* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Prospective and Retrospective ECG Gating for Thoracic CT
Survey
Document related concepts
Cardiac contractility modulation wikipedia , lookup
Myocardial infarction wikipedia , lookup
History of invasive and interventional cardiology wikipedia , lookup
Mitral insufficiency wikipedia , lookup
Electrocardiography wikipedia , lookup
Cardiac surgery wikipedia , lookup
Coronary artery disease wikipedia , lookup
Hypertrophic cardiomyopathy wikipedia , lookup
Management of acute coronary syndrome wikipedia , lookup
Marfan syndrome wikipedia , lookup
Turner syndrome wikipedia , lookup
Dextro-Transposition of the great arteries wikipedia , lookup
Transcript
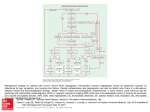
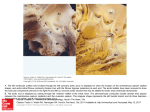
6A S C U L A R A N D ) N T E R VE N T I O N A L 2 A D I O L O G Y s / R I G I N A L 2 E S E A R C H Wu et al. ECG Gating for Thoracic CT Angiography FOCUS ON: Vascular and Interventional Radiology Original Research Wenhui Wu1 Joseph Budovec2 W. Dennis Foley 2 Wu W, Budovec J, Foley WD Keywords: cardiac gating, CT angiography, emergency radiology, hemodynamics, thoracic CT angiography, vascular disease DOI:10.2214/AJR.08.2158 Received November 24, 2008; accepted after revision February 2, 2009. W. Wu was at the Medical College of Wisconsin at the time of the study for a visiting fellowship that was sponsored in part by GE Healthcare. W. D. Foley has an Investigator Agreement with GE Healthcare CT Division. 1 Department of Radiology, Cardiovascular Institute & Fuwai Hospital, Chinese Academy of Medical Sciences & Peking Union Medical College, Beijing, China. 2 Department of Radiology, Medical College of Wisconsin, 9200 W Wisconsin Ave., Milwaukee, WI 53226. Address correspondence to W. D. Foley ([email protected]). AJR 2009; 193:955–963 0361–803X/09/1934–955 © American Roentgen Ray Society AJR:193, October 2009 Prospective and Retrospective ECG Gating for Thoracic CT Angiography: A Comparative Study /"*%#4)6% The objective of our study was to compare radiation dose, contrast load, thoracic aortic attenuation value, and image quality parameters of MDCT thoracic aortography performed with prospective and retrospective cardiac gating. -!4%2)!,3 !.$ -%4(/$3 Studies were performed on 80 patients (prospective ECG gating, n = 40; retrospective ECG gating, n = 40) either being evaluated for thoracic aortic aneurysm (n = 23) or aortic dissection (n = 36) or undergoing postsurgical or postintervention follow-up (n = 21). Image acquisition parameters and radiation dose (CT dose index volume [CTDIvol] and dose–length product [DLP]) were obtained from image archival data. Contrast load and aortic attenuation values were obtained from a data registry. The comparative degrees of motion artifact and banding artifact were assessed on parasagittal maximumintensity-projection (MIP) images and reformatted images in the plane of the aortic valve. 2%35,43 CTDIvol and DLP in the prospective ECG-gating group was 28.8 ± 2.12 mGy (mean ± SD) and 833.7 ± 115.77 mGy/cm, respectively, which are significantly lower (p < 0.001) than those values in the retrospective ECG-gating group (74.7 ± 13.42 mGy and 2,547.3 ± 553.27 mGy/cm). The average contrast load in the prospective gating group was 109.1 ± 14.74 mL and in the retrospective gating group, 101.3 ± 10.45 mL (p < 0.05). The average aortic attenuation values (in Hounsfield units) for the prospective and retrospective ECG-gated groups were 447.6 and 350.2 HU, respectively, for the mid ascending aorta, 413.6 and 325.7 HU for the mid aortic arch, 418.2 and 327.6 HU for the mid descending aorta, and 355.0 and 306.2 HU for the supraceliac aorta. Subjective scores of motion artifact and banding artifact were equivalent between the two groups. #/.#,53)/. Compared with retrospective ECG-gated thoracic CT angiography, prospective ECG-gated thoracic CT angiography was associated with a lower radiation dose, slightly increased contrast load, increased aortic attenuation values, and equivalent image quality. M DCT angiography is a technically robust, noninvasive imaging technique for the evaluation of vascular disease, including diseases affecting the thoracic aorta. Advances in image acquisition technology have resulted in improvements in image acquisition speed and spatial resolution. Consequently, patient studies can now be performed rapidly for both emergent and elective indications. MDCT of the thoracic aorta is used to diagnose various acute and chronic conditions including aortic aneurysm, aortic dissection, penetrating atherosclerotic ulcer, intramural hematoma, traumatic aortic injury, aortic rupture, inflammatory disorders, and congenital malformations [1, 2]. By creating a 3D image, CT angiography may be superior to conventional angiography [3, 4]. MDCT using a 64-channel system is used not only for the diagnosis of aortic disease but also for surgical planning and postintervention evaluation [5]. In addition, CT angiography is less invasive, consumes fewer resources, and is more cost efficient than conventional angiography, especially after the introduction of 64-MDCT scanner [6, 7]. In patients undergoing CT evaluation for suspected aortic dissection or ascending aortic aneurysm, vessel motion can result in blurred aortic outlines, particularly of the aortic root and proximal ascending aorta. In addition, the aortic valve and coronary arteries frequently are not clearly displayed even when images are reoriented in the longitudinal and shortaxis plane of these structures. ECG-gated data acquisition addresses these issues. Roos et al. [8] reported that image 955 7U ET AL quality of the