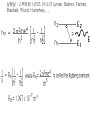* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download atomicspectra1-2
Quantum group wikipedia , lookup
Renormalization wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Particle in a box wikipedia , lookup
History of quantum field theory wikipedia , lookup
EPR paradox wikipedia , lookup
Bremsstrahlung wikipedia , lookup
Probability amplitude wikipedia , lookup
Nitrogen-vacancy center wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Quantum state wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
Franck–Condon principle wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Tight binding wikipedia , lookup
Wave–particle duality wikipedia , lookup
Chemical bond wikipedia , lookup
Electron scattering wikipedia , lookup
Magnetic circular dichroism wikipedia , lookup
Ferromagnetism wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Auger electron spectroscopy wikipedia , lookup
Renormalization group wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Hydrogen atom wikipedia , lookup
Population inversion wikipedia , lookup
Atomic orbital wikipedia , lookup
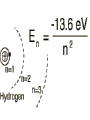
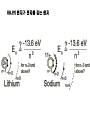
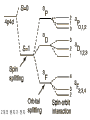
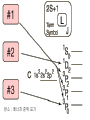
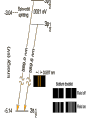
원자 구조 및 스펙트럼 • • • • • • 수소 원자 다 전자 원자. 슈레딩거 방정식 파울리 배타 원리 주기율표 LS Coupling & jj Coupling 실험실 : 스펙트럼 시리즈 (수소의 Lyman, Balmer, Pachen, Brackett, Pfund, Humphrey, … Lyman Series Balmer Series n λ(nm) n λ(nm) 2 122 3 656 3 103 4 486 4 97.2 5 434 5 94.9 6 410 6 93.7 7 397 91.1 365 Paschen Series Brackett Series n λ(nm) n λ(nm) 4 1870 5 4050 5 1280 6 2630 6 1090 7 2170 7 1000 8 1940 8 954 9 1820 820 1460 Pfund Series Humphreys Series n λ(nm) n λ(nm) 6 7460 7 12372 7 4650 8 7503 8 3740 10 5129 9 3300 11 4673 10 3040 13 4171 2280 3282 보아 모형 • • • • • • • • • • • • 전자의 각운동량(mvr)은 정수배의 h/2p mvr = n h/(2p) Coulomb force : Z e2 / (4peo r2 ) = mv2 / r r = eo n2 h2 / (p Ze2m) 궤도의 크기는 n2 에 비례 (n=주양자수) 궤도상 전자의 총에너지 = 포텐셜 에너지 + 운동에너지 = - Ze2 /(4peor) + Ze2 /(8peor) = - Ze2 /(8peor) E = - Z2 e4m / (8eo2n2h2) 에너지는 n-2 에 비례, 두 에너지 준위는 (n1-2 – n2-2 ) 에 비례 Sommerfeld의 개선 • 전자의 질량 m 환원질량 m = mM/(m+M) • 원의 궤도 타원 궤도(운동에너지 r, f) • 주 양자수 n + azimuthal 양자수 k (양자역학의 궤도 양자수 l = k-1) • 상대론적 효과 에너지 준위의 약한 k에 대한 의존성 Hydrogen Energy Levels Electron Volts 의 개념 쉬뢰딩거 방정식 파동방정식 파동 함수의 의미 수소 원자의 3 주양자 껍질에 전자 밀도 분포 파울리의 배타원리와 주기율표 • • • • • 주 양자수 n 궤도 양자수 l (k-1) 궤도 자기 양자수 mㅣ 스핀 자기 양자수 ms no two electrons in an atom may have the same four quantum mumbers, n, l, ml, ms • 원소의 주기율표 하나의 원자가 전자를 갖는 원자 one-electron atomic state • defined by the quantum numbers nlmlms or nljmj, with n and l representing the principal quantum number and the orbital angular momentum quantum number, respectively. • allowed values of n positive integers, and l = 0, 1, ..., n - 1 • The quantum number j represents the angular momentum obtained by coupling the orbital and spin angular momenta of an electron, i.e., j = l + s, so that j = l ± 1/2. • magnetic quantum numbers ml, ms, and mj represent the projections of the corresponding angular momenta along a particular direction • ml = -l, -l + 1 ... l and ms = ± 1/2. Pauli exclusion principle • prohibits atomic states having two electrons with all four quantum numbers the same • Electrons having both the same n value and l value : equivalent • the maximum number of equivalent electrons is 2(2l + 1) • parity of a configuration is even or odd : S lj is even or odd Hydrogen and Hydrogen-like Ions • A particular level is denoted either by nlj or by nl 2LJ with L = l and J = j. • The