonal activation13 and coupling14 in organic chemistry that distinguish two functional sites based on chemoselectivity. This paper reviews the concept, methods, and strategies in orthogonal ligation methods for peptide and protein synthesis. CONCEPTUAL FRAMEWORK OF PEPTIDE LIGATION Orthogonal peptide ligation generally consists of a two-reaction sequence of a chemoselective capture and an intramolecular acyl rearrangement (Figure 2). The chemoselective capture requires a nucleophile or electrophile proximally placed at an NT-amine segment and another compatible electrophile or nucleophile also proximally located at a carboxylic terminal (CT)-ester segment. The chemoselective capture of the nucleophile and electrophile pair forming an ester or a thioester brings the NT-amino group and CTester into such a close proximity to permit a spontaneous intramolecular acyl transfer to form an amide bond. In most orthogonal ligation, the products usually do not need a deprotecting step after the ligation reaction. SPECIFIC REQUIREMENTS FOR THE NT-AMINE AND CT-ACYL SEMENTS IN ORTHOGONAL LIGATION As previously discussed, there are specific requirements for placing additional functional groups as a complementary nucleophile and electrophile partners 195 in the reacting pairs of NT-amine and CT-carboxylic acid for an orthogonal ligation to occur. There are essentially two configurations. For the NT-amine (or CT-COOH), the functional group can be placed either at the side chain or at the ␣-amine (or as an ester). A common combination for an NT-amine-nucleophile is to place the nucleophile at the side chain of an NT-amino acid. This spatial configuration is found in many naturally occurring amino acids. Essentially, all ␣-amino acids containing heteroatomic side chains occupying NT positions can be considered as NTamine-nucleophiles. Since not all amino acids contain a side-chain nucleophile and since NT-amine-nucleophiles have different reactivity toward the complementary CT-acyl-electrophiles, they provide a mechanism to distinguish one NT-amine from another. Thus far, NT-Cys, Ser, Thr, His, and Trp, containing weak-base nucleophiles such as a thiol, amine, or hydroxyl group on their side chains spatially separated by two atoms from their ␣-amines, have been found to be most useful. Their function as the NTamine-nucleophiles is lost when they are placed in internal positions of a peptide sequence because their ␣-amines would be converted to amides. A different spatial configuration for an NT-amine-nucleophile is to place a nucleophile directly at the NT-amine as an N-substituted nucleophile. This spatial combination is discussed under the section describing cysteine-mimetic ligation. A second combination of a paired functional arrangement for the NT-amine is the amine-electrophile as an ␣-amine plus side-chain electrophile combination or an ␣-amino-substituted electrophile. None of the naturally occurring amino acids contains a reactive NT-amine-electrophile combination suitable for the orthogonal ligation scheme. Thus, NT-amine-electrophiles are either modified from the naturally occurring ␣-amino acids or are prepared directly from total syntheses. Examples of amine-electrophile include -arylsulfenyl- or alkoxycarbonyl sulfenyl-thiol, FIGURE 2 General concept of orthogonal peptide ligation. X and Y represent a pair of nucleophile and electrophile. Z is the bond of the capture intermediate. R1 and R2 represent side chains. In this and all subsequent figures, N, M, and C denote amino, middle, and carboxylic segments of unprotected peptides or proteins, respectively. 196 Tam, Xu, and Eom FIGURE 3 Pseudoproline ligation via imine capture for NT-Cys, NT-Ser, and NT-Thr. -bromo- and ␥-formyl amino acids, aziridine and ␣-azidoalkyl acids. For the CT-COOH, a requirement is that it must be an ester or thioester. A typical CT-acyl-electrophile is an ester or thioester with a spacer ⱕ 3 atoms linking the acyl and electrophile. Two popular arrangements are O-glycoaldehyde ester and thioester in which the electrophiles are part of the ester moiety. Their capture through either an imine or thioester by an NTnucleophile segment such as NT-Cys would result in an ester or thioester that leads to an acyl migration through a five-membered ring intermediate. A different spatial arrangement of an acyl-electrophile is the placement of the electrophile on the side chain of a CT-ester. An example is the formyl group of ␥-formyl ester. Such a CT-electrophile can react with an NTCys to form a bicyclic lactam.15 Another combination for an acyl segment is an acyl-nucleophile. The CT-thioacid with a thiolate directly linked to the CT-carbonyl group represents the simplest form of an acyl-nucleophile. CT-nucleophile is versatile and has been useful for many