aorta was superior using retrospective ECG-gated acquisition compared with non-ECG-gated acquisition. The benefit of ECG-gated synchronization was most pronounced in the proximal ascending aorta and was less so at the aortic arch and proximal descending aorta [1]. ECG gating improves the diagnosis of type A aortic dissections, particularly the proximal extent of the mural flap in relationship to the aortic root, coronary ostia, and aortic valves. In addition, it improves the delineation of the proximal margin of ascending aortic aneurysms. Thoracic aortic CT angiography with cardiac gating may now be considered the preferred technique for thoracic CT angiography when diagnostic concern relates to the aortic sinus, ascending aorta, or coronary arteries. An alternative technique is gated thoracic aortic MR angiography using steadystate free precession acquisition. Advances in parallel imaging techniques permit imaging of the thoracic aorta and left ventricular outflow tract with a 5- to 6-second breathhold [9–11]. A more comprehensive evaluation of the thoracic aorta and aortic valve requires multiplanar, multisequence imaging with ECG and respiratory gating, including sagittal oblique images through the aorta and aortic root, that prolong acquisition time. Thoracic aortic MR angiography is not used in patients presenting with acute chest pain and is limited in patients with implanted cardiac devices (e.g., automatic implantable cardioverter-defibrillators, pacemakers) and prosthetic valves. Although MR angiography can define the anatomy of the aortic valve and aortic root, the technique is not well suited to coronary arteriography. Until recently, the initial implementation of cardiac gating for CT angiography was with a retrospective ECG-gating technique. Compared with nongated techniques, retrospective gating has an acquisition duration and radiation dose that are relatively high because of the low pitch value. Prospective ECG gating is a newer technique in which the tube current is triggered for only a short segment of the R-R interval at a preset time after the R wave. Consequently, radiation is emitted every alternate heartbeat and the table moves without radiation being emitted during the intervening heartbeat [12]. The purpose of this study was to compare prospective and retrospective ECG-gated MDCT data acquisition techniques for CT angiography of the thoracic aorta with regard to radiation dose, contrast load, thoracic aortic attenuation, and image quality parameters. 956 Materials and Methods Patient Population The institutional review board approved this study with a waiver of authorization for patient informed consent. Retrospective review of CT examinations performed between March 2007 and August 2007 yielded 40 patients who had undergone MDCT thoracic CT angiography using prospective ECG gating. A comparison group of 40 patients was selected from patients who had undergone MDCT thoracic CT angiography using retrospective ECG gating performed from June 2006 to March 2007. The 80 patients in the study group had been referred for evaluation of suspected thoracic aortic disease, either dissection (n = 36) or aneurysm (n = 23), or for postsurgical or postintervention followup (n = 21). Nine patients in the prospective ECGgated group and 11 patients in the retrospective ECG-gated group were referred from the emergency department. Imaging Techniques The CT examinations were performed using a 64-MDCT system (LightSpeed VCT, GE Healthcare). All examinations were performed during patient inspiration. Circulation timing was determined via a preliminary mini bolus in which 15 mL of contrast material, either iodixanol (Visipaque 320, 320 mg I/mL, GE Healthcare) or iopamidol (Isovue 370, 370 mg I/mL, Bracco Diagnostics), was administered IV at 6 mL/s using an 18-gauge catheter placed in an antecubital vein. Visipaque was used for patients with marginal renal function (i.e., estimated glomerular filtration rate [GFR] < 60 mL/min/1.73 m2). A time–attenuation curve was derived from the comparative attenuation values of 10 sequential images obtained from the mid ascending aorta over a 20-second interval beginning 5 seconds after the beginning of contrast injection. The contrast arrival time in the ascending aorta was calculated as the time from the beginning of the injection until peak attenuation on the time– enhancement curve after the mini bolus injection. The delay time between contrast arrival and the beginning of acquisition was set at 4 seconds for both protocol groups. The contrast injection interval was the combination of the time between contrast arrival in the ascending aorta and the beginning of the acquisition plus the acquisition time (Fig. 1). The imaging volume extended from the proximal brachiocephalic arteries in a cephalad to caudad direction to the level of the origin of the celiac artery in the upper abdominal aorta. Retrospective ECG-gated acquisition used a detector configuration of 64 × 0.625 mm, scan rotation speed of 350 milliseconds, and pitch adjust- ed from 0.18 to 0.24 on the basis of the patient’s heart rate. The average heart rate during acquisition was 63 beats per minute (bpm). Table speed averaged 2.5 cm/s. ECG modulation of x-ray tube current was used, decreasing tube current during systole and early diastole and maintaining full selected tube current in mid to late diastole. Prospective ECG-gated acquisition was performed in a sequential mode with a beam collimation of 40 mm (detector configuration, 64 × 0.625 mm). Prospective gating used axial plane acquisition in an incremental dynamic mode; 75% of the R-R interval was chosen as the midpoint for