multiplicity of the L term is equal to 2S + 1 = 2s + 1 = 2.: doublet : two levels, with J = L ± 1/2, respectively • The Coulomb interaction between the nucleus and the single electron is dominant, so that the largest energy separations are associated with levels having different n • hyperfine splitting of the 1H 1s ground level [1420.405 751 766 7(10) MHz] results from the interaction of the proton and electron magnetic moments. = 21cm line Alkalis and Alkali-like Spectra • In the central field approximation there exists no angular-momentum coupling between a closed subshell and an electron outside the subshell, since the net spin and orbital angular momenta of the subshell are both zero. nlj quantum numbers are appropriate for a single electron outside closed subshells. However, the electrostatic interactions of this electron with the core electrons and with the nucleus yield a strong l-dependence of the energy levels. The spin-orbit fine-structure separation between the nl (l > 0) levels having j = l - 1/2 and l + 1/2, respectively, is relatively small. 원자가(valance) 전자 하나인 알카리 금속 Hydrogen-like atoms Other terms Hyperfine structure Closed shell Terms Equivalent electrons Terms Helium and Helium-like Ions; LS Coupling • condition for LS coupling • (a) The orbital angular momenta of the electrons are coupled t give a total orbital angular momentum L = S ili. • (b) The spins of the electrons are coupled to give a total spin S = Si si. • combination of a particular S value with a particular L value , a spectroscopic term 2S+1L.(2S + 1 is the multiplicity of the term) • total angular momentum, J = S + L : level is denoted as 2S+1LJ. • For 1s2 nl configurations, L = l and S = 0 or 1, i.e., the terms a singlets (S = 0) or triplets (S =1) • ionization energy 24.5874 eV, the 1s2s 3S - 1S separation is 0.7962 eV, the 1s2p 3P° - 1P° separation is 0.2539 eV, and the 1s2p 3P°2 - 3P°0 fine-structure spread is only 1.32 × 10-4 eV. wavelengths (nm) 438.793 w 443.755 w 447.148 s 471.314 m 492.193 m 501.567 s 504.774 w 587.562 s 667.815 m s=strong, m=med, w=weak LS Hierarchy of Atomic Structure in LS Coupling • Atomic structural hierarchy in LS coupling and names for the groups of all transitions between structural entities. • Structural entity Quantum numbers a Group of all transitions • Configuration (nili)Ni Transition array • Polyad (nili)Ni g S1 L1 nl S L, S L... Supermultiplet • Term (nili)Ni g S L multiplet • Level (nili)Ni g S L J line • State (nili)Ni g S L J M Line component • a The configuration may include several open subshells, as indicated by the i subscripts. The letter g represents any additional quantum numbers, such as ancestral terms, necessary to specify a particular term. Ca I 3d4p 3D°2 level belongs to the 3D° term which, in turn, belongs to the 3d4p 3(P° D° F°) triplet triad 3d4p configuration also has a 1(P° D° F°) singlet triad. 3d4s configuration has only monads, one 1D and one 3D 3d4s 3D2 - 3d4p 3D°3 line belongs to the corresponding 3D - 3D° triplet multiplet, this multiplet belongs to the great Ca I 3d4s 3D - 3d4p 3(P° D° F°) supermultiplet of three triplet multiplets 3d4s - 3d4p transition array includes both the singlet and triplet supermultiplets, as well as any (LS-forbidden) intercombination or intersystem lines arising from transitions between levels of the singlet system and those of the triplet system Allowed Terms of Levels for Equivalent Electrons • LS Coupling • two nonequivalent groups of electrons coupling the S and L vectors of the groups in all possible ways, and the procedure may be extended to any number of such groups. • The configuration l N has more