* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Immunotherapy
Survey
Document related concepts
Transcript
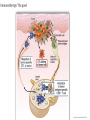
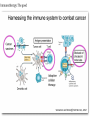
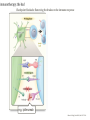
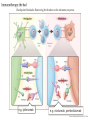
Immunotherapy: the good, the bad, the ugly GAMC Meeting Jan 2017 Why immunotherapy? Chrysoula I. Liakou, MD, PhD Disclosures: - Research Associate: Bristol-Myers Squibb - Research Associate: Sanofi Immunotherapy: The good Die krankhaften Geschwülste. (The Morbid Tumors) Virchow, Rudolph Published by Hirschwald 1863-67, Berlin, 1863. • In 1863 Virchow began publication of what was to be a comprehensive review on the subject. • It was never completed. • Virchow stopped when he reached the point where carcinoma was to be discussed, probably because of the vigorous attack he suffered from the medical community. • He described white blood cells in the tumor cells. Immunotherapy: The good Coley WB. The treatment of malignant tumors by repeated inoculations of erysipelas: with a report of ten original cases. Am. J. Med. Sci. 1893; 105: 487-511. • 1890s William Coley (1862–1936), a leading New York surgeon, had a cancer patient (sarcoma) who had a complete remission following two attacks of erysipelas caused by acute infection with the bacteria Streptococcus pyogenes. • Coley injected streptococcal cultures provided by Robert Koch into cancer patients and observed tumour regression in some cases. His findings were published in 1893, with his paper being the first that describes a serious attempt at cancer immunotherapy. • During the next 43 years Coley treated almost 900 cancer patients with his bacterial preparation which became known as ‘Coley’s toxin’. Most of the treated patients had inoperable sarcomas, with the bacterial toxin achieving a cure rate of over 10%. • Despite these successes ‘Coley’s toxin’ was not widely accepted by the scientific and clinical communities of the time, possibly due to the severe fever induced by the treatment and the perceived low cure rates. • The general feeling amongst immunologists was that it would be impossible for the immune system to recognize and respond to malignant cells. Woglom expressed this view in dramatic terms in a review in 1929 by stating that ‘It would be as difficult to reject the right ear and leave the left ear intact as it is to immunize against cancer’! (Woglom WH. Immunity to transplantable tumors. Cancer Res. 1929; 4: 129-38). • Coley’s early studies led to the use of bacille Calmette-Guérin (BCG) for cancer immunotherapy, with this treatment being used to the present day as the most effective therapy against superficial bladder cancer. Cancer Immunotherapy – Yes or No? A time line of the changing attitudes towards cancer immunotherapy. 1890s YES. William Coley’s toxin induces tumor regression in some patients. 1900–50 NO. NO - Difficult to reproduce tumor regression with Coley’s toxin. - Treatment highly toxic. - No accepted theoretical framework for cancer immunotherapy. 1950s NO. Sir Frank Macfarlane Burnet, MD, PhD. Virologist. Nobel Prize 1960. 1960s YES. Lloyd Old. MD. TNF, BCG, p53 1970–85 NO – The viral era. 1949: Burnet proposes a model for cancer immunity but when they inject cancer cells in animal models, “body recognizes cancer cells as “self”, therefore cannot reject them. The Nobel Prize in Physiology or Medicine 1960 was awarded jointly to Sir Frank Macfarlane Burnet and Peter Brian Medawar "for discovery of acquired immunological tolerance“: Failure of immunological responsiveness, that is, inability of antigensensitive cells to synthesize antibodies; induced by exposure to large amounts of an antigen. Immunological paralysis. • Old LJ, Clarke DA, Benacerraf B. Effect of Bacillus Calmette Guerin infection on transplanted tumors in the mouse. Nature 1959 Jul 25; 184: 291-292. (PMID: 14428599) • Proposed that antitumor immune responses are directed against endogenous viruses in tumors. • Charlotte Friend characterizes a murine leukemia virus, later named the Friend leukemia virus (FLV). The Friend leukemia virus is one of several discoveries leading to a resurgence in the virus theory of cancer in the 1960s and 1970s. Epstein MA, Achong BG, Barr YM. Virus particles in cultured lymphoblasts from Burkitt’s lymphoma. Lancet 1964 Mar 28; 1: 702-703. (PMID: 14107961) • 1995–2002 YES!! Ralph Steinman, MD, PhD. Immunologist, 2011 – Nobel Prize in Physiology or Medicine. James Alison, PhD. Immunologist, 2015 – Lasker De Bakey Award of Medicine. • • • • • • • 2002 – 2017 The hipster era. Dendritic cells can present tumor antigens to the adaptive immune system. Many immunodeficient mice have much higher incidences of tumors Emergence of the innate immune