the data acquisition time of 175 milliseconds, with an additional padding factor of 125 milliseconds. This padding factor added 125 milliseconds both before and after the initial designated acquisition time. The data acquisition occurs during every alternate heartbeat with incremental table motion of 3.5 cm in the intervening heartbeat. For a heart rate of 60 bpm, beam collimation of 4 cm, and acquisition of every alternate heartbeat, the effective table speed approximates 2 cm/s. The average heart rate in the prospective ECG-gated group was 62 bpm. Beta-blocker medication was administered to patients with sinus rhythm and heart rates between 70 and 100 bpm. Patients with arrhythmia were excluded. For elective studies, patients received either 100 mg of metoprolol orally or, if on current E-blocker medication, 50 mg of metoprolol orally unless contraindicated. After 45 minutes of observation, if the heart rate remained above 70 bpm, an additional 50 mg of metoprolol was administered orally. Additional E-blocker medication (5 mg of IV metoprolol) was administered if necessary in the CT scanner suite to a maximum dose of 15 mg. For patients presenting to the emergency department with acute symptoms, IV E-blocker medication (5 mg of metoprolol repeated × 2 as required) was administered. Contraindications to metoprolol therapy included severe aortic stenosis, systolic blood pressure less than 100 mm Hg, and a history of asthma requiring daily nebulizer therapy. The image acquisition parameters for prospective ECG-gated acquisition and retrospective ECG-gated acquisition are detailed in Table 1. Radiation Dose Estimation CT dose index volume (CTDIvol) and dose– length product (DLP) were recorded as direct data output from prospective ECG-gated examinations. Only the CTDIvol and DLP values obtained from the contrast-enhanced CT angiographic studies were used in study analysis. Radiation from the preliminary localization CT radiographs and from single-level dynamic scanning to determine circulation time was not included. DLP in milligrays AJR:193, October 2009 ECG Gating for Thoracic CT Angiography Contrast Contrast Saline Saline 10 seconds 10 20 30 10 20 30 6 mL/s, 14 seconds 6 mL/s, 16 seconds First circulation First circulation 6 mL/s, 8 seconds 6 mL/s, 8 seconds 64 × 0.625 mm, 0.20, 0.35 second, 2.5 cm/s 64 × 0.625 mm, 1.0, 0.35 second, 2 cm/s A " Fig. 1—Diagrams show injection/acquisition technique retrospective and prospective gating for thoracic CT angiography. Cephalocaudad coverage is 25 cm. A, Schematic diagram illustrates injection acquisition technique for thoracic CT angiography using retrospective gating. Time in seconds is indicated on x-axis. Arrow indicates calculated arrival time of injected contrast in ascending aorta using aortic peak on time–attenuation curve after mini bolus injection (6 mL/s for 15 mL). There is 4-second delay between calculated arrival time and beginning of acquisition. Acquisition interval is calculated at 10 seconds for 25-cm cephalocaudad acquisition using image acquisition parameters detailed on bottom line: detector configuration, 64 × 0.625 mm; average pitch value, 0.20; scan rotation speed, 0.35 second; table speed, 2.5 cm/s. Contrast injection rate is set at 6 mL/s with injection duration equal to time delay between aortic arrival and beginning of acquisition (4 seconds) plus acquisition interval (10 seconds). Contrast injection is followed by saline flush at same flow rate over 8-second interval. Aim of this injection and acquisition protocol is to capture image data during first circulation of injected bolus. B, Schematic diagram illustrates injection and acquisition parameters for prospective cardiac gating. Time in seconds is indicated on x-axis. Arrow represents aortic arrival of contrast material after preliminary mini bolus injection (6 mL/s for 16 seconds) and is calculated as time to peak on time–attenuation curve in ascending aorta. There is 4-second delay time between aortic arrival and beginning of acquisition. Acquisition uses step-and-shoot, or incremental dynamic, mode in which 4 cm of axial scan data is acquired in 1 rotation at fixed preset time after R wave of ECG; incremental table motion occurs during next heartbeat, and in third heartbeat, another 4 cm of axial data is again acquired at same preset time after R wave of ECG. There is 0.5-cm overlap of image anatomy between each incremental acquisition. Process is replicated until required cephalocaudad dimension is imaged. Acquisition parameters are detector configuration of 64 × 0.625 mm and scan rotation speed of 0.35 second, 2 cm/s. At heart rate of 60 beats per minute, table speed approximates 2 cm/s. Contrast medium is injected at 6 mL/s over time interval equal to delay between aortic arrival and beginning of acquisition plus acquisition time. After contrast injection, saline is injected at same rate to enable effective utilization of injected bolus and clear superior vena cava. As with retrospective gating, injection and acquisition technique are aimed to provide first circulation acquisition of imaged anatomy. per centimeter (mGy/cm) was converted to an estimated effective dose in millisieverts (mSv) using a standard conversion equation that includes the patient’s age and sex [13]. Patient radiation dose for retrospective ECGgated MDCT thoracic angiography was estimated using the lowest and highest tube current (mA) values and on times in the R-R interval during acquisition to define the estimated CTDIvol. DLP in mGy/cm was then determined by the total length of the acquisition. As with prospective ECG-gated acquisition, the DLP in mGy/cm was converted into an