transformations in orthogonal ligation scheme. CT-Cys ester or its analog such as CT-Hcy (homocysteine) with the thiolate located at the side chain is also useful for the synthesis of various bicyclic analogs in the orthogonal ligation schemes. CT-phosphino ester, thioester or imidazole derivatives as the CT-nucleophiles have also been designed for Staudinger ligation. METHODS IN ORTHOGONAL PEPTIDE LIGATION All orthogonal peptide ligation methods have an invariable intramolecular acyl transfer reaction, but their capture reactions vary and can be grouped into two general reactions, imine and thioester captures. The imine capture affords an imine intermediate formed between the amine and acyl segments containing an aldehyde as an electrophile. The thioester capture produces a thioester or perthioester. These two capture methods have been developed to include a repertoire of NT-amino acids. For convenience, we have named each method according to its ligation product or the NT-nucleophile when several methods yield a common product. Peptide Ligation Methods by Imine Capture Imine ligation involves an imine capture between a CT-ester aldehyde with an NT-amine-nucleophile. In pseudoproline ligation, this imine then undergoes a rapid ring-chain tautomerization due to the addition of the nucleophile paired to the NT-amine. The resulting heterocycle facilitates an acyl transfer of the ester intermediate to form a proline-like imidic bond. The imine capture is well represented by ligation methods involving NT-amine-nucleophile of NTCys, Ser, Thr, Trp, His, and Asn, yielding a variety of mono-, bi-, and tricyclic side chains at the ligation site.3 In the Staudinger ligation, the capture results in an iminophosphorane through a CT-phosphino thioester. Pseudoproline Ligation The first pseudoproline ligation being developed is thiaproline ligation. It is also the first example that demonstrates the principle of orthogonal peptide ligation by coupling an NT-Cys segment and a CT-ester glycoaldehyde to form a thiaproline at the ligation site (Figure 3).16 –19 The capture between an NT-Cys segment and a CTglycoaldehyde ester to form a thiazolidine ester is facile and regiospecific. The specificity of three aldehyde esters reacting with all 20 NT-amino acids has been examined by a library containing 400 dipeptides under aqueous conditions.20 The fastest capture reaction is between the glycoaldehyde ester and cysteine to form a stable thiazolidine ester that is completed in ⬍l5 min at pH 5 and ⬍5 min under neutral or basic conditions. The thiazolidine ligation of NT-Cys is ⬎1000-fold faster than NT-Thr and NT–Trp. NT-Ser and NT-Asn are essentially unreacted in 72 h. This high reactivity between the CT-aldehyde and the NT-Cys under aqueous conditions provides the exclusive orthogonality of the amide ligation. Pseudoproline ligation has the advantage that it is performed under acidic conditions that Methods and Strategies of Peptide Ligation 197 FIGURE 4 Staudinger ligation via iminophosphorane for simple NT-amino acids. avoid side reactions of aldehyde with other nucleophiles present in peptides. These features make the imine capture very useful for the purpose of chemoselective ligation of two molecules or regiospecific conjugation reactions in protein chemistry, even without the acyl transfer reaction.21 After heterocyclic ring formation, the secondary amine in the formed thiazolidine ring acts as a weak base and can undergo an O,N-acyl migration through a favorable fivemembered ring transition state at pH 5 to form a proline mimic, 2-hydroxymethyl thiaproline, at the ligation site.16,17 While the ring formation is fast, the rearrangement is generally slow and constitutes the rate-determining step of the ligation. The rate of rearrangement increases with the increased pH and depends on the steric and electronic environment of neighboring components. Simple amino acids such as Gly and Ala are the preferred choices. However, other amino acids such as Leu and Val have been used successfully in the synthesis of HIV-1 protease analog.18 The effectiveness of this strategy has been confirmed by the synthesis of analogs of 50-residue transforming growth factor-␣ TGF␣16,17 and 99-residue peptide HIV-1 protease.18 The 2-hydroxymethyl thiaproline formed at the ligation site can be considered as a pseudothiaproline (SPro) structure. The enzyme activity test shows that the substitution of proline with SPro in the sequence of HIV-1 protease gives identical activity.18 Oxaproline ligation involves an NT-Ser or NT-Thr with a CT-ester glycoaldehyde to form an oxaproline at the ligation site (Figure 3).22 It proceeds through an imine capture, imine-oxazolidine ring-chain tautomerization, and then an O,N-acyl transfer reaction. Since oxazolidine has