system as an important mediator of antitumor immunity5 ‘Stress’ induced markers can be recognized on tumor cells by the immune system ‘Immunosurveillance hypothesis’ resurrected but modified to incorporate the new view that the immune system selects for tumors that can evade immune elimination Steinman RM, Cohn ZA. Identification of a novel cell type in peripheral lymphoid organs of mice. I. Morphology, quantification, tissue distribution. J Exp Med 1973 May 1; 137: 1142-1162. (PMID: 4573839) Allison JP, McIntyre BW, Bloch D. Tumor-specific antigen of murine T-lymphoma defined with monoclonal antibody. J Immunol 1982 Nov; 129: 2293. (PMID: 6181166) NIH has 816 million funding of immunotherapy research that comes down the pipeline. Source: Cancer Research Institute. The history of cancer immunotherapy: from empirical approaches to rational, science-based therapies. Immunotherapy was declared the Breakthrough of the Year by Science Imatinib approved for CLL Treatment Treatment of of bladder cancer with bacterial products cancer with BCG (“Coley’s toxin”) 1863 1898 1957 1976 1983 1991, 4 Description of immune infiltrates Cancer immunoin tumors by surveillance Virchow hypothesis (Burnet, Thomas) Source: Cancer Research Institute Rituximab approved for NHL FDA approval of sipuleucel-T (DC vaccine) in prostate cancer Adoptive T cell therapy 2001 2002 IL-2 therapy Discovery of for cancer human tumor antigens 2008 2003 2009 2010 HPV vaccination in VIN -antiCD19 approved for NHL. -Bortezomib approved for multiple myeloma. - Cefuximab – Erbitux approved metastatic colorectal cancer. - AVASTIN colorectal cancer. FDA approval of anti-PD1 for melanoma 2013 2011 2014 2016 FDA approval of anti-CTLA4 (ipilumimab) for melanoma 1. FDA approval Pembrolizumab for head and neck cancer 2. Atezolizumab antiIgG1 bladder and lung cancer 3. First line tx of melanoma and lung cancer is immunotherapy The Carter story and the conspiracy theory March 2016, USA Today. Just months after finding out he had metastatic cancer, former President Jimmy Carter announced this weekend that his doctors have said he no longer needs cancer treatment thanks in part to a groundbreaking new kind of medication that trains the immune system to fight cancer tumors. Carter announced in August that he had melanoma that had spread to his liver and brain. He underwent surgery, radiation therapy and a new kind of cancer treatment called immunotherapy to fight the disease. Speaking at his church this weekend, Carter announced that his doctors are stopping his immunotherapy treatment called pembrolizumab after they saw no signs of tumors over a period of three months. While he has no evidence of the disease, doctors will monitor Carter closely to see if the cancer reoccurs, a representative for the former president said. "President Carter said today he did not need any more treatments, which he had August 2015 through February 2016, but will continue scans and resume treatment if necessary, Pardoll , Nature Immunol 2012. Immune phenotypes that predict better survival Analysis of 124 published articles on correlation of T cell subsets and prognosis of 20 cancer types Fridman et al. Nat Rev Cancer 12:298, 2012 Identification of tumor neoantigens Ton N. Schumacher, and Robert D. Schreiber Science 2015;348:69-74 Immunotherapy: the bad • • • • • • • • • • • Cytokine storm – T cells respond to target antigen: – Requires anti-inflammatory therapy (anti-IL-6R). – Risk of long-term damage (especially brain). Unclear how well it will work against solid tumors: – Problem of T cell entering tumor site. Will tumors lose target antigen and develop resistance? Technical and regulatory challenges of producing genetically modified CAR-T cells for each patient. Prospect of gene-edited “universal” CAR-T cells? One study found that fatigue was the most common side effect, along with fever, chills, nausea, and reactions at the site of the infusion. Patients who receive these therapies need to be monitored for potentially more serious adverse effects such as: Pneumonitis (inflammation of the lungs resulting in cough and difficulty breathing). Colitis (inflammation of the large bowel leading to diarrhea). Hepatitis and pancreatitis. Skin rashes. Endocrine disorders including thyroid abnormalities and adrenal insufficiency. Checkpoint blockade: Removing the brakes on the immune response e.g. ipilimumab Ribas A. N Engl J Med 2012;366:2517-2519. Checkpoint blockade: Removing the brakes on the immune response e.g. ipilimumab 13 e.g. nivolumab, pembrolizumab Ribas A. N Engl J Med 2012;366:2517-2519. Bernardo SG, Moskalenko M, Pan M, et al. Elevated rates of transaminitis during ipilimumab therapy for metastatic melanoma. Melanoma Res 2013; 23:47. Kim KW, Ramaiya