* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Print - Circulation
Heart failure wikipedia , lookup
Cardiac contractility modulation wikipedia , lookup
Saturated fat and cardiovascular disease wikipedia , lookup
Drug-eluting stent wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Electrocardiography wikipedia , lookup
Cardiovascular disease wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
Quantium Medical Cardiac Output wikipedia , lookup
Cardiac surgery wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
Ventricular fibrillation wikipedia , lookup
Coronary artery disease wikipedia , lookup
Arrhythmogenic right ventricular dysplasia wikipedia , lookup
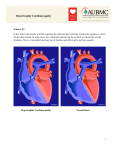
ORIGINAL ARTICLES Sudden Death in Young Athletes BARRY J. MARON, M.D., WILLIAM C. ROBERTS, M.D., HUGH A. MCALLISTER, M.D., DOUGLAS R. ROSING, M.D., AND STEPHEN E. EPSTEIN, M.D. Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 SUMMARY The causes of sudden and unexpected death in 29 highly conditioned, competitive athletes, ages 13-30 years, are summarized. Sudden death occurred during or just after severe exertion on the athletic field in 22 of the 29 athletes. Structural cardiovascular abnormalities were identified at necropsy in 28 of the 29 athletes (97%), and in 22 (76%) were almost certainly the cause of death. The most common cause of death in this series was hypertrophic cardiomyopathy, which was present in 14 athletes. Other cardiovascular abnormalities that occurred in more than one athlete were anomalous origin of the left coronary artery from the right (anterior) sinus of Valsalva, idiopathic concentric left ventricular hypertrophy, coronary heart disease and ruptured aorta. Cardiac disease was suspected during life in only seven of the 29 patients, and in only two of the seven was the correct diagnosis made clinically. Hence, in this series of young athletes, sudden death was usually due to structural cardiovascular disease, and hypertrophic cardiomyopathy was a frequent cause of sudden death; atherosclerotic coronary heart disease was relatively uncommon. ALTHOUGH YOUNG, competitive athletes epitomize the conditioned, healthy segment of society, sudden and unexpected death does occur in such individuals.1-4 However, no large series of sudden deaths in athletes has been systematically compiled, and therefore the principal causes of such deaths have not been adequately defined. Because such information might help to identify athletes who are "at risk" of sudden death, the present study was undertaken. In this investigation we have attempted to determine the causes of sudden, unexplained death in a group of highly competitive athletes. initially referred to the NHLBI for consultation because of the occurrence of sudden death and suspicion of heart disease; subsequently these patients were identified as having been competitive athletes during life. Six of the 29 study patients have been included in published studies.2 5, 6 For inclusion in the study each patient must have been an active and highly conditioned member of an organized athletic team for at least 2 years at the junior high school, high school, collegiate or professional level. Four of the 29 athletes had withdrawn from organized team sports 1-5 years before death, but had continued to maintain a high level of conditioning. It also was necessary that a complete necropsy examination be performed in each patient to exclude trauma-related causes of death and that, in particular, the heart be in suitable condition to permit examination. Toxicologic tests for levels of alcohol, barbiturates and amphetamines were performed postmortem on blood from 15 patients, and the levels of each were normal. None of the 29 athletes had a history of drug usage. Materials and Methods Selection of Patients The 29 athletes with sudden and unexpected death who constitute the study group were initially identified from the following sources: (1) prospectively from news media reports of sudden death in competitive athletes from 1976-1979 (17 athletes: two died in the Washington D.C. metropolitan area and the other 15 died elsewhere); (2) retrospectively from review of the autopsy registry of the Cardiovascular Division of the Armed Forces Institute of Pathology, 1950-1978 (seven athletes who were military dependents and whose hearts were referred for consultation because of the occurrence of sudden death and suspicion of heart disease); and (3) retrospectively from review of the autopsy registry of the Pathology Branch, National Heart, Lung, and Blood Institute (NHLBI) (five athletes). The hearts of four of these patients had been Necropsy Studies In addition to gross visual examination, measurements were made of the maximum thickness of the ventricular septum and posterior left ventricular free wall. Trabeculae, papillary muscles and crista supraventricularis were not included in the measurement of wall thickness. The extramural coronary arteries were studied in the intact specimen by making multiple cross sections of the vessels (3-5 mm apart) and viewing the arterial lumens grossly. Blocks of myocardium were taken from the ventricular septum for histologic study of cardiac muscle cell arrangement. When pertinent, the extent of cellular disorganization was assessed in tissue sections by a previously described quantitative method.7 In four athletes (three with idiopathic concentric From the Cardiology and Pathology Branches, NHLBI, NIH, Bethesda, Maryland, and the Department of Cardiovascular Pathology, Armed Forces Institute of Pathology, Washington, D.C. Address for correspondence: Barry J. Maron, M.D., Cardiology Branch, Building 10, Room 7B-15, NHLBI, Bethesda, Maryland 20205. Received October 24, 1979; revision accepted January 30, 1980. Circulation 62, No. 2, 1980. 