* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Protocol Sheet 2a2014
Survey
Document related concepts
Transcript
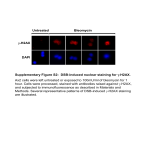
PBMC Phospho Flow Protocol www.cytobank.org Analyze multiple cell subsets and signaling pathways simultaneously in primary peripheral blood mononuclear cells (PBMCs) using this simple protocol. Biochemistry at the single cell level: enabling you to profile disease samples or monitor the effects of drugs in clinical samples. Materials Permeabilize Primary cell sample (PBMCs) Stimuli: human recombinant IL-6 and IL-10 (BD Biosciences), and lipopolysaccharide (LPS, Sigma) Phospho-specific antibodies: pStat3 Ax488 (clone 49) and p-p38 Ax647 (36) (BD Biosciences) Surface marker antibodies: CD3 PECy7 (UCHT-1), CD20 PerCPCy5.5 (H1), CD33 PE (P67.6), CD4 Pacific Blue (RPA-T4) (BD Biosciences) Tissue culture media (RPMI + 5% FBS) Staining media (PBS + 0.5% BSA) 5-mL polystyrene FACS tubes (BD Falcon) 16% paraformaldehyde in water (PFA), EM grade (Electron Microscopy Sciences) 100% methanol, cooled to 4°C Tabletop centrifuge with 5ml tube holder Flow cytometer with 405, 488, and 633 laser lines (e.g., Becton Dickinson LSRII) 5. Resuspend the cells in the residual volume left after decanting by flicking or vortexing. Then add 1 mL of cold methanol (4°C) to each tube, and vortex to mix. 6. Incubate cells at 4°C for 20 min. Methods Expected Results Stimulate The surface gating strategy and expected results are shown in the Figure below. This figure was generated in Cytobank software, with histograms and heatmaps colored according to the fold change in phosphorylation relative to unstimulated controls. LPS specifically activated p38 in the monocyte population, while IL-6 and IL-10 activated Stat3 in multiple cell subsets. 1. Thaw or prepare PBMC via Ficoll density purification. Wash twice with RPMI + 5%FBS. Rest at 10^6 cells/mL in polypropylene conical tube for 1 hour in incubator. Aliquot 1 mL cells to four FACS tubes. 2. Quickly add stimuli to cells as described in the following table: Tube # 1 2 3 Stimulus None 50ng IL-6 50ng IL-10 3. 4 1µg LPS Vortex or pipette cells to mix, and then incubate for 15 min in a 37°C, 5% CO2 incubator. Fix SSC SSC 200k Intact cells 100k 100k FSC 200k Monocytes Lymphocytes 0 103 104 CD33 PE 105 103 T cells 100k CD4- CD4+ T cells T cells 0 0 103 104 CD3 PECy7 105 9. Add 20μl pStat3, 20μl p-p38, 10μl CD3, 5μl CD4, 20μl CD20, and 5 μl CD33 antibodies. 10. Incubate cells at room temperature for 1 hour. Wash the cells twice in staining media, as in step 7. Resuspend in 100μl staining media. 11. Prepare compensation tubes for each color. Acquire data on cytometer. Interact with sample data from this experiment at: https://www.cytobank.org/cytobank/experiments/811 (If you are not yet registered to use Cytobank software, register and use it for free at www.cytobank.org) References Krutzik PO, Trejo A, Schulz KR, Nolan GP. Phospho flow cytometry methods for analysis of kinase signaling in cell lines and primary human blood samples. Methods Mol Bio (2010) in press. Krutzik PO, Nolan GP. Intracellular phospho-protein staining techniques for flow cytometry: monitoring single cell signaling events. Cytometry (2003). 55(2):61-70. Monocytes B cells CD4+ T cells CD4- T cells 1.2 0 -1.2 pStat3 Ax488 200k SSC 104 Add 50 μl staining media to the cell pellet and transfer 50μl to a fresh FACS tube. unstim IL-6 IL-10 LPS 105 B cells 8. 0 103 104 CD4 PacBlu 105 0 102 103 104 unstim IL-6 IL-10 LPS log10FC 100k Wash the cells by adding 3 mL of staining media, pelleting (>400 x g, 5 min, 4°C), and decanting the supernatant. Repeat this wash. M 200k 7. on B ocy ce te s C lls D 4+ C D Tc 4- e T lls ce lls Remove tubes from incubator and fix cells by adding 100 μL of fresh 16% PFA to each mL of media (for a final concentration of 1.5%); vortex and incubate for 10 min at room temperature. Centrifuge for 5 min at 400 x g, 4°C, and decant the supernatant. CD20 PerCPCy5.5 4. Surface and intracellular staining 0 102 103 104 0 102 103 104 pStat3 Ax488 p-p38 Ax647 Cytobank and the Cytobank Logo are property of Cytobank, Inc. © 2014 Cytobank, Inc. 0 102 103 104