been found to be 104 times less stable than the corresponding thiazolidine analog,23,24 ring closure of the imine to oxazolidine is not a favored process because the oxazolidine ring-chain tautomerization generally favors an open imine form under aqueous conditions. Thus, in contrast to thiaproline ligation, oxaproline ligation does not proceed under aqueous conditions. However, it can be achieved under nearly nonaqueous conditions, such as 90% dimethylsulfoxide (DMSO) or dimethylformamide (DMF) in 10% acetate buffer at pH 5.5.20 However, oxaproline ligations are typically carried out under a mixture of pyridine-acetic acid (1:1, mol/mol) that has appropriate pH and solubility for unprotected segments.22 Recently, it has been confirmed that the pseudoproline ligation with an NT-Cys, NT-Ser, and NT-Thr (L configuration) segment is stereospecific and affords an R-epimer at the newly created stereocenter at the C2 carbon of the SPro or OPro at the ligation site.22 Staudinger Ligation for Simple NT-Amino Acids Recently, an exciting peptide ligation method based on the Staudinger reaction has been developed independently by two independent groups.25–28 It is a promising method for ligating simple NT-aliphatic amino acids such as Gly and Ala without side- chain functional groups. This ligation requires an NT-azido group, which can be considered to be an NT-amino electrophile and can be generated from the NT-amino acids via diazo-transfer reaction.29,30 For the CT-ester, it requires a functionalized phosphine-containing (thio)ester or acyl imidazole required for an imino capture (Figure 4). The putative mechanism of Staudinger ligation involves the formation of an iminophosphorane intermediate, an aza-phosphorous ylide, followed by nucleophilic attack of the iminophosphorane nitrogen on the thioester via a five-membered ring S,N-acyl transfer leading to an aminophosphonium salt that is hydrolyzed to produce the desired dipeptide and phosphine oxide. Thus far, successful examples of Staudinger ligation have been limited to Nacetylated amino acid thioester with NT-azido Gly derivatives under essentially organic conditions to afford model dipeptides and only in their protected forms. Pepetide Ligation Methods by Thioester Capture The CT-thioester readily reacts with a thiol through a thiolthioester exchange reaction to form a new thioester. With an NT-Cys, this thioester capture forming a new S-acyl inter- 198 Tam, Xu, and Eom FIGURE 5 Cysteine and selenocysteine ligation via thiol-thioester or selenol-thioester exchange. mediate would spontaneously undergo an S,N-acyl migration to form a peptide bond through a five-membered ring. This reaction, first reported by Wieland in 1953,31 involves a CT-thioester and a cysteine derivative that results in the formation of a cysteine-containing peptide between two amino acids. The significance of this finding went unnoticed for 40 years but recently was exploited by two laboratories for the orthogonal peptide ligation.9,32 At present, the thioester as a method of capture in the peptide ligation has been applied successfully in cysteine,9,32 cysteine mimetic,33–35 methionine,11 selenocysteine,36 and histidine37 ligation for unprotected peptides. Cys ligation possesses two ideal elements in the orthogonal ligation method. First, NT-Cys bearing a thiol readily undergoes a thiol–thioester exchange reaction with a CTthioester to produce a new thioester. After the S,N-acyl migration, it regenerates the thiol moiety and preserves the integrity of the side-chain functional group of cysteine at the ligation site. Second, the thioester as a CT-electrophile contains perhaps the most favorable spatial arrangement of these two functional groups, with the alkyl thiolate electrophile directly linked to the acylating carbonyl group. Such an arrangement provides not only an activated ester but also a highly desirable five-membered ring intermediate for an intramolecular acyl migration after the thioester capture. Consequently, cysteine ligation by thioester capture has been extensively exploited in chemical and, more recently, semisyntheses of proteins.38 – 41 There are three approaches of thioester captures for cysteine ligation and each approach is described in the following sections. Cysteine Ligation through Thioester Capture by Thiol–Thioester Exchange Cysteine ligation through capture by thiol-thioester exchange involves a CT-thioester as an electrophile and an NT-Cys as a nucleophile that produces Cys at the ligation site (Figure 5). Several conditions for ligating CT-thioester with NT-cysteine peptide have been reported. Although Cys ligation can occur under acidic conditions,32 most ligations are performed under aqueous conditions buffered at pH 7– 8 by sodium phosphate. However, in general, Cys