218 SUDDEN DEATH IN YOUNG ATHLETES/Maron et al. left ventricular hypertrophy and one with hyper- trophic cardiomyopathy), the heart was in suitable condition to permit removal of the sinoatrial and atrioventricular node areas for histopathologic study of the conduction system.8' 9 Serial sections 6 m thick were cut from each conduction system block; every tenth section was stained with hematoxylin and eosin or elastic van Gieson. Echocardiographic Studies M-mode echocardiograms were obtained in a total of 75 first-degree relatives (ages 4-58 years, mean 28 years) of 15 athletes in whom the necropsy diagnosis was hypertrophic cardiomyopathy or idiopathic concentric (symmetric) left ventricular hypertrophy. Thicknesses of the ventricular septum and posterior left ventricular free wall were measured as previously described.'0 219 curred during or just after severe exertion. Each of these 22 deaths occurred during an athletic contest or during training. Two of the remaining seven patients died during or just after relatively mild exertion unrelated to participation in sports, and five athletes died during sedentary activities. Necropsy Findings: Causes of Sudden Death Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 Structural cardiovascular alterations were identified the 29 athletes (97%), but no significant anatomic abnormality was found in an organ other than the heart (fig. 1). The remaining patient, -a 14-year-old high school football player, had a normal heart by both gross and histologic examination. ECGs in five first-degree relatives of this athlete showed no abnormalities (including no evidence of prolonged QT interval syndrome), but a systematic examination of the conduction system was not possible in his heart. at necropsy in 28 of Results Demographic Data and Circumstances of Death The 29 athletes ranged in age from 13-30 years (mean 19 years). Twenty-six athletes were male and three were female; 20 were white and nine were black. Body surface areas ranged from 1.7-2.4 m2 (average 2.0 m2). At the time of death the athletes had most commonly reached the high school or junior high school level of competition (17 athletes), although eight were participating in intercollegiate sports programs and four were professional athletes. The athletes had participated in a wide variety of sports, but basketball and football were most common (table 1). The duration of participation at competitive levels ranged from 2-18 years. In 28 of the 29 athletes, death occurred suddenly without warning and was virtually instantaneous; the other athlete survived 12 hours in a terminal state after her collapse. In 22 of the 29 athletes, death ocTABLE 1. Sports Participated in by 29 Competitive Athletes n* Sport 11 Basketball Football 10 Running (track) 4t 3 Wrestling 1 Swimmring 1 Boxing 1 Hockey 1 Soccer 1 Tennis 1 Baseball 1 Gymnastics *Each sport was tabulated for athletes participating in more than one. tIncludes one athlete who participated primarily in the pole vault. Patients with Unequivocal Evidence of Cardiovascular Disease Of the 28 athletes with structural cardiovascular alterations, 22 were considered to have unequivocal cardiovascular disease that caused sudden death (fig. 1); the diseases were congenital in 19 of the 22 patients and acquired in the other three. Hypertrophic Cardiomyopathy The cardiovascular disease most commonly identified was hypertrophic cardiomyopathy, which was present in 14 athletes (figs. 1 and 2; table 2). Each of these patients had a hypertrophied, nondilated left ventricle in the absence of another cardiac or systemic disease that could produce left ventricular hypertrophy (such as valvular, hypertensive or coronary heart disease or infiltrative cardiomyopathy).11 For the diagnosis of hypertrophic cardiomyopathy to be made, each of these patients also had to have at least one of the following characteristic features of the disease: (1) asymmetric septal hypertrophy (ventricular septal-to-left ventricular free wall thickness ratio > 1.3; range 1.3-2.2);1o 12, 13 (2) marked disorganization of cardiac muscle cells in the ventricular septum,7' 14 involving at least 5% of the relevant areas of the tissue section7; and (3) clinical or echocardiographic evidence of hypertrophic cardiomyopathy in one or more closely related family members.", 16 Seven of the 14 athletes with hypertrophic cardiomyopathy had both asymmetric septal hypertrophy and marked ventricular septal disorganization and three had asymmetric septal hypertrophy without disorganization. The other four athletes had concentric (symmetric) left ventricular hypertrophy (septal-free wall ratio < 1.3), including two with marked septal disorganization and two without disorganization; each had echocardiographic evidence of hypertrophic cardiomyopathy in relatives. The heart weights in these 14 patients were all increased over normal17 and ranged from 360-630 g 220 CIRCULATION VOL 62, No 2, AUGUST 1980 1Lnan,neiv2cal CV Disease, 22 1 Idiopathic Hypoplastic Concentric LVH Coronaries ~~~ Disease Ruptured Atherosclerotic Anomalous Hypertrophic Aorta Origin of Cardiomyopathy Coronary Disease Left Coronary Artery Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 FIGURE 1. Diagram summarizing the cause of death in 29 competitive athletes. CV= cardiovascular; L VH = left ventricular hypertrophy. *Includes one patient who also had anomalous origin of the left coronary arteryfrom the anterior sinus of Valsalva. The hearts offour other patients in this subgroup were initially referred for evaluation because of suspected cardiac disease; subsequently, these patients were identified as having been competitive athletes during life. tlncludes one patient who had gross and histologic findings at necropsy consistent with mitral valve prolapse, as well as a family history of that condition. This patient (as well as one other with idiopathic concentric left ventricular hypertrophy) also had marked narrowing of the artery to the atrioventricular node. (mean 475 g). None of the 12 patients in whom blood pressure recordings were available had systemic hypertension. Mild-to-moderate right ventricular dilatation was also present in five patients. Abnormal intramural coronary arteries (two or more per tissue section) were identified in the ventricular septal myocardium of four athletes. The abnormal