estimated dose in mSv using a standard conversion equation. Contrast Material Load The contrast material for the examinations was either Isovue 370 or Visipaque 320. A data registry recorded the type and amount of contrast material for each separate patient study. Visipaque was used in patients with marginal renal function, which was defined as an estimated GFR of less than 60 mL/min/1.73 m 2. Before and after the procedure, oral hydration (two glasses of 16 oz [471 mL] of tap water) was given for renal protection in all patients who received Visipaque. AJR:193, October 2009 Thoracic Aortic Attenuation The mean CT attenuation values (in Hounsfield units [HU]) of the mid ascending aorta, mid aortic arch, mid descending aorta, and supraceliac abdominal aorta were measured using a circular region of interest approximating 50% of the aortic cross-sectional area. The mean and SD of attenuation at all four levels in both patient groups were recorded. Image Quality Parameters Image quality parameters were assessed subjectively; motion artifact and resultant blurring, as well as misregistration or stairstep artifact, affecting both the thoracic aorta and aortic valve were evaluated. Motion artifact—Motion artifact causing image blurring of the thoracic aorta and brachiocephalic arteries was assessed from parasagittal subvolume maximum-intensity-projection (MIP) reformatted images using the following scale adapted from the work of Roos et al. [8]: 1, no motion artifact, optimum for diagnosis; 2, minimum aortic wall blurring, satisfactory for diagnosis; 3, moderate motion artifact, adequate for diagnosis; 4!",% )MAGING 0ARAMETERS FOR 0ROSPECTIVE AND 2ETROSPECTIVE %#''ATED Techniques CT Mode Peak Tube Potential (kVp) Tube Current (mA) Sequential prospective ECG-gated 120–140 a Helical retrospective ECG-gated 120–140 ECG dose modulationa,b Pitch 0.18–0.24 Rotation Time (s) Detector Configuration (mm) Beam Width (mm) 0.35 0.625 × 64 40 0.35 0.625 × 64 40 aTube current selection was based on patient weight: 350–450 mA for small patients (< 120 lb [54 kg]), 450–550 mA for medium patients (120–220 lb [54–99 kg]), and 600–750 mA for large patients (> 220 lb [99 kg]). bIf heart rate was less than 65 bpm, peak mA was 70–80% R-R interval; if heart rate was greater than 65 bpm, peak mA was 40–80% R-R interval. 957 7U ET AL A " C D Fig. 2—Prospective (A, B) and retrospective (C, D) ECG-gated acquisitions in patients with suspected aortic dissection. Optimal image quality without motion blurring or misregistration is shown on both types of examinations. A, Prospective ECG-gated thoracic CT aortography in 75-year-old woman with suspected aortic dissection using parasagittal subvolume maximum intensity projection (MIP). Left anterior oblique projection shows there is no motion or banding artifact. Study is considered optimum for diagnosis. Differential opacity of aortic root as compared with mid and distal ascending aorta represents time interval of two heartbeats approximating 2 seconds with differential aortic opacity between two acquisitions. Resultant linear band across aortic lumen does not represent stairstep or banding artifact. This patient had prior sternotomy and coronary artery bypass grafting (arrow) to distal right coronary artery. B, Prospective ECG-gated thoracic aortic CT angiography of same patient shown in A. Reformatted image through plane of aortic valve shows sharp delineation of trileaflet valve and aortic wall with normal leaflet coaptation. Left atrium is on posterior aspect of aortic root. Small segment of proximal left main coronary artery (arrow) is opacified. There is mild uniform opacity of right atrium and right ventricle after bolus passage through right heart. C, Retrospective ECG-gated thoracic CT aortogram study in 61-year-old man with suspected aortic dissection using parasagittal subvolume MIP. Left anterior oblique projection shows neither motion artifact nor banding artifact. Study is considered optimum for diagnosis. D, Retrospective ECG-gated thoracic aortic CT angiography in 59-year-old man undergoing surveillance study after aortic graft repair of type A dissection. Reformatted image of aortic root through plane of valve leaflets shows sharp definition of normal trileaflet valve and aortic wall and normal coaptation of leaflets. Opacified left atrium marginates right coronary and noncoronary aortic sinuses. Small segment of proximal right coronary artery (arrow) is imaged. Mild residual uniform enhancement of right heart chambers and pulmonary outflow tract is seen after bolus passage through right heart. 4, distinct motion artifact, questionable for diagnosis; and 5, severe motion artifact, diagnostically uninterpretable. Aortic valve motion artifact was determined from in-plane batch reformatted images using the following 5-point scale: 1, no motion artifact, aortic valve leaflets, orifice, and annulus clearly vis- 958 ible; 2, minimum motion artifact, satisfactory visualization of the annulus, leaflets, and orifice; 3, moderate motion artifact, adequate visualization of the annulus, orifice, and leaflets; 4, distinct motion artifact, inadequate visualization of the annulus, orifice, and leaflets; 5, severe motion artifact, no anatomic detail of the aortic wall or aortic valve. Banding artifact—Banding artifact occurs because of anatomic misregistration of contiguous cardiac structures obtained during sequential heartbeats. This artifact is identified as one or more parallel or straight lines in which the cardiac or aortic contour showed abrupt steplike distortions. In the thoracic aorta, banding artifact was assessed as follows: 1, no banding artifact, distinct