ligation can be, and has been, performed successfully under nondena- turing conditions.42 A critical condition necessary for this ligation is the maintenance of a strong reducing environment to prevent N,S-bisacylated byproduct and reduce disulfide formation. A combination of R3P and a large excess of an alkyl thiol improved the yield from 75 to ⬎95% in a model peptide32 and was considered as an optimal condition for thiol–thioester exchange. Tri(2-carboxylethyl)phosphine (TCEP)43 is convenient to use for unprotected peptide ligation because it is water soluble and rapidly reduces the blocked disulfide forms of the NT-Cys segments to their reactive forms. There are no significant side reactions in the peptides containing side-chain functionalities such as the amino in lysine, thiol in cysteine, guanidine in arginine, and imidazole in His at internal positions of the peptide sequence. Internal thiols present in NT-Cys and CT-thioester segments are reactive moieties that can impede the reaction course in the Cys or other thioester ligation methods. Thus, ligation reactions often become sluggish when both segments contain multiple thiols competing with the NT-Cys for the thiol–thioester exchange reactions. CT-thioesters with internal thiols quickly undergo ring-chain tautomerization to form thiolactones that can often serve as the effective acylation moieties in the capture step.44 Recently, selenium analogue of cysteine, selenocysteine (Sec), which contains a selenol group on its side chain spatially separated by two atoms from its NT-amine, was also used in peptide ligation. After the ligation it generates a Sec in the ligated site.36 Cysteine Ligation through Thioester Capture by Thioalkylation Another approach for thioester capture is through thioalkylation utilizing a CT-thioacid with an NT-BrAla or NTaziridine. In this ligation method an amine-electrophile, NT--bromoalanine (BrAla), is readily S-alkylated by a thioacid to form a covalent thioester that will rearrange rapidly to give the cysteine at the ligation site (Figure 6).32 The side reaction in this ligation method is an isomer formation at the ligation site because the N-terminal BrAla peptide can readily undergo 1,3-elimination of HBr at pH ⬎ 6 to form an NT-cyclic imine, an aziridine ring. Subse- Methods and Strategies of Peptide Ligation 199 FIGURE 6 Cysteine ligation via thioalkylation capture of bromoalanine or aziridine. quent opening of the aziridine ring at the ␣ or  position by the thioacid forms the desired peptide product with the -isomer as a byproduct. However, when thioesterification was conducted at pH ⬍ 5, the aziridine formation was slow and the byproduct was minimized to ⬍3%. Interestingly, the ring opening becomes regiospecific at the  position with a steric hindered proline at the second position and only the desired ␣-peptide is obtained. A strategic advantage of the thioester capture by a CT-thioacid over a CTthioester is that it is generally performed in acidic conditions. This difference may have advantages in devising tandem ligation strategies for multiple peptide ligation and for synthesis of proteins that are not stable under basic conditions. Cysteine Ligation Through Perthioester Capture by Thiol–Acyl Disulfide Exchange Another thioester capture is found in Cys-perthioester ligation that involves an acyl disulfide intermediate in the capture step.45 An NT-amine-electrophile such as NTCys(Scm) or NT-Cys(Npys) containing an activated thiol side chain of mixed disulfides readily undergoes a thio–thiol exchange reaction with the C␣-thiocarboxylic acid of an acyl segment. The mixed acyl disulfide (perthioester) then undergoes a rapid intramolecular S,N-acyl transfer through a six-membered ring intermediate. Thiolytic reduction of the resulting hydrodisulfide (S-SH) gives a Cys residue at the ligation site (Figure 7). A characteristic of this reaction is that the capture and S,N-acyl migration can occur under acidic conditions (pH ⬍ 4). In our laboratory, we performed the reaction at pH ⬍ 2 with mixed acetonitrile–water solution, with the segment concentration of 10 ⬃ 15 mM. The high efficiency of the first capture step is attributed to the Npys-activated sulfide and to the strong nucleophilicity and low pKa of the thiocarboxylic acid compared to a normal alkyl thiol. The acyl transfer is spontaneous and 90% completed in 5 min even at pH ⬍ 4. The efficiency of this acyl transfer step is attributed to the activated acyl disulfide and proximity of the CT-acyl and NT-amine in a six-membered ring intermediate. This orthogonal peptide ligation and disulfide reduction is a one-pot process that does not require isolation of the intermediates. This method was used for the synthesis of a 32-residue peptide45 as analog of the 99-residue HIV protease from two purified segments, the acyl segment (15 residues) containing a thioacid and the amino segment containing an NT-Cys(Npys). Methionine Ligation The principle of Cys-thioester ligation has been successfully extended to form a methionine (Met) at the ligation site.11 In Met ligation the latent thiol moiety of Met as NT-homocysteine (Hcy) is exploited for the capture by transthioesterification with an acyl segment bearing a CT-thioester to give an S-acyl intermediate. The condition used for transesterification is similar to those previously described for cysteinyl peptide. After the S,N-acyl migration through a favorable FIGURE 7 Cysteine perthioester ligation. 