arteries had thickened walls (due to intimal proliferation or medial hypertrophy or both) and narrowed lumens.'8 1' One other athlete with hyper- FIGURE 2. Heart of a 13-year-old male football and baseball player showing disproportionate thickening of the ventricular septum (VS) with respect to the posterior left ventricular free wall (L V). R V = right ventricular wall. trophic cardiomyopathy showed marked narrowing of the arteries to the sinoatrial and atrioventricular nodes due to fibromuscular hyperplasia (figs. 3A-C); the conduction system was otherwise normal, as were the other intramural coronary arteries. Anomalous Origin ofthe Left Coronary Artery Four athletes had anomalous origin of the left coronary artery from the right (anterior) sinus of Valsalva,2-4, 20-22 including one of the patients with hypertrophic cardiomyopathy. Three were 17 years old and the fourth was 22 years old. In these patients the left coronary artery made an acute leftward and posterior angulation from its origin in the right sinus of Valsalva and followed a course through the narrowed area between the aorta and pulmonary artery (fig. 4); the left main coronary artery appeared to be narrowed and obstructed by virtue of its oblique course. In each of the four patients with anomalous origin of the left coronary artery, the distribution of both right and left coronary arteries was normal, and the entire coronary tree was free of significant atherosclerosis. Coronary Heart Disease Three patients had acquired abnormalities of the coronary arteries. These patients were 24, 26 and 28 years of age. Two of the three patients showed severe atherosclerosis in each of the three major extramural coronary arteries (> 75% cross-sectional area luminal narrowing by atherosclerotic plaque). One of these SUDDEN DEATH IN YOUNG ATHLETES/Maron et al. 221 Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 TABLE 2. Necropsy Findings in 19 Athletes with Hypertrophic Cardiomyopathy or Idiopathic Concentric Left Ventricular Hypertrophy Ventricular wall thicknesses Heart VS Age weight LVFW¶ Thickened % VS ASH in AML Pt (years) Sex (g) (mm) (mm) VS/LVFW VS plaquet disorganized7 relatives* Hypertrophic cardiomyopathy M SD 21 14 13 450 1.5 0 0 49 + M 18 DM 14 390 8 2.2 + + 0 + MM M 14 360 18 12 1.5 0 0 53 + AF 17 F 440 15 8 1.9 0 0 14 + M PB 17 530 21 12 1.8 + 0 75 0 M JG 17 370 16 10 1.6 0 0 49 M 0 JD 17 14 460 21 1.6 + + M 17 490 23 18 1.3 0 + 13 + SWt M AJ 19 475 23 20 1.2 + 0 + + M EW 20 630 30 25 1.2 + + 6 + 22 M WD 0 89 500 21 0 13 1.6 + OB M 23 0 0 500 20 15 1.3 + + M WH 24 0 490 15 15 1.0 0 0 + KK 30 F 530 17 15 1.1 0 0 24 Idiopathic concentric LVH M RS 0 16 440 16 16 1.0 0 0 0 M GL 16 420 18 18 1.0 0 0 0 0 M 0 KR§ 17 530 20 20 1.0 0 0 0 M BB 0 17 465 23 20 1.2 0 0 + SC M 28 0 465 21 19 1.1 0 0 *Echocardiographic identification of a septal-free wall thickness ratio of > 1.3 in at least one first-degree relative of the athlete. tThickening involving the mural endocardium in left ventricular outflow tract, presumably resulting from contact of the free edge of the anterior mitral leaflet with the ventricular septum. tAlso had anomalous origin of the left coronary artery from the anterior sinus of Valsalva. §Also had gross and histologic findings consistent with mitral valve prolapse, as well as a family history of that disease. ¶Measured behind the midpoint of the posterior mitral leaflet at the level of the inferior margins of the mitral leaflets. Abbreviations: AML = anterior mitral leaflet; ASH = asymmetric septal hypertrophy; LVH = left ventricular hypertrophy; LVFW = left ventricular free wall; VS = ventricular septum; VS/LVFW = ventricular septal-to-left ventricular free wall thickness ratio; + = present; 0 = absent;- = data not available. patients had total occlusion of the right coronary artery by an antemortem thrombus and hemorrhage into an atherosclerotic plaque. This athlete, a 28-yearold professional football player, had a large myocardial infarct that was about 2 months old in the posterior left ventricular wall at necropsy. The other athlete with severe coronary atheroslcerosis also showed extensive atherosclerotic plaque in the ascending aorta. There was no evidence of necrosis or scarring in this heart. The remaining patient had about 50% luminal narrowing of the left anterior descending coronary artery by atherosclerotic plaque and no, or only minimal, atherosclerosis in each of the other two major extramural coronary arteries. There was no myocardial necrosis or scarring. However, the left anterior descending coronary artery was completely occluded by an antemortem thrombus 2 cm from the bifurcation of the left coronary artery. This thrombus was 1 cm in length and adherent to the underlying atherosclerotic plaque. Aortic Rupture Two athletes (ages 18 and 21 years) died of aortic rupture, with massive mediastinal hemorrhage. In each, the ascending aorta was markedly dilated (about 10 cm in diameter) (fig. SA) and had a linear tear in its wall. In one patient the aortic rupture was associated with a dissecting aneurysm of the ascending aorta. Histologic examination of the aortic wall in both athletes showed markedly diminished numbers of elastic fibers in the media, compatible with "cystic medial necrosis" (fig. SB). One of these athletes had the typical physical stigmata of the Marfan syndrome; the other athlete was tall (81 inches) and relatively thin (210 pounds), but did not have the other physical features of that syndrome. Patients with Probable Cardiovascular Disease Six of the 29 athletes had cardiovascular alterations that were considered probable (but not definitive) 222 VOL 62, No 2, AUGUST 1980 CI RCULATION Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 FIGURE 3. Small arteries in the conduction system of two athletes who died suddenly. (A-C) The artery to the sinoatrial node of a 24-year-old basketball player with hypertrophic cardiomyopathy (hematoxylin and eosin stain). (D and E) Arteries to the atrioventricular node of a 1 7-year-old basketball player with idiopathic concentric left ventricular