aortic wall delineation and continuity; 2, minor banding artifact, slight discontinuity and misregistration on one margin of the aorta at one level; 3, moderate banding artifact, distinct misregistration and discontinuity of both margins of the aorta at one level; 4, severe banding artifact, stairstep artifact at multiple levels. In the aortic valve, banding artifact was assessed as follows: 1, no banding artifact, aortic annulus and leaflets intact and continuous; 2, minor banding artifact, slight misregistration and discontinuity of aortic annulus but the three aortic leaflets are clearly defined; 3, moderate banding artifact, distinct misregistration and discontinuity of the aortic annulus and three aortic valve leaflets are in the same cardiac phase; 4, severe banding artifact with prominent banding artifact across the aortic annulus and different segments of the aortic valve visualized in different cardiac phases. The differences in average heart rate and heart rate variability of the patients with images showing no banding artifact and those with images showing banding artifact were recorded. Statistical Analysis Statistical analysis was performed on commercially available software (SPSS, version 14.5, SPSS) for Microsoft Windows. Continuous variables were expressed as means and SDs. Comparative analysis for the numeric variables of two groups used the unpaired Student’s t test or the Mann-Whitney U test. For nonparametric data, the Kruskal-Wallis H test was used to compare scores of image quality of the thoracic aorta and aortic valve between the two groups. A p value of less than 0.05 was considered statistically significant. Results The prospective ECG-gating and retrospective ECG-gating techniques were successful in all patients without any complications (Fig. 2). The 40 patients in the prospective ECG-gated group (24 men, 16 women) ranged in age from 34 to 88 years (mean ± SD, 60.3 ± 13.78 years). All the patients had normal sinus rhythm at the time of examination. The retrospective ECG-gated group was composed of 40 patients (22 men, 18 women) who ranged in age from 25 to 93 years (mean, 55.3 ± 14.42 years). The mean AJR:193, October 2009 ECG Gating for Thoracic CT Angiography 4!",% 2ADIATION $OSE OF 4WO 'ROUPS Value (mean ± SD) Radiation Dose CTDIvol (mGy) Prospective Group Retrospective Group p 28.8 ± 2.12 74.7 ± 13.42 < 0.001 DLP mGy/cm mSv < 0.001 833.7 ± 115.77 2,547.3 ± 553.27 14.2 ± 1.96 43.3 ± 9.4 Note—CTDIvol = CT dose index volume, DLP = dose–length product. weight of the patients was 194 ± 50.64 lb (87 ± 22.79 kg) in the prospective ECG-gated group and 197 ± 60.34 lb (89 ± 27.15 kg in the retrospective ECG-gated group. No statistically significant difference between the two groups in either age or body weight was detected. The mean delay time between the beginning of the contrast injection and scanning acquisition was 24 ± 3.2 seconds in the prospective ECG-gated group and 23 ± 3.4 seconds in the retrospective ECG-gated group. The duration of acquisition in the prospective ECG-gated group (mean ± SD) was 15.4 ± 2.2 seconds and in the retrospective ECG-gated group, 13.4 ± 2.2 seconds. In the prospective ECG-gated group, 12 patients had a thoracic aortic aneurysm, eight of which involved the ascending aorta, and 11 patients had aortic dissection, only one of which involved the ascending aorta. One patient with a type B dissection had a postoperative pseudoaneurysm of the aortic root. In the retrospective ECG-gated group, 10 patients had an aortic aneurysm, five of which involved the ascending aorta and an additional two postoperative pseudoaneurysms of the aortic root. Of the seven aortic dissections, one involved the ascending aorta. Radiation Dose The average CTDIvol was 28.8 mGy in the prospective ECG-gated group and 74.7 mGy in the retrospective ECG-gated group. The DLP was 833.7 mGy/cm in the prospective ECG-gated group and 2,547.3 mGy/cm in the retrospective ECG-gated group. The radiation dose in the prospective ECG-gated group was significantly reduced compared with that of the retrospective ECG-gated group (p < 0.001) (Table 2). Contrast Volume In the prospective ECG-gated group, 34 patients received Visipaque 320 and six patients received Isovue 370. In the retrospective ECG-gated group, 28 patients received Visipaque and 12 patients received Isovue 370. AJR:193, October 2009 The average total contrast load in the prospective ECG-gated group was 109.1 ± 14.74 mL, which was significantly greater (p < 0.05) than that in the retrospective ECG-gated group (101.3 ± 10.45 mL). Contrast Enhancement Table 3 lists the attenuation values in the mid ascending aorta, mid aortic arch, mid descending aorta, and supraceliac abdominal aorta for both the prospective and retrospective ECG-gated groups. The mean attenuation value was significantly greater in the prospective ECG-gated group in the mid ascending aorta, mid aortic arch, and mid descending aorta (p < 0.001). There was no significant difference in the mean attenuation value at the level of the supraceliac abdominal aorta between the two groups (p = 0.122). Image Quality There was no difference in the extent of motion artifact in the thoracic aorta or at the aortic valve annulus between the prospective and retrospective ECG-gated groups (Fig. 3). Motion artifact scores in the thoracic aorta did not exceed 2 in either group. In the prospective ECG-gated group, one patient had an aortic valve motion artifact score of 2. All the other patients has scores of 1. Regarding banding artifacts, there was no statistically significant difference in the overall score between the two groups (p = 0.46). Banding artifacts