200 Tam, Xu, and Eom FIGURE 8 Methionine ligation. six-membered ring transition state to form the homocysteinyl product and subsequent S-methylation of the ligated Hcy residue with excess methyl p-nitrobenezensulfonate, Met is obtained at the ligation site (Figure 8). Thus, this ligation method requires an additional S-alkylation step after completion of the orthogonal ligation scheme. However, it would be useful for Met-containing peptides or proteins without any cysteinyl residues. Selective methylation on Cys has been previously demonstrated by Heinrikson in 1971 with methyl p-nitrobenzenesulfonate as a methylation reagent.46 Although the thioalkylation has been carried out under basic conditions, other methods of selective S-methylation under neutral or acidic conditions are also available for transforming of Hcy to Met.47 Cysteine Mimetic Ligation for NTNoncysteine Amino Acids with N-Linked Auxiliary Thiols An NT-cysteine mimetic is an N-substituted amino acid with the thiol side chain being placed at the ␣-amine. Such a mimetic could then take part in the thioester capture to afford a modified amino acid at the ligation site.33–35 When the N-linked auxiliary thiol at the ␣-amine is removable, this method provides a highly promising approach for ligating simple NT-amino acids (Figure 9). Two different removable N-linked auxiliary thiols have been developed for the NT-cysteine mimetics, an N-linked mercaptoethoxy and a mono- or dimethoxy substituted benzyl thiol auxiliary.33,35 To reduce the N—O bond of the O-alkyl hydroxamate to give the ligation product with an amide bond, the N-mercaptoethoxy peptide product is reduced by zinc dust.33 A notable feature of this cysteine mimetic ligation is the acyl transfer rearrangement that is not observed when a bulky C␣-substituted amino acid is used on either side of the ligation site. The acid susceptibility of the benzylic thiol auxiliary groups has been modulated by introduction of electron-donating groups at the aromatic ring.35 These benzylic-protecting groups are similar in concept and design to the hydroxymethoxy benzylic groups developed previously by Sheppard’s laboratory as auxiliary groups to provide the secondary amine.48 How- FIGURE 9 Cysteine mimetic ligation with removable auxiliary thiol linker. Methods and Strategies of Peptide Ligation 201 FIGURE 10 Histidine ligation. ever, the susceptibility of these benzylic thiol auxiliary groups during the acidolytic cleavage step remains to be resolved. An N-linked arylthiol group has also been developed, but subsequent detachment of this auxiliary group from the product is not successful.34 Histidine Ligation An alternative nucleophile to thiol is the His side-chain imidazole group, a weak base under acidic conditions that can catalyze the acyl transfer in the enzymatic process.49 Ligation schemes that use NT-His as a nucleophile and a CT-thioester to form His at the ligation site are inefficient and activation of CT-ester becomes necessary.37 Thus, an acyl segment that contains a CT-thioacid that can be activated by a thiophilic promoter such as Ellman’s reagent50 has been developed successfully for the capture reaction by the imidazole of the NT-His to form an Xaa—His bond after subsequent Nim to N␣-acyl transfer (Figure 10). Under acidic conditions and in the absence of thiol nucleophiles, the imidazole moiety would be the sole nucleophile. Thus, when the ligation reaction is performed under acidic conditions in phosphate buffer maintained at pH 5.7, it is generally completed within 8 h. This method has been used to generate His-containing peptides such as calcitonin and parathyroid hormone with yields of 60 ⬃ 75%. Sequential Peptide Ligation Sequential ligation strategy is an approach to couple segments consecutively, employing a deprotection step between each ligation cycle. At present, only cysteinyl ligation is being exploited for the sequential ligation strategy. However, the cysteine-thioester method without any protecting group is limited to the ligation of two segments because the peptide bearing both the NT-cysteine and CT-thioester