hypertrophy (elastic van Gieson stain). (A) Artery to sinoatrial node has markedly narrowed lumen due to extreme thickening of the arterial wall (magnification X 95). (B) Same artery as in panel A, but at a different level and at lower magnification (X 55), showing calcium deposits in adjacent myocardial fibers (outlined by broken line). (C) Higher magnification view (X 400) of the calcium-containing fiber shown in panel B. (D) Low-magnification view (X 40) of the portion of atrioventricular nodal tissue that penetrates the centralfibrous body. Three distinctly abnormal small arteries are evident (arrows). (E) Highmagnification view (x 130) of one of the vessels shown in panel D, but at a different level. The lumen is markedly narrowed due to thickening of the arterial wall. .! h SUDDEN DEATH IN YOUNG ATHLETES/Maron et al. AORTA L CIRC. R.C.A. PUL. A. L.AD. ANOMALOUS ORIGIN LEFT CORONARY ARTERY FROM RIGHT (ANTERIOR) SINUS OF VALSALVA AORTA L. CIRC. Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 R.C.A IR C. PUL. A. L.A.D. NORMAL FIGURE 4. Diagrammatic representation of anomalous origin of the left coronary artery from the right (anterior) sinus of Valsalva. The left coronary artery may have a common (or separate) ostia with the right coronary artery that also arises from the right sinus of Valsalva. Note the acute leftward and posterior course of the left coronary artery between the aorta and pulmonary artery. Normal origins of the left coronary artery from the left sinus of Valsalva and of the right coronary artery from the right sinus of Valsalva are shown below for comparison. A = anterior coronary cusp; L = left coronary cusp; P = posterior (noncoronary) cusp; L.A.D. left anterior descending coronary artery; L. CIRC. left circumflex coronary artery; PUL. A. - pulmonary artery; R. CA. = right coronary artery. evidence of cardiovascular disease. Five had a hypertrophied, nondilated left ventricle unassociated with a cardiac lesion that could produce left ventricular hypertrophy; heart weights were increased over normal'7 and ranged from 420-530 g. However, each of these five hearts differed from the other hearts in this study considered to represent hypertrophic car- FIGURE 5. (A) Posteroanterior chest radiograph obtained from an 18-year-old male collegiate swimmer with the Marfan syndrome who died 2 months later of a ruptured aorta. Note prominent dilatation of the ascending aorta. (B) Histologic section of ascending aorta from the second patient in this study who died of a ruptured aorta. Note the markedly diminished numbers of elastic fibers (which stain darkly) in the media. Intima is at the top and adventitia is at the bottom. Elastic van Gieson stain; magnification X 80. 223 224 VOL 62, No 2, AUGUST 1980 CIRCULATION Normal Arterial Supply Poor Arterial Supply Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 FIGURE 7. Diagrammatic representation of coronary arterial distribution to the posterior surface of the heart. (left) Normal arterial distribution. (right) Markedly decreased distribution of coronary arterial vessels in a 17year-old female distance runner who died suddenly. FIGURE 6. Heart of a 17-heart-old soccer player showing marked concentric (symmetric) thickening of the ventricular septum (VS) and left ventricular free wall (L V). R V = right ventricular wall; A V aortic valve. = diomyopathy by virtue of the absence of: (1) asymmetric septal hypertrophy (septal-free wall ratios ranged from 1.0-1.2) (fig. 6); (2) disorganization of cardiac muscle cells in the ventricular septum; and (3) clinical or echocardiographic evidence of hypertrophic cardiomyopathy in closely related family members. These five patients with idiopathic concentric left ventricular hypertrophy ranged in age from 16-28 years. Two were normotensive, but blood pressure recordings had not been obtained in the other three. In three of these five patients the degree of left ventricular wall thickening was particularly marked (average of ventricular septal and posterior left ventricular free wall thickness > 20 mm); in the other two patients, left ventricular wall thickening was only moderate (thicknesses of 16 mm and 18 mm) (table 2). Mild-to-moderate right ventricular dilatation was present in three of these patients. Two of the five athletes with idiopathic concentric left ventricular hypertrophy had associated cardiac malformations. In one, a 17-year-old football player with a 530-g heart, the anterior and posterior mitral leaflets had an appearance consistent with relatively mild mitral valve prolapse thickened, gelatinous and mildly scalloped leaflets with excessive mucoid - material, histologically. This patient's mother and two siblings had clinical or echocardiographic evidence of mitral prolapse. In addition, this athlete had an anatomic alteration of the artery to the atrioventricular node, involving severe fibromuscular hyperplasia of the vessel wall and marked narrowing of the lumen (figs. 3D and 3E). One other athlete with concentric ventricular wall thickening had a similar alteration in the artery to the atrioventricular node. The conducting tissue was otherwise normal in these latter two athletes. None of the athletes who had idiopathic concentric left ventricular hypertrophy, with or without alterations in the artery to the atrioventricular node, had abnormal intramural coronary arteries in other areas of ventricular myocardium. The remaining athlete with probable evidence of cardiovascular disease was a 17-year-old distance runner whose heart was of normal weight (280 g), without luminal narrowing of the extramural coronary arteries. The left main and left anterior descending coronary arteries were normal. However, only two small posterior descending branches to the posterior left ventricular wall arose from the left circumflex coronary artery. The right coronary artery was short and small in caliber and it gave off only two small branches to the posterior wall of right ventricle and none to the posterior wall of left ventricle (fig. 7). Hence, the coronary arterial distribution