were observed in studies of 10 patients in the prospective ECG-gated group and in studies of 13 patients in the retrospective ECG-gated group (Fig. 4). No significant difference was recorded between the prospective and retrospective ECG-gated groups in the banding artifact scores of the aortic valve (1.38 ± 0.774 vs 1.43 ± 0.78, respectively; p = 0.869) or of the thoracic aorta (1.38 ± 0.93 vs 1.45 ± 0.93; p = 0.712). The average heart rate of patients in the prospective ECG-gated group was 62.3 bpm. In patients whose images showed banding artifact, the average heart rate was 68.9 bpm, which was significantly higher than the average heart rate in those whose images were free of banding artifact (60.2 bpm; p < 0.05). In the retrospective ECG-gated group, the average heart rate was 63.4 bpm. In those with banding artifact, the average heart rate was 69.62 bpm, whereas it was 60.4 bpm in patients without banding artifact (Table 4). The heart rate variability of patients with banding artifact in the prospective ECG-gated group (18.0 bpm) was significantly greater than those without banding artifact (6.3 bpm; p < 0.05). In the retrospective ECGgated group, the average heart rate variability was higher in patients whose images showed banding artifact (11.18 bpm) than in those whose images did not show banding artifact (6.1 bpm). This difference was not statistically significant. Discussion Nongated thoracic aortic CT angiography is typically performed with a pitch value of 1.0–1.5. In a system with a 40-mm beam collimation, total acquisition can be performed within 3 seconds. However, motion of the ascending aorta can result in blurring of the vessel outlines and apparent thickening of the aortic wall, which could simulate a thrombosed dissection or obscure a mural flap (Fig. 5). In addition, the aortic valve and coronary arteries are not clearly displayed. Thoracic aortic CT angiography with cardiac gating can allow diagnostic evaluation of the 4!",% %NHANCED !ORTIC !TTENUATION 6ALUE AT &OUR -EASURED 3ITES Attenuation Value (HU) (mean ± SD) Prospective Group Retrospective Group p Mid ascending aorta Site 447.6 ± 93.02 350.2 ± 71.09 < 0.001 Mid aortic arch 413.6 ± 92.68 325.7 ± 67.13 < 0.001 Mid descending aorta 418.2 ± 94.98 327.6 ± 65.25 < 0.001 Supraceliac abdominal aorta 355.0 ± 129.88 306.2 ± 83.08 0.122 57.3 ± 34.43 31.5 ± 21.12 < 0.001 SD 959 7U ET AL A " C D Fig. 3—Prospective (A, B) and retrospective (C, D) ECG-gated acquisitions in patients with suspected aortic dissection demonstrate minimal motion blurring in both examinations. A, Prospective ECG-gated thoracic CT angiography with subvolume maximum intensity projection (MIP) in 65-year-old man with suspected aortic dissection in left anterior oblique projection. Minimum aortic wall blurring is noted, but image is satisfactory for diagnosis. B, Prospective ECG-gated thoracic CT aortography of same patient as shown in A. Reformatted image in plane of aortic valve shows minimal motion blurring of valve leaflets and aortic wall with normal coaptation of leaflets. Note small segment of proximal left coronary artery in left atrioventricular groove (arrow). Opacified left atrium is posterior to aorta, and there is residual contrast medium in right atrium, right ventricle, and pulmonary outflow tract. C, Retrospective ECG-gated thoracic aortic CT angiography in 61-year-old woman with suspected aortic dissection. Subvolume MIP in left anterior oblique projection shows minimal motion blurring of ascending aorta. Image is satisfactory for diagnosis. D, Retrospective ECG-gated CT thoracic aortography image shows minimal motion blurring in same patient as shown in C. Reformatted image in plane of aortic valve shows minimal motion blurring of valve leaflets and aortic wall. Opacified left atrium is left and posterior to aortic valve. Short segment of proximal left coronary artery (arrow) is imaged. Note mild uniform attenuation of right heart chambers after passage of contrast bolus. entire thoracic aorta, including the aortic sinus and aortic valve, and of the coronary arteries. Consequently, thoracic aortic CT angiography shows promise as a method for the comprehensive evaluation of the entire chest within a single breathhold including evaluation of the thoracic aorta, coronary arteries, and pulmonary arteries [14]. This practice mode includes both elective patient studies and evaluation of patients for acute chest pain in the emergency department in whom 960 E-blocker medication is administered IV by emergency department physicians. Radiation Dose The radiation dose from CT examinations has recently come to national attention. In 1989, CT accounted for 4% of diagnostic radiologic examinations performed in the United Kingdom, but CT contributed 40% of the collective population radiation dose from medical radiation [15]. In 1999, inves- tigators from one North American institution reported that 11.1% of the department workload was CT examinations but that CT contributed 67% of the collective patient radiation dose [16]. More recent reports indicate that CT accounts for 17% of departmental workload but accounts for 70–75% of the collective dose from medical radiation [17, 18]. These figures mirror the introduction of the first single-detector CT scanner in the late 1980s and MDCT in the late 1990s [19]. Radiation dose is becoming an issue of increasing importance for contrast-enhanced cardiac MDCT (coronary CT angiography) because 64-MDCT has shown promising results for coronary artery evaluation [20, 21]. Both the CTDIvol and the DLP were significantly reduced in the prospective ECGgated group compared with the retrospective ECG-gated