is prone to cyclization and polymerization. For sequential ligation the NT-cysteine of the thioester segment must be protected. This protecting group should be removable under mild aqueous conditions in accordance with the nature of STRATEGIES FOR LIGATING MULTIPLE PEPTIDE SEGMENTS In general, stepwise solid phase synthesis consistently produces unprotected peptides of 60 amino acid residues in good yield and high purity.1,2,51 With these synthetic building blocks, a single ligation step can construct a protein of 100 –120 amino acid residues. Sequential and tandem ligation strategies have been developed for ligating multiple peptide segments in which three or more peptide segments are used to afford proteins larger than 120 amino acid residues. FIGURE 11 An example of sequential ligation exploiting a photolabile protecting group. 202 Tam, Xu, and Eom FIGURE 12 N 3 C three-segment tandem pseudoproline ligation. the unprotected peptide. Photolabile- and reduction-sensitive protecting groups for NT-amines and thiols as well as thiazolidine-based phenacyl-protecting groups (Figure 11) for the NT-Cys have been developed to fulfill these requirements.52,53 These photolabile-protecting groups have been successfully applied for the sequential ligations giving a human ␣-defensin (38aa) and NK-lysin (77aa).52 Similarly, Muir and co-workers have used a base-labile Msc (2-(methylsulfonyl)ethyloxycarbonyl) as NT-protecting group.53 illustrated in preparing proline-rich peptide, bactenecin (Bac 7), to form two pseudoproline bonds, a thiaproline and an oxaproline (Figure 12).56 Ligation at the C 3 N direction demonstrated the regioselectivity of an NT-Cys segment in forming a thiaproline bond over an NT-Ser or NT-Thr segment in forming an oxaproline bond with a segment that bears a CT-glycoaldehyde ester. The successive ligations of three unprotected segments in the sequential order forms a thiaproline and then an oxaproline bond. However, ligation through the N 3 C direction is more flexible and can be extended beyond three segments. An NT-Cys, NT-Ser, or NT-Thr segment bearing a CT-glycerol ester as a masked form of CT-glycoaldehyde has been used to form a pseudoproline bond with another CT-glycoaldehyde ester segment. Oxidative conversion of the glycerol ester product to a CTglycoaldehyde ester completes a round of pseudoproline ligation with an NT-Ser, NT-Thr, or NT-Cys segment. A second strategy, a semiorthogonal tandem ligation of pseudoproline and cysteine strategy, has also been developed by employing imine and thioester captures based on the chemoselectivity of NT-Cys peptides toward two CTelectrophiles to form, in tandem, an Xaa—SPro (thiaproline) and an Xaa—Cys bond at the ligation sites.57 In addition, this tandem ligation strategy shows that the biological activity of Spro 3 Pro peptides including somatostatin-28 (Sm-28) and two proteins, human macrophage inflammatory proteins MIP-1␣ and MIP-1 is not altered by the proline 3 thiaproline substitutions in these three bioactive peptides.57 As discussed in previous sections, inter- and intramolecular cysteine-thioester ligations are best performed under basic conditions (pH ⬎ 7), whereas SPro-imine ligation prefers acidic conditions at pH ⬍ 5. Under acidic conditions, the thiol-thioester exchange between an NT-Cys and the CT-thioester is slow, while the imine capture between an aldehyde and an NT-Cys is fast. This differential reactivity Tandem Peptide Ligation With an expanded repertoire for peptide ligation methods, a tandem ligation scheme of coupling multiple segments to form two or more regiospecific amide bonds without a protection scheme would be desirable to increase the flexibility of the orthogonal ligation for forming complex peptides and proteins. Such a strategy would be different from a tandem three-segment ligation that combines chemoselective and orthogonal ligation methods to afford macromolecules with one or two nonamide linkages at both ligation sites.54,55 Recently, three successful strategies of tandem ligation of unprotected peptide segments have been reported.56 –58 The first strategy, known as tandem pseudoproline ligation, forms two pseudoproline bonds by imine captures involving the orthogonality of NT-Cys peptide and an NTSer/Thr peptide without a protecting group scheme between each ligation step. This strategy is bidirectional and is FIGURE 13 A tandem thiapseudoproline and cysteinethioester ligation. Methods and Strategies of Peptide Ligation 203 FIGURE 14 Four-segment tandem ligation. of an NT-Cys peptide under acidic and basic conditions toward two different CT-esters provides the basis for the three-segment ligation involving two NT-Cys to form two amide bonds in tandem. This tandem three-segment ligation strategy is performed in the