to the posterior wall of both ventricles was diminished. Clinical Findings: Suspicion of Cardiovascular Disease During Life Of the 29 athletes, 21 had been entirely asymptomatic, even upon retrospective questioning of closely related family members. The remaining eight athletes had transient symptoms that may have been due to cardiac disease, including syncope in three, presyncope in one (a single episode 2 months before SUDDEN DEATH IN YOUNG ATHLETES/Maron et al. 225 Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 TABLE 3. Findings in Seven Competitive Athletes in Whom Cardiovascular Disease Was Suspected or Identified During Life Reason disease Age first Clincal Necropsy Cardiac cath ECG Patient (years) Sex suspected diagnosis diagnosis 14 M Syncope WPW pattern; LAD; Mild bilateral DM WPW syndrome Hypertrophic PA branch T-wave abn. I, aVL, cardiomyopathy stenosis V4-Ve; LVH M Family 14 MM LVH with "strain"; Rest LVOT grad. Hypertrophic Hypertrophic = 15 mm Hg; history Q waves II, III, cardiomyopathy cardiomyopathy aVF, Ve LVEDP = 24 mm Hg M Heart Q waves III, aVF, None VSD 17 JG Hypertrophic murmur V5, VB; - LVH cardiomyopathy M for Marfan SS 18 Exam Normal None syndrome; Ruptured ascendMarfan AR ing aorta syndrome M Heart 20 EW None Normal athlete's Deep, symmetric Hypertrophic T-wave inversion II, murmur heart cardiomyopathy III, aVF, Vs-V6; LVH 23 M VT on Rest LVOT grad. Normal athlete's OB Deep, symmetric Hypertrophic = 0; LVEDP routine T-wave inversion I, heart cardiomyopathy = 5 mm Hg; exercise II, III, aVF, V4-V6; LVH* LV cavity test oblteration (EF = 95%) Rest LVOT grad. Normal athlete's Hypertrophic = 0; LVEDP heart cardiomyopathy = 10 mm Hg *Initially this patient underwent a treadmill exercise ECG as part of the preparticipation examination for his university basketball team. During exercise, ST-segment depression of 2 mm with negative slope and frequent premature ventricular complexes were present. During the second minute of recovery, a 4-second period of ventricular tachycardia occurred (fig. 8). A subsequent 24-hour ambulatory ECG showed only occasional premature ventricular complexes and episodes of junctional rhythm; a second 24-hour ECG recorded during a basketball scrimmage showed no arrhvthmia. Abbreviations: AR = aortic regurgitation; abn. = abnormality; cath = catheterization; EF = ejection fraction; grad. = gradient; LAD = left-axis deviation; LV = left ventricular; LVEDP left ventricular end-diastolic pressure; LVH = left ventricular hypertrophy; LVOT = left ventricular outflow tract; PA = pulmonary artery; WPW = Wolff-Parkinson-White syndrome; VSD = ventricular septal defect; VT = ventricular tachycardia; t probable. KK 30 F Mild fatigue LVH with "strain"; diffuse T-wave abn. = death), chest pain in two, periodic mild fatigue in one, and mild fatigue, presyncope and palpitations in one. In two athletes with transient symptoms, as well as in five other asymptomatic athletes, cardiovascular disease had been suspected by an examining physician during life (table 3). In the latter five athletes, the suspicion of cardiovascular disease initially arose because of a heart murmur in two, a family history of heart disease in one,5 identification of ventricular tachycardia on an exercise ECG performed as a routine part of a preparticipation athletic examination in one (fig. 8B), and during evaluation for the Marfan syndrome in one. Six of the seven athletes in whom cardiovascular disease was suspected during life had hypertrophic cardiomyopathy confirmed at necropsy. However, in only one of those six athletes with hypertrophic cardiomyopathy was the correct diagnosis made clinically. In the other five athletes, the diagnosis made during life was "normal athlete heart" in three, ventricular septal defect in one and Wolff-ParkinsonWhite syndrome in one. Each of the latter five athletes with hypertrophic cardiomyopathy had a distinctly abnormal ECG (figs. 8A and 9). Two athletes with abnormal ECGs also underwent cardiac catheterization; neither had a left ventricular outflow gradient under basal conditions. (Provocative interventions were not performed.) These findings led to the erroneous conclusion that hypertrophic cardiomyopathy was absent, even though one of these athletes also had a markedly hyperdynamic left ventricle (fig. 8C). The seventh athlete had been identified as having the Marfan syndrome with aortic root dilatation (fig. 4A) and aortic regurgitation during life; he had been advised not to compete in intercollegiate sports, but died of a ruptured aorta 2 months later during swimming practice. In the remaining 22 athletes there had been no suspicion of cardiovascular disease during life. However, clinical data in two suggested the presence of coronary heart disease, even though it was not identified until necropsy. One of these two athletes was a 28-year-old professional football player who had an abnormally elevated serum cholesterol (350 mg%; normal < 280 mg%), as well as a family history of premature coronary heart disease and death, identified at a routine preparticipation examination for professional athletes. Subsequently, type II hyperlipoproteinemia was documented in two surviving first-degree relatives. Upon retrospective analysis of the athlete's medical history, it was ap- 226 CIRCULATION 11 I -. .. .. V, .. 