NH, Krajewski KM, et al. Ipilimumab associated hepatitis: imaging and clinicopathologic findings. Invest New Drugs 2013; 31:1071. Chmiel KD, Suan D, Liddle C, et al. Resolution of severe ipilimumab-induced hepatitis after antithymocyte globulin therapy. J Clin Oncol 2011; 29:e237. Naidoo J, Wang X, Woo KM, et al. Pneumonitis in patients treated with anti-programmed death-1/programmed death ligand 1 therapy. J Clin Oncol 2016. Nishino M, Giobbie-Hurder A, Hatabu H, et al. Incidence of Programmed Cell Death 1 Inhibitor-Related Pneumonitis in Patients With Advanced Cancer: A Systematic Review and Metaanalysis. JAMA Oncol 2016; 2:1607. Nishino M, Ramaiya NH, Awad MM, et al. PD-1 Inhibitor-Related Pneumonitis in Advanced Cancer Patients: Radiographic Patterns and Clinical Course. Clin Cancer Res 2016; 22:6051. Corsello SM, Barnabei A, Marchetti P, et al. Endocrine side effects induced by immune checkpoint inhibitors. J Clin Endocrinol Metab 2013; 98:1361. Blansfield JA, Beck KE, Tran K, et al. Cytotoxic T-lymphocyte-associated antigen-4 blockage can induce autoimmune hypophysitis in patients with metastatic melanoma and renal cancer. J Immunother 2005; 28:593. Dillard T, Yedinak CG, Alumkal J, Fleseriu M. Anti-CTLA-4 antibody therapy associated autoimmune hypophysitis: serious immune related adverse events across a spectrum of cancer subtypes. Pituitary 2010; 13:29. Hughes J, Vudattu N, Sznol M, et al. Precipitation of autoimmune diabetes with anti-PD-1 immunotherapy. Diabetes Care 2015; 38:e55. Okamoto M, Okamoto M, Gotoh K, et al. Fulminant type 1 diabetes mellitus with anti-programmed cell death-1 therapy. J Diabetes Investig 2016; 7:915. Gullo L. Day-to-day variations of serum pancreatic enzymes in benign pancreatic hyperenzymemia. Clin Gastroenterol Hepatol 2007; 5:70. Wilgenhof S, Neyns B. Anti-CTLA-4 antibody-induced Guillain-Barré syndrome in a melanoma patient. Ann Oncol 2011; 22:991. Maur M, Tomasello C, Frassoldati A, et al. Posterior reversible encephalopathy syndrome during ipilimumab therapy for malignant melanoma. J Clin Oncol 2012; 30:e76. Bhatia S, Huber BR, Upton MP, Thompson JA. Inflammatory enteric neuropathy with severe constipation after ipilimumab treatment for melanoma: a case report. J Immunother 2009; 32:203. Liao B, Shroff S, Kamiya-Matsuoka C, Tummala S. Atypical neurological complications of ipilimumab therapy in patients with metastatic melanoma. Neuro Oncol 2014; 16:589. Johnson DB, Balko JM, Compton ML, et al. Fulminant Myocarditis with Combination Immune Checkpoint Blockade. N Engl J Med 2016; 375:1749. Gordon IO, Wade T, Chin K, et al. Immune-mediated red cell aplasia after anti-CTLA-4 immunotherapy for metastatic melanoma. Cancer Immunol Immunother 2009; 58:1351. Akhtari M, Waller EK, Jaye DL, et al. Neutropenia in a patient treated with ipilimumab (anti-CTLA-4 antibody). J Immunother 2009; 32:322. Delyon J, Mateus C, Lambert T. Hemophilia A induced by ipilimumab. N Engl J Med 2011; 365:1747. Robinson MR, Chan CC, Yang JC, et al. Cytotoxic T lymphocyte-associated antigen 4 blockade in patients with metastatic melanoma: a new cause of uveitis. J Immunother 2004; 27:478. Immunotherapy: the ugly • • • • • • “Me-too” drugs. Dr. Richard Pazdur, head of the Food and Drug Administration's office of oncology products, was referring to therapies tumors use to evade the immune system. The FDA has approved such treatments from Merck & Co, Bristol-Myers Squibb Co and Roche Holding AG , each of which have list prices of $150,000 per year. At least five other drug makers are developing similar medicines. Pazdur acknowledged that the success of a few drugmakers in the worldwide $110 billion market for cancer treatments makes it attractive for rivals to continue developing similar therapies rather than invest heavily in unproven approaches. Merck and Roche are testing their PD-1s combined with chemotherapy for the same purpose. If combinations of expensive therapies become the norm, drugmakers could theoretically keep prices lower by mixing their own treatments rather than seeking permission from a rival. New combination strategies for cancer therapy. Checkpoint blockade: prospects and challenges • Checkpoint blockade (anti-PD1/CTLA-4) + vaccination (DCs presenting tumor antigen). • Checkpoint blockade + agonist antibody against activating receptor. • Checkpoint blockade + kinase inhibitor to target oncogene. • Exploiting combinations of checkpoints – Poor biology underlying choice of combinations to block. – Difficult to reliably produce agonistic antibodies. Typically, 20-40% response rates; risk of developing resistance? • Possible biomarkers of response vs resistance: – Nature of cellular infiltrate around tumor. – Frequency of tumor-reactive (“exhausted”) T cells.