N 3 C direction (Figure 13) on three unprotected segments to form first an Xaa–SPro that serves as an Xaa—Pro bond surrogate, and then an Xaa— Cys bond. For convenience, these three segments are also designated in the N 3 C direction as the N-segment containing a CT-glycoaldehyde ester, the M-segment containing both a CT-thioester and an NT-cysteine, and the Csegment also containing an NT-cysteine. SPro-imine ligation of the N and M segments proceeds through an imine capture of the CT-glycoaldehyde ester. A thiazolidine ester intermediate is formed, and then an O,N-acyl transfer takes place to form a C2-hydroxymethyl thiaproline at the ligation site through a favorable five-membered ring intermediate. Cys-thioester ligation between the newly formed segment and C-segment was performed consecutively (Figure 13). A third tandem ligation strategy for coupling four segments has been developed by combining chemoselective and NTspecific peptide ligation.58 This tandem ligation scheme, suitable for synthetic vaccines and combinatorial protein synthesis, couples consecutively four different segments S1–S4 to produce two amide bonds and a thioether bond at three ligation sites (Figure 14). The versatility of this new strategy has been validated in the synthesis of branched proteins of 56 –70 residues with the S1–S4 segments derived from Bac7, NK-lysin, melittin, and gp120 sequences.58 The four-segment tandem ligation scheme proceeding from the C 3 N direction generates consecutively at the ligation site Cys, Gly, and OPro. Thus, the first ligation between S1 and S2 segments to form LS1 is an orthogonal cysteine-thioester ligation. The newly generated cysteinyl thiol group of LS1 is then used for the chemoselective ligation with the chloroacetyl peptide S3 to form a branched peptide LS2 containing the side-chain thioether bond as Gly-Gly turn mimetic. Finally, the OPro ligation involves the NT serine/threonine of the LS2 segment with the peptide S4 mediates through an OPro ligation to produce the final product LS3. CONCLUSIONS AND PERSPECTIVE Since our last review on peptide ligation,3 promising new methods and practical convergent tandem ligation strategies have emerged. New concepts for ligating simple NT-amino acids such as Gly and Ala based on imine capture by Staudinger reaction and thioester capture by cysteinyl mimetics warrant further development to extend the current repertoire of ligation methods. The expansion of new peptide ligation methods also leads to novel tandem ligation strategies for coupling multiple peptide segments without the use of a protecting scheme between each ligation step. We envision that a tandem ligation strategy in a fully developed form should enable the synthesis of peptides, proteins and their mimetics of all shapes and sizes.52–58 At present, the principle of peptide ligation has already been extended for the synthesis of cyclic and branched peptides,58 – 64 and peptide mimetics. Further applications of peptide ligation concepts to peptide mimetics, organic synthesis, and combinatorial libraries can also be envisioned with existing or new combinations of amino- and acyl-nucleophiles and electrophiles. In addition, on-resin sequential or tandem ligation of the building blocks obtained efficiently by either stepwise solid phase synthesis or recombinant methods will further enhance the versatility of peptide ligation and remove the size limitation in producing synthetic or semisynthetic proteins. It is about forty years since the first public introduction of solid phase synthesis by Bruce Merrifield at the Federation Meeting of 1962 in Atlantic City.65 Applications of this enabling methodology together with ligation chemistries now make it possible to synthesize with confidence both native and synthetic peptides and proteins containing unusual amino acids 204 Tam, Xu, and Eom and architectures as well as ligating peptides to biopolymers, cell surfaces, phages, and biologicals regioselectively through an amide bond. REFERENCES 1. Merrifield, R. B. J Am Chem Soc l963, 85, 2149 –2154. 2. Merrifield, R. B. In Peptides: Synthesis, Structures and Applications; Gutte, B., Ed.; Academic Press: San Diego, CA, 1995; pp 93–168. 3. Tam, J. P.; Yu, Q.; Miao, Z. Biopolymers (Peptide Sci) 1999, 51, 311–332. 4. Dawson, P. E.; Kent, S. B. H. Ann Rev Biochem 2000, 69, 923–960. 5. Miranda, L. P.; Alewood, P. F. Biopolymers (Peptide Sci) 2000, 55, 217–226. 6. Coltart, D. M. Tetrahedron 2000, 56, 3449 –3491. 7. Kemp, D. S. Biopolymers 1981, 20, 1793–1804. 8. Tam, J. P.; Liu, C.-F.; Lu, Y.-A.; Shao, J.; Zhang, L.; Rao, C.; Shin, Y. S. In Peptides: Chemistry, Structure and Biology—Proceedings of the 14th American Peptide Symposium; Kaumaya, P. T. P., Hodges, R. S., Eds.; Mayflower Worldwide: Kingswinford, UK, 1996; pp 15–16. 