111 .. V2 .. V3 aVR .. ... aVL --- VOL 62, No 2, AUGUST 1980 aVF .. V6 a Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 Cont. -Co. O.B. 10/14/74 FIGURE 8. Electrocardiographic and angiographic finding.v in a 23-year-old collegiate basketball player (O.B.) in whom the diagnosis of hypertrophic cardiomyopathy was made at necropsy. (A) Standard 12-lead ECG obtained under basal conditions showing marked symmetric T-wave inversion (as much as 15 mm in depth), as well as left ventricular hypertrophy. (B) Four-second period of ventricular tachycardia that occurred in the second minute of recovery after a routine treadmill exercise test and terminated spontaneously. (C) Left ventricular angiocardiogram in diastole (top) and end-systole (C1) showing marked cavity obliteration (ejection fraction of 95%). LV - left ventricular cavity. The ECGs have been retraced for clarity. parent that he had experienced symptoms compatible with myocardial infarction about 45 days before death (which was documented at necropsy), and frequent episodes of chest pain with exertion thereafter. During life, the acute myocardial infarction and chest pains had not been considered evidence of heart disease, but rather manifestations of a traumatic injury. An ECG recorded before the acute myocardial infarction (the only one obtained from this patient) was normal. A second athlete who died of severe coronary atherosclerosis had an abnormal ECG and elevated serum cholesterol (380 mg%) identified at a preparticipation athletic examination, but no further diagnostic evaluation was undertaken. Discussion This study shows that most sudden and unexpected deaths in young, competitive athletes are due to car- c diovascular disease. In 28 of the 29 athletes, a structural cardiovascular abnormality was identified at necropsy, and in 22 athletes it was almost certainly responsible for their sudden death. Although the etiology of sudden death in one patient without structural cardiac disease is unknown, it is possible that a primary rhythm disturbance was responsible. While a wide variety of cardiovascular malformations (primarily congenital) was identified in our 29 athletes, the most frequently encountered disease was hypertrophic cardiomyopathy, a condition known to be responsible for sudden death in young, asymptomatic subjects.5 6,23,24 Although this disease accounted for almost one-half of the deaths in the present series, our data cannot be used to define precisely the prevalence with which different diseases cause sudden death in young athletes. First, our series is relatively small, and cannot be assumed to be an en- 227 SUDDEN DEATH IN YOUNG ATHLETES/Maron et al. aVR I. I. 1. |l 1-1 1 , - 11t-31 1: I - LIi 1 ~l 1-:1 1 :t........................ 1:t-- 1 -,. - 1---1 aVL aVF I 1 aVR aVL 11 E~ ~ ~ ~ ~ ~ ~ ~ ~ ~ ~ +J 'Wk -eX aVF - :1 1.t .... t .... IS, -1hSTD 1 Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 3 .;. I aVR aVL aVF -1 I. .t .... 111 11 1 Il 11 I 1 I aVR aVL aVF :,1 -i4-| ,. R.l:. T" 4- .... IIS----- ---l-i l--t Ali--t~Y i =~ tio-1-7 tii ai[ff i li T hl, ::11: V, V2 V3 hf i_ lz II I Ii i I .I, . .... . .. VAIJ~ .... -I t ::i' ii:: Ri fm H ;! 1, t t~111 1 011 Ptft IiLIz T III1 .... Hi -N:: F lF :j ::: :: lifil- -11- Lo!i. V4 -T .. 4 1- 3i I2STD V6 Vs rx' IzI¶ liiitli! - iI ~Ii V, V2 V3 V4 VS V6 4A'P. - Li11 ail,, 1---. ,l :: 1* I ll --- i;. ::: -l le :l ...c l'e~ 1!.a f l,1-' .1H.' -.:!!! :::i 1,1-. .: 1; :::: i'' 9-15-75 E. W. 1/2STD K.K. FIGURE 9. ECGs showing the spectrum of abnormalities from four athletes who were identified as having hypertrophic cardiomyopathy at necropsy. Although cardiovascular disease was suspected in each athlete during life, another clinical diagnosis had been made in each. (top left) From a 14-year-old wrestler (D.M.) showing Wolff-Parkinson- White pattern, left-axis deviation, diffuse T-wave inversion andflattening in leads I, aVL and V4-V6, andprobable left ventricular hypertrophy. (top right) From a 17-year-oldfootballplayer (J. G.) showing deep Q waves in leads III, aVF, V, and VB, and probable left ventricular hypertrophy. (bottom left) From a 20-year-old basketball player (E. W.) showing deep symmetric T-wave inversion in leads II, III, aVF and V-V6, and left ventricular hypertrophy. Compare with a similar ECG from another athlete shown in figure 8. (bottom right) From a 30-year-old tennis player (K.K.) showing left ventricular hypertrophy with "strain" and diffuse T-wave flattening and inversion. The ECGs have been retraced for clarity. tirely representative sampling of sudden death in athletes. Second, unavoidable biases, intrinsic to our study design, may have influenced the selection of subjects for this investigation. For example, athletes with unequivocal cardiac disease recognized at necropsy may have been preferentially referred to the NHLBI and the Armed Forces Institute of Pathology. However, 17 of 29 subjects were obtained by follow-up of news media reports describing sudden death in athletes. Hence, most patients in this series were identified initially as competitive athletes who had died suddenly, and not as patients with heart disease who subsequently proved to have been athletes. Even when this latter segment of our patient population was considered separately, our conclusions were similar to those from the entire study population. For example, eight of the 17 athletes initially identified through the news media had hypertrophic cardiomyopathy, compared with 14 of the 29 athletes in the overall study were group. The fact th4t we chose to limit our investigation to competitive athletes, who were necessarily relatively 228 CIRCULATION Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 young, also importantly influenced the relative prevalence of cardiovascular diseases found. In such young athletes, atherosclerotic coronary disease would be expected to be an infrequent cause of death. Conversely, had we studied recreational athletes over the age of 30 years at death, the frequency of coronary heart disease would surely have been much higher. Certain cardiovascular diseases, such as valvular heart disease, coarctation of the aorta, fixed subvalvular aortic stenosis or other acyanotic or cyanotic heart malformations, did not appear in our series of athletes. Such lesions would likely have been identified earlier in life due to the presence of a loud heart murmur or other obvious physical findings and, hence, before participation in competitive athletics had begun. Other known causes of sudden death, such as prolonged QT-interval syndrome, probably did not appear in our series due to their relatively low prevalence in the population. Cardiovascular disease was unrecognized during life in most of our athletes, and suspected clinically in only seven of the 29. In only two of these seven athletes was