than one allowed term of certain LS types if l > 1 and 2 < N < 4l (d 3 - d 7, f 3 - f 11, etc.). • LS term type from d N and f N : tables of Nielson and Ko ster Equivalent electrons • jj Coupling • The allowed J values for a group of N equivalent electrons having the same j value, ljN, are given in the table below for j = 1/2, 3/2, 5/2, and 7/2 (sufficient for l < 3). • Eg : (6p2 ½)0 Allowed J values for ljN equivalent electrons (jj coupling). • • • • • • • • • • • • • • • l jN Allowed J values l1/2 ½ l 21/2 0 l3/2 and l 33/2 3/2 l23/2 0, 2 l 43/2 0 l5/2 and l 55/2 5/2 l 25/2 and l 45/2 0, 2, 4 l 35/2 3/2, 5/2, 9/2 l 65/2 0 l7/2 and l 77/2 7/2 l 27/2 and l 67/2 0, 2, 4, 6 l 37/2 and l 57/2 3/2, 5/2, 7/2, 9/2, 11/2, 15/2 l 47/2 0, 22, 42, 24, 44, 5, 6, 8 l 87/2 0 Allowed levels • The allowed levels of the configuration nl N may be obtained by dividing the electrons into sets of two groups nlQl+1/2 nlRl-1/2 , Q + R = N. • The possible sets run from Q = N - 2l (or zero if N < 2l) up to Q = N or Q = 2l + 2, whichever is smaller. Notations for Different Coupling Schemes • LS Coupling (Russell-Saunders Coupling) • jj Coupling • J1 j or J1 J2 Coupling • J1l or J1L2 Coupling (J1K Coupling) • LS1 Coupling (LK Coupling) • http://sed.nist.gov/Pubs/AtSpec/total. html Coupling Schemes and Term Symbols • Coupling • Scheme • Quantum number for vectors that coupl to give J • • • • L, S J1,, J2 K, S2 K, S2 LS J1J2 J1L2(-> K) LS1(->K) Term Symbol 2S+1L (J1,, J2 ) 2S2+1 [K] 2S2+1 [K] 2개의 원자가 전자 탄소 ; 에너지 준위 표기 Zeeman Effect • "weak" magnetic fields (the anomalous Zeeman effect) : split into magnetic sublevels : -J, -J + 1, ..., J: =M DE = gM µBB • magnetic flux density is B, and µB is the Bohr magneton (µB = e /2me). • wavenumber shift Ds corresponding to this energy shift is Ds = gM(0.466 86 B cm-1) 자기장에 의한 영향 g value of a level bJ belonging to a pure LS-coupling term g value for a pure electron spin as ge Term Series, Quantum Defects, and Spectral-line Series • hydrogenic (one-electron) ion Zc is the charge of the core and n* = n -d is the effective principal quantum number Sequences • Isoelectronic Sequence :A neutral atom and those ions of other elements having the same number of electrons as the atom comprise an isoelectronic sequence : the Na I isolectronic sequence. • Isoionic, Isonuclear, and Homologous Sequences : • An isoionic sequence comprises atoms or ions of different elements having the same charge. • The atom and successive ions of a particular element comprise the isonuclear sequence for that element • The elements of a particular column and subgroup in the periodic table are homologous :C, Si, Ge, Sn, and Pb atoms belong to a homologous sequence having np2 Selection Rules Emission Intensities (Transition Probabilities) • The total power e radiated in a spectral line of frequency n per unit source volume and per unit solid angle is • Aki is the atomic transition probability and Nk the number per unit volume (number density) of excited atoms in the upper (initial) level k • For a homogeneous light source of length l and for the optically thin case, where all radiation escapes, the total emitted line intensity 숙제 3 • 수소의 Ha 가 n=3 -> n=2 로 천이되 는 모든 경우를 고려하여 선택 규율을 따르는 모든 천이를 에너지도를 그려 표 시하시오. • 또 Zeeman 효과가 있을 경우 천이를 모두 도표로 그리고, 선의 분리를 보이 시오. Absorption f values fik is the atomic (absorption) oscillator strength (dimensionless). Line Strengths Y i and Y k are the initial- and final-state wave functions and Rik is the transition matrix element of the appropriate multipole operator P (Rik involves an integration over spatial and spin coordinates of all N electrons of the atom or