group. In this study, ECG-dependent tube current modulation was used in the retrospective ECG-gated group. ECG tube current modulation is currently the most effective technical tool for radiation dose reduction during retrospective ECG-gated acquisitions without loss of image quality. Tube current is reduced during phases that are not usually necessary for interpretation of cardiac structure (i.e., phases associated with heart motion). During mid to late diastole, when motionfree images of the coronary artery and aortic valve may be acquired, the tube current is increased to the full prescribed radiation dose. ECG-dependent dose modulation may reduce radiation dose in retrospective ECGgated CT angiography by up to 50% [22]. Even with optimum ECG-dependent tube current modulation, the radiation dose from retrospective ECG-gated acquisition is still higher than that of prospective ECG-gated acquisition. In prospective ECG-gated acquisition, a single image is acquired sequentially during the same phase of the cardiac cycle at contiguous anatomic levels. A single exposure time is used for one image. In our study, the data acquisition time was lengthened beyond the required 175 milliseconds to allow the possible use of different midpoints of the R-R interval for data acquisition in the alternate heartbeats. Apart from phases selected for acquisition, the patient does not receive unnecessary radiation exposure during other phases of the cardiac cycle. Relatively slow heart rates (< 65 bpm) are optimal for both retrospective and prospective cardiac gating. A relatively slow heart rate with retrospective cardiac gating allows the AJR:193, October 2009 ECG Gating for Thoracic CT Angiography ble translation so that contiguous anatomy can be imaged in every alternate heartbeat. At faster heart rates, imaging may occur every third rather than every second heartbeat, thus prolonging the overall image acquisition. A " C D Fig. 4—Prospective (A, B) and retrospective (C, D) ECG-gated acquisitions in patients with suspected thoracic aortic aneurysms. Banding artifacts are shown on both types of examinations. A, Prospective ECG-gated thoracic CT angiography in 69-year-old woman with suspected thoracic aortic aneurysm. Subvolume maximum-intensity-projection (MIP) image in left anterior oblique projection shows misregistration stairstep artifact at junction of ascending aorta and aortic arch involving both anterior and posterior wall (left arrow). Identical stairstep artifact can be seen at same anatomic level in proximal descending thoracic aorta (right arrow). Additional minor stairstep artifacts are visible in mid and distal descending aorta. B, Prospective ECG-gated CT thoracic aortography with banding artifact in same patient as shown in A. Reformatted image in plane of aortic valve shows stairstep discontinuity of aortic wall predominantly anterior (single arrow). Diastolic phase is recorded in left half of aortic sinus with coapted right and left aortic valve leaflets, and systolic phase is recorded in right side of aortic sinus with indistinct and relatively blurred aortic valve leaflets. Left anterior descending coronary artery and proximal circumflex coronary artery (double arrows) are imaged in proximal atrioventricular groove. Left atrium is posterior to aortic root. Mild uniform attenuation of right heart chamber secondary to passage of contrast bolus is seen. C, Retrospective ECG-gated thoracic aortic CT angiography in 41-year-old woman with bicuspid aortic valve and aortic stenosis. Minimal banding artifact of aortic root and proximal ascending aorta is seen. Slight stairstep misregistration artifact (arrow) of anterior aortic wall through sinotubular junction and proximal ascending aorta are evident. D, Retrospective ECG-gated thoracic CT aortography with banding artifact of same patient as shown in C. Image shows partially calcified bicuspid aortic valve. Stairstep artifact (arrow) of right lateral margin of aortic root and mild blurring of anterior aspect can be seen. Remainder of aortic root and visualized valve leaflets are sharply delineated. Left atrium is posterior to aortic root. Mild uniform opacity of right heart chambers is seen after passage of contrast bolus. ECG-dependent tube modulation technique to be applied for the highest mA only in a relatively restricted mid to late diastolic phase of acquisition. With faster heart rates and a single-source system, a higher mA is usually applied to a longer segment of the cardiac cycle AJR:193, October 2009 because the optimum phase for image acquisition may be late systolic or mid to late diastolic in these circumstances; as a result, the patient receives a higher radiation dose. With prospective cardiac gating, heart rates of less than 65–70 bpm allow rapid ta- Contrast Load Adequate and uniform aortic enhancement throughout the entire period of image acquisition is highly desirable for the purpose of image processing and display, in which 3D postprocessing is frequently based on a threshold CT attenuation value. In our study, contrast material was injected at a fixed rate (6 mL/s) in both protocol groups, and the overall contrast volume was dependent on the combination of delay time from aortic arrival of contrast material until image acquisition plus the image acquisition interval. Aortic arrival of contrast medium can be determined by either a preliminary mini bolus or bolus-tracking software. In our practice, we use a preliminary mini bolus because it should be more precise. The image acquisition interval was longer in the prospective ECG-gated group than the retrospective ECG-gated group because the table speed in the prospective