the correct diagnosis established during life: one with obvious stigmata of the Marfan syndrome and the second in whom hypertrophic cardiomyopathy was identified after his brother had died suddenly of that disease. The five other athletes in whom cardiac disease was suspected clinically all died of hypertrophic cardiomyopathy. All five of these athletes had distinctly abnormal ECGs and one had an abnormal left ventricular angiocardiogram. Thus, it is likely that if high-quality echocardiograms had been obtained in these athletes, the correct diagnosis of hypertrophic cardiomyopathy would have been made during life. Each of these five athletes, after examination for possible heart disease, were permitted by their examining physician to continue participation in competitive athletics and each subsequently died on the practice field or during an athletic contest. The five athletes who had concentric (symmetric) left ventricular hypertrophy without ancillary evidence of a primary, genetically transmitted cardiomyopathy represent a diagnostic dilemma. The significance of the ventricular hypertrophy in these patients is unclear. First, we cannot exclude the possibility that the increased cardiac mass identified at necropsy represented only the "physiologic" hypertrophy that would be expected in many highly conditioned competitive athletes.25 SO Indeed, in two of these athletes, the degree of left ventricular wall thickening was consistent with that usually present in actively competing athletes. However, in the other three athletes with idiopathic concentric hypertrophy, the magnitude of left ventricular wall thickening (> 20 mm) clearly exceeded that which would appear to be "physiologic" hypertrophy, even after making reasonable allowances for known discrepancies between ventricular wall thicknesses assessed by echocardiography and at necropsy.31 Second, systemic hypertension could not be definitely excluded as the cause of left ventricular hypertrophy in three patients because blood pressure had not been recorded during life; however, two of VOL 62, No 2, AUGUST 1980 these three were less than 20 years old at death, and it is unlikely that hypertension played an etiologic role in their disease process. In two athletes with idiopathic concentric left ventricular hypertrophy, associated abnormalities (which may have been primary or contributing factors in their sudden deaths) also complicate interpretation of the significance of the cardiac hypertrophy found in this subgroup. One of these athletes had, at necropsy, alterations of the mitral valve leaflets consistent with mitral valve prolapse and a family history of that disease. The abnormality of the mitral valve in this patient was relatively mild and therefore was probably not responsible for significant mitral regurgitation or the degree of left ventricular hypertrophy that was present. In addition, this athlete and one other with idiopathic concentric hypertrophy had severe narrowing of the artery to the atrioventricular node. Abnormal small intramural arteries in the atrioventricular node (as well as in the sinoatrial node), morphologically similar to those present in our patients, have been described in young, healthy subjects32' 33 or in patients with hypertrophic cardiomyopathy34 who died suddenly, as well as in patients with a variety of other cardiac or systemic diseases.35 Unlike the findings in our athletes, the small-vessel abnormalities described in these reports were usually associated with significant fibrosis of the conducting tissue. Our findings raise questions regarding the prospective identification of potentially lethal cardiovascular diseases in competitive athletes during life. Screening of athletes by personal and family history, physical examination, ECG and echocardiogram would have identified most of the athletes in our study group. However, routine comprehensive screening of this type would probably be impractical. Although screening by personal history and physical examination alone would identify many of the more obvious cardiovascular abnormalities (such as systemic hypertension or aortic valvular stenosis), it would be relatively insensitive for detecting other diseases. For example, the most frequent cause of death in this series, hypertrophic cardiomyopathy, is usually not associated with a loud heart murmur,36" 37 or even always with an abnormal ECG38 when present in its nonobstructive form. Definitive identification of hypertrophic cardiomyopathy without obstruction could be made noninvasively in athletes only by echocardiography. Similarly, echocardiography could identify many of the patients with aortic root dilatation who may be at high risk of aortic rupture. The data in this study do not provide definitive information regarding the feasibility of screening programs for detecting cardiovascular disease in competitive athletes. However, the basic knowledge and awareness of the diseases that may cause sudden death in athletes help to create the groundwork necessary for the initiation of studies that will provide such answers. References 1. James TN, Froggatt P, Marshall TK: Sudden death in athletes. Ann Intern Med 67: 1013, 1967 young SUDDEN DEATH IN YOUNG ATHLETES/Maron et al. Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 2. Cheitlin MD, De Castro CM, McAllister HA: Sudden death as a complication of anomalous left coronary origin from the anterior sinus of Valsalva. A not-so-minor congenital anomaly. Circulation 50: 780, 1974 3. Jokl E, McClellan JT, Williams WC, Gouze F, Bartholomew R: Congenital anomaly of left coronary artery in a young athlete. Cardiologia 49: 253, 1966 4. Jokl E, McClellan JT, Ross GD: Congenital anomaly of left coronary artery in a young athlete. JAMA 182: 572, 1962 5. Maron BJ, Lipson LC, Roberts WC, Savage DD, Epstein SE: "Malignant" hypertrophic cardiomyopathy: identification of a subgroup of families with unusually frequent premature deaths. Am J Cardiol 41: 1133, 1978 6. Maron BJ, Roberts WC, Edwards JE, McAllister HA, Foley DD, Epstein SE: Sudden death in patients with hypertrophic cardiomyopathy: characterization of 26 patients without previous functional limitation. Am J Cardiol 41: 803, 1978 7. Maron BJ, Roberts WC: Quantitative analysis of cardiac muscle cell disorganization in the ventricular septum of patients with hypertrophic cardiomyopathy. Circulation 59: 689, 1979 8. Lev M, Widran J, Erickson EE: A method for the histopathologic study of the atrioventricular node, bundle, and branches. Arch Pathol 52: 73, 1951 9. Lev M, Watne AL: Method for routine histopathologic study of human sinoatrial node. Arch Pathol 57: 168, 1954 10. Henry WL, Clark CE, Epstein SE: Asymmetric septal hypertrophy (ASH): echocardiographic identification of the pathognomonic anatomic abnormality of IHSS. Circulation 47: 225, 1973 11. Maron BJ, Epstein SE: Hypertrophic cardiomyopathy: a discussion of nomenclature. Am J Cardiol 43: 1242, 1979 12. Roberts WC: Valvular, subvalvular and supravalvular aortic stenosis: morphologic features. Cardiovasc Clin 5: 104, 1973 13. Menges H, Brandenburg RO, Brown AL: The clinical, hemodynamic and pathologic diagnosis of muscular subvalvular aortic stenosis. Circulation 24: 1126, 1961 14. Teare D: Asymmetrical hypertrophy of the heart in young patients. Br Heart J 20: 1, 1958 15. van Dorp WG, ten Cate FJ, Vletter WB, Dohmen H, Roelandt J: Familial prevalence of asymmetric septal hypertrophy. Eur J Cardiol 4: 349, 1976 16. Clark CE, Henry WL, Epstein SE: Familial prevalence and genetic transmission of idiopathic hypertrophic subaortic stenosis. N Engl J Med 289: 709, 1973 17. Gould SE: Pathology of the Heart and Blood Vessels, 3rd ed. Springfield, Illinois, Charles C Thomas, 1968, p 1139 18. McReynolds RA, Roberts WC: The intramural coronary arteries in hypertrophic cardiomyopathy. (abstr) Am J Cardiol 35: 154, 1975 19. James TN: Pathology of small coronary arteries. Am J Cardiol 20: 679, 1967 20. Benson PA: Anomalous aortic origin of coronary artery with sudden death: case report and review. Am Heart J 79: 254, 1970 21. Benson PA, Lack AR: Anomalous aortic origin of left coronary 229 artery. Arch Pathol 86: 214, 1968 22. Cohen LS, Shaw LD: Fatal myocardial infarction in an 11year-old boy associated with a unique coronary artery anomaly. Am J Cardiol 19: 420, 1967 23. Lambert EC, Menon VA, Wagner HR, Vlad P: Sudden unexpected death from cardiovascular disease in children. Am J Cardiol 34: 89, 1974 24. Fiddler GI, Tajik AJ, Weidman WH, McGoon DC, Ritter DG, Giuliani ER: Idiopathic hypertrophic subaortic stenosis in the young. Am J Cardiol 42: 793, 1978 25. Parker BM, Londeree BR, Cupp GV, Dubiel JP: The noninvasive cardiac evaluation of long-distance runners. Chest 73: 376, 1978 26. Roeske WR, O'Rourke RA, Klein A, Leopold G, Karliner JS: Noninvasive evaluation of ventricular hypertrophy in professional athletes. Circulation 53: 286, 1976 27. Allen HD, Goldberg SJ, Sahn DJ, Schy N, Wojcik R: A quantitative echocardiographic study of champion childhood swimmers. Circulation 55: 142, 1977 28. Morganroth J, Maron BJ, Henry WL, Epstein SE: Comparative left ventricular dimensions in trained athletes. Ann Intern Med 82: 521, 1975 29. Gilbert CA, Nutter DO, Felner JM, Perkins JV, Heymsfield SB, Schlant RC: Echocardiographic study of cardiac dimensions and function in the endurance-trained athlete. Am J Cardiol 40: 528, 1977 30. Ikaheimo MJ, Palatsi IJ, Takkunen JT: Noninvasive evaluation of the athletic heart: sprinters versus endurance runners. Am J Cardiol 44: 24, 1979 31. Maron BJ, Henry WL, Roberts WC, Epstein SE: Comparison of echocardiographic and necropsy measurements of ventricular wall thicknesses in patients with and without disproportionate septal thickening. Circulation 55: 341, 1977 32. James TN, Armstrong RS, Silverman J, Marshall TK: De subitaneis mortibus. VI. Two young soldiers. Circulation 49: 1239, 1974 33. James TN, Hackel DB, Marshall TK: De subitaneis mortibus. V. Occluded A-V node artery. Circulation 49: 772, 1974 34. James TN, Marshall TK: De subitaneis mortibus. XII. Asymmetrical hypertrophy of the heart. Circulation 51: 1149, 1975 35. James TN: An etiologic concept concerning the obscure myocardiopathies. Prog Cardiovasc Dis 7: 43, 1964 36. Epstein SE, Henry WL, Clark CE, Roberts WC, Maron BJ, Ferrans VJ, Redwood DR, Morrow AG: Asymmetric septal hypertrophy. Ann Intern Med 81: 650, 1974 37. Braunwald E, Lambrew CT, Rockoff SD, Ross J Jr, Morrow AG: Idiopathic hypertrophic subaortic stenosis. I. A description of the disease based upon an analysis of 64 patients. Circulation 30 (suppl IV): IV-3, 1964 38. Savage DD, Seides SF, Clark CE, Henry WL, Maron BJ, Robinson FC, Epstein SE: Electrocardiographic findings in patients with obstructive and nonobstructive hypertrophic cardiomyopathy. Circulation 58: 402, 1978 Sudden death in young athletes. B J Maron, W C Roberts, H A McAllister, D R Rosing and S E Epstein Downloaded from http://circ.ahajournals.org/ by guest on June 18, 2017 Circulation. 1980;62:218-229 doi: 10.1161/01.CIR.62.2.218 Circulation is published by the American Heart Association, 7272 Greenville Avenue, Dallas, TX 75231 Copyright © 1980 American Heart Association, Inc. All rights reserved. Print ISSN: 0009-7322. Online ISSN: 1524-4539 The online version of this article, along with updated information and services, is located on the World Wide Web at: http://circ.ahajournals.org/content/62/2/218 Permissions: Requests for permissions to reproduce figures, tables, or portions of articles originally published in Circulation can be obtained via RightsLink, a service of the Copyright Clearance Center, not the Editorial Office. Once the online version of the published article for which permission is being requested is located, click Request Permissions in the middle column of the Web page under Services. Further information about this process is available in the Permissions and Rights Question and Answer document. Reprints: Information about reprints can be found online at: http://www.lww.com/reprints Subscriptions: Information about subscribing to Circulation is online at: http://circ.ahajournals.org//subscriptions/