ion). Relationships between A, f, and S • The relationships between A, f, and S for electric dipole (E1, or allowed) transitions in SI units (A in s-1, l in m, S in m2 C2) units (A in s-1, in Å, S in atomic units), • S and E in atomic units 천이 확율(Transition Probabilites) • • • • • • • • • • 선과 multiplet의 상대적 세기 <= 천이 확율 <= 흡수, 방출계수 천이 확율 - 들뜸 에너지 준위의 수명 Life time of the transition = inverse of the sum of the transition probabilities life time 10-8s : allowed transitions 10-5s : intercombination transitions > 10-3s : forbidden transitions 21cm - 11 *106 years 아인슈타인 천이 확률 • Spontaneous Emission Transition Probability • = reciprocal of the lifetime of the transition • = A21 ( 108 s-1 for allowed • 10-15 s-1 for most extremely forbidden) • • • number of spontaneous transition /time/volume • = N2 A21 • N2 = number density of atoms in upper level • Absorption Transition Probability • = need radiation • propotional to the radiation intensity • • • number of absorptions/time/volume • = N1 B12 I21 • • B12 = absorption transition probability from 1 to 2 • N1 = number Density of atoms in level 1 • I21 = intensity of radiation (emitted 2->1) Negative Absorption = Stimulated Emission • =>LASER (Light Amplification by Stimulated Emission of Radiation) • =>MASER (Microwave Amplificaton by Stimulated • Emission of Radiation) • number of stimulated emisions/time/volume • =N2 B21 I21 • B21 = Stimulated Emission Trasition Probability from 2 to 1 • ==> photons added to the radiation field with the same direction, polarization and phase as those of the stimulating photons • cf: photons emitted spontaneouly ==> have random Principle of Detailed Balancing in TE • • • • • • =every process balanced by its inverse =every upward transition have a downward transtion occurring nearby and nearly simultaneously ==> total number of absorptions = total number of emissions • N1 B12 I21 = N2 B21 I21 + N2 A21 • TE : radiation field = BB radiation • = Plank Function TE : radiation field = BB radiation = Plank Function • 여기서 m 는 매질의 굴절율로 보통 1 근처의 값 A21 = (N1/N2 B12 - B21) I21 = 아인슈타인 계수 • transition probability 는 원자의 성질이므 로 환경의 특성인 온도에 무관 주파수가 높아지면 ( 보통 적외선 보다) stimulated emission 은 무시될 수 있다. 이로써 maser가 성간 가스 운에서는 발생하지만 높은 주파수의 laser는 발생하지않는 것을 보게된다. • A21 B21 B12 • HI 1215(Ly ) 6*108 2.8*1012 1.4*1012 • 1025(Ly ) 1.7*108 4.5*1011 2.2*1011 • 972(Ly ) 6.8*107 1.6*1011 7.9*1010 • OI 3947 3.7*105 5.7*1010 8.0*1010 • MgI 4571] 2.1*102 5.1*107 1.7*107 • [NI 5198] 1.6*10-5 5.7 5.7 • 흡수와 방출 계수 • 아인슈타인 천이 확율은 각 천이가 일어날 확율을 결정하므로 스 펙트럼의 흡수와 방출선의 결과가 된다 • 고전 회전자 - 약한 선의 경우 흡수 계수 :선윤곽 • 질량 흡수 계수 • • • • • N : 흡수 회전자의 개수 밀도 m, e : 회전자(전자)의 질량과 전하 no : 공명 주파수 (즉 흡수선의 중심 주파수) g: 회전자 복사의 감쇄 상수 양자 역학:진동자 세기(Oscillator Strength) • Damping 상수는 준위 수명과 관계가 고전과 다소 다르다 • 허가 천이의 경우 값이 108 로 가시 영역 천이의 g정도다. • n lower than 준위 1, m higher than 준위 1 • G1 도 유사한 관계를 갖음 • Thomas-Reiche-Kuhn Sum Rule • N 대신 Nf 로 대치 : • f는 고전을 양자 역학적 값으로 환원하는 보정 계수인 진동자 세기 ( Oscillator Strength) • 관측되는 선세기를 만드는 고전 진동자의 수 (보통 쪼각) • 따라서 한 원자 또는 이온에서 한 준위에서 발 생되는 모든 가능한 천이에 대한 진동자 세기 를 전부 합한 것(방출의 진동자 세기는 음으로 취급)은 원자나 이온의 전자 수와 같아진 다.==> Thomas-Reiche-Kuhn Sum Rule 발머선의 진동자 세기 ( Oscillator Strength) • • • • • • • • • • • • Bamer series n=2 Ha 0.637 Hb 0.119 Hg 0.044 Hd 0.021 He 0.012 ... 흡수 0.866 이온화 0.238 방출(Lyman ) -0.104 총합(수소에 전자수) 1.000 양자역학적 자연 흡수 계수 선의 반폭 = G/2p 이며, G 가 선에 따라 달라지므로 고전적 결과와 달리 양자영학적 흡수 계수는 선에 따라 달라진다. Oscillator Strength 와 아인슈타 인 천이 확률계수들과의 관계 • 두 준위 사이의 흡수와 방출의 진동자 세기 (Oscillator Strength)는 두 준위의 통계 가중치 와 다음과 같이 관련된 다. • • Oscillator Strength 는 천이 확율과의 관계 • 천이 확율과 흡수 및 방출 계수의 관계 자연 선 윤곽의 형태 함수 stimulated emission, negative 흡수 포함 방출 계수 Kirchhoff's 법칙