9. Dawson, P. E.; Muir, T. W.; Clark-Lewis, I.; Kent, S. B. H. Science 1994, 266, 776 –779. 10. Liu, C.-F.; Rao, C.; Tam, J. P. In Peptides: Chemistry, Structure and Biology– Proceedings of the 14th American Peptide Symposium; Kaumaya, P. T. P., Hodges, R. S., Eds.; Mayflower Worldwide: Kingswinford, UK, 1996; pp 835– 836. 11. Tam, J. P.; Yu, Q. Biopolymer (Peptide Sci) 1998, 46, 319 –327. 12. Barany, G.; Merrifield, R. B. J Am Chem Soc 1977, 99, 7363–7365. 13. Kanie, O.; Ito, Y.; Ogawa, T. J Am Chem Soc 1994, 116, 12073–12074. 14. Zeng, F.; Zimmerman, S. C. J Am Chem Soc 1996, 118, 5326 –5327. 15. Hanessian, S.; McNaughton, G.; Lombart, H.-G; Lubell, W. D. Tetrahedron 1997, 53, 12789 –12854. 16. Liu, C. F.; Tam, J. P. Proc Natl Acad Sci USA 1994, 91, 6584 – 6588. 17. Liu, C. F.; Tam, J. P. J Am Chem Soc 1994, 116, 4149 – 4153. 18. Liu, C. F.; Rao, C.; Tam, J. P. J Am Chem Soc 1996, 118, 307–312. 19. Liu, C.F.; Rao, C.; Tam, J. P. In Peptides: Chemistry, Structure and Biology—Proceedings of the 13th American Peptide Symposium; Hodges, R. S., Smith, J. A., Eds.; ESCOM: Leiden, 1994; pp 1075–1077. 20. Tam, J. P.; Rao, C.; Shao, J.; Liu, C. F. Int J Pept Protein Res 1995, 45, 209 –216. 21. Shao, J.; Tam, J. P. J Am Chem Soc 1995, 117, 3894 – 3899. 22. Tam, J. P.; Miao, Z. J Am Chem Soc 1999, 121, 9013–9022. 23. Fulop, F.; Marttinen, J.; Pihlaja, K. Tetrahedron 1990, 46, 6545– 6552. 24. Baldwin, J. E. Tetrahedron 1982, 38, 2939 –2947. 25. Saxon, E.; Bertozzi, C. R. Science 2000, 287, 2007– 2010. 26. Saxon, E.; Armstrong, J. I.; Bertozzi, C. R. Org Lett 2000, 2, 2141–2143. 27. Nilsson, B. L.; Kiessling, L. L.; Raines, R. T. Org Lett 2000, 2, 1939 –1941. 28. Nilsson, B. L.; Kiessling, L. L.; Raines, R. T. Org Lett 2001, 3, 9 –12. 29. Cavender, C. J.; Shiner, V. J., Jr. J Org Chem 1972, 22, 3567–3569. 30. Pelletier, J. C.; Lundquist, J. T., IV Org Lett 2001, 3, 781–783. 31. Wieland, T.; Bokelmann, E.; Bauer, L.; Lang, H. U.; Lau, H. Liebigs Ann Chem 1953, 583, 129 –149. 32. Tam, J. P.; Lu, Y.-A.; Liu, C. F.; Shao, J. Proc Natl Acad Sci USA 1995, 92, 12485–12489. 33. Canne, L. E.; Bark, S. J.; Kent, S. B. H. J Am Chem Soc 1996, 118, 5891–5896. 34. Offer, J.; Dawson, P. E. Org Lett 2000, 2, 23–26. 35. Botti, P.; Carrasco, M. R.; Kent, S. B. H. Tetrahedron Lett 2001, 42, 1831–1833. 36. Gieselman, M. D.; Xie, L. L.; van der Donk, W. A. Org Lett 2001, 3, 1331–1334. 37. Zhang, L.; Tam, J. P. Tetrahedron Lett 1997, 38, 3– 6. 38. Muir, T. W.; Sondhi, D.; Cole, P. A. Proc Natl Acad Sci USA 1998, 95, 6705– 6710. 39. Severinov, K.; Muir, T. W. J Biol Chem 1998, 273, 16205–16209. 40. Evans, T. C.; Benner, J., Jr.; Xu, M.-Q. Protein Sci 1998, 7, 2256 –2264. 41. Evans, T. C.; Benner, J., Jr.; Xu, M.-Q. J Biol Chem 1999, 274, 3923–3926. 42. Dawson, P. E.; Churchill, M. J.; Ghadiri, M. R.; Kent, S. B. H. J Am Chem Soc 1997, 119, 4325– 4329. 43. Burns, J. A.; Butler, J. C.; Moran, J.; Whitesides, G. M. J Org Chem 1991, 56, 2648 –2650. 44. Tam, J. P.; Lu, Y.-A.; Yu, Q. J Am Chem Soc 1999, 121, 4316 – 4324. 45. Liu, C. F.; Rao, C.; Tam, J. P. Tetrahedron Lett 1996, 37, 933–936. 46. Heinrikson, R. L. J Biol Chem 1971, 246, 4090 – 4096. 47. Naider, F. R.; Becker, J. M. Biopolymers 1997, 43, 3–14. 48. Johnson, T.; Quibell, M.; Owen, D.; Shepard, R. C. J Chem Soc Chem Commun 1993, 369 –372. 49. Wieland, T.; Schneider, G. Liebigs Ann Chem 1953, 580, 159 –168. 50. Ellman, G. L. Arch Biochem Biophys 1959, 82, 70 –77. 51. Merrifield, R.B. Angew Chem Int Ed 1985, 24, 799 – 810. 52. Hennard, C.; Tam, J. P. In Peptides: Chemistry, Structure and Biology–Proceedings of the 15th Am Peptide Symposium; Tam, J. P., Kaumaya, P. T. P., Eds.; Kluwer Academic Press: New York, 1997; pp 247–248. Methods and Strategies of Peptide Ligation 53. Muir, T. W.; Dawson, P. E.; Kent, S. B. Methods Enzymol 1997, 289, 266 –298. 54. Baca, M.; Muir, T. M.; Schnolzer, M.; Kent, S. B. H. J Am Chem Soc 1995, 117, 1881–1887. 55. Canne, L. E.; Ferre-D’Amare, A. R.; Burley, S. K.; Kent, S. B. H. J Am Chem Soc 1995, 117, 2998 –3007. 56. Miao, Z.; Tam, J. P. J Am Chem Soc 2000, 122, 4253– 4260. 57. Tam, J. P.; Yu, Q.; Yang, J.-L. J Am Chem Soc 2001, 123, 2487–2494. 58. Eom, K. D.; Miao, Z.; Tam, J. P. In Peptides: Chemistry, Structure and Biology–Proceedings of the 17th American Peptide Symposium, in press. 205 59. Zhang, L.; Tam, J. P. J Am Chem Soc 1997, 119, 2363–2370. 60. Botti, P.; Pallin, T. D.; Tam, J. P. J Am Chem Soc 1996, 118, 10018 –10024. 61. Tam, J. P.; Lu, Y.-A. Tetrahedron Lett 1997, 38, 5599 – 5602. 62. Camarero, J. A.; Muir, T. W. Chem Commun 1997, 1369 –1370. 63. Camarero, J. A. Pavel, J.; Muir, T. W. Angew Chem Int Ed 1998, 37, 347–349. 64. Shao, Y.; Lu, W.; Kent, S. B. H. Tetrahedron Lett 1998, 39, 3911–3914. 65. Merrifield R. B. Fed Proc 1962, 21, 412.