ECGgated group approximated 2 cm/s, whereas the table speed was 2.5 cm/s in the retrospective ECG-gated group. Faster table speed results in a shorter acquisition time. Aortic arrival of contrast material may be determined by a preliminary mini bolus technique or bolus-tracking software. The preliminary mini bolus technique was used in this study because it should be more precise. The increase in total contrast load in the prospective ECGgated group (109.1 ± 14.74 mL) versus the retrospective ECG-gated group (101.3 ± 10.45 mL) reflects the difference in the acquisition interval. Thoracic Aortic Attenuation The mean attenuation values in the mid ascending aorta, mid aortic arch, and mid descending aorta were higher in the prospective ECG-gated group than in the retrospective ECG-gated group. This increase in aortic attenuation, with the longer duration of acquisition and the longer injection interval, with prospective gating is supported by the work of Bae [23]. Bae reported that peak aortic enhancement was increased with a more prolonged contrast injection duration and that the time to peak aortic enhancement corresponded to the weighted sum of the injection duration and the time to peak test bolus enhancement. Of note in our study is that 961 7U ET AL 4!",% (EART 2ATE AND (EART 2ATE 6ARIABILITY "ETWEEN THE 4WO 'ROUPS Value (mean ± SD) Heart Rate Banding Artifact No Banding Artifact p 68.9 ± 10.58 60.2 ± 9.86 < 0.05 69.62 ± 13.49 60.4 ± 10.06 < 0.05 Prospective group 18.0 ± 19.4 6.3 ± 5.59 < 0.05 Retrospective group 11.18 ± 11.85 6.1 ± 6.35 NS Rate (bpm) Prospective group Retrospective group Variability (bpm) Note—NS = not significant. A " Fig. 5—Non-ECG-gated acquisition in 34-year-old woman with Ehlers-Danlos syndrome and left hemothorax. Neither thoracic aortic dissection nor aneurysm is present. A, Axial image of nongated acquisition for CT thoracic aortography shows pseudodissection (arrows) of ascending thoracic aorta with apparent curvilinear thickening of aorta wall on anterior and right posterior wall secondary to motion artifact. B, Nongated thoracic aortic CT angiography with subvolume maximum intensity projection in left anterior oblique projection shows motion blurring predominantly of anterior wall of ascending aorta and pseudointimal flap (arrow) at junction of aortic sinus and ascending aorta. no significant difference in aortic enhancement between the two groups was found at the level of the supraceliac aorta. This may be due to the longer scan duration of prospective ECG-gated acquisition, with the resultant data acquisition in the supraceliac aorta occurring after a relative plateau of thoracic aortic enhancement. With prospective ECG-gated acquisition, the time window for each segmental image data acquisition is separated by a separate heartbeat for table feed from the next image acquisition. The 2-second lag time between sequential acquisitions may result in prominent changes in aortic attenuation at the two sequential levels, resulting in prominent attenuation changes with linear margins at contiguous anatomic levels on subvolume MIP images obtained in the parasagittal plane. This appearance does not affect the diagnostic quality of the study. 962 Image Quality and Motion Artifact Motion artifacts that cause diagnostic uncertainty on CT images may occur in various parts of the body. Cardiac motion creates a challenge for diagnostic cardiac CT angiography. Cardiac motion also affects the perceived image quality of the adjacent anatomic structures—that is, the aortic valve and ascending aorta [24]. Pseudodissection of the thoracic aorta is a well-known pitfall. This artifact occurs at the left anterior and right posterior aortic circumference. A summation effect of both pendular and circular aortic motion produces a curvilinear thickening of the aortic wall, which simulates a dissection or hampers detailed visualization of an intraluminal flap [25–27]. Roos et al. [8] used a 4-MDCT scanner to show a significant reduction in motion-related artifacts for the entire thoracic aorta with ECG-gated MDCT. Maximum reduction was achieved at the level of the heart that physiologically moves the most—the aortic valve—followed by the ascending aorta. Thomas et al. [28] have also shown that ECG gating can be performed without delaying the workflow in an emergency setting, resulting in improved MDCT images of the thoracic aorta. In our study with both prospective and retrospective ECG-gated acquisitions, the aortic valve and ascending thoracic aorta were generally free of artifact with good to excellent image quality, reflecting fast gantry rotation speed and ECG-gated acquisition. Banding artifact is caused by misregistration between the software-detected ECG signal and cardiac motion. This artifact is also called synchronization or stairstep artifact. This artifact is identified as one or more parallel or straight lines along which the cardiac and aortic contours show abrupt steplike distortions. The most common reason that this artifact occurs is nonoptimal phase selection during either prospective or retrospective ECG-gated acquisition because of either tachycardia or cardiac arrhythmia [29]. Theoretically, prospective ECG gating is more dependent on a regular heart rate than retrospective ECG gating, and artifacts and misregistration occur in the presence of cardiac arrhythmias [29]. In this study, however, no significant difference between the two protocol groups was seen either in the number of patients with banding artifacts or in the banding artifact score. An increased heart rate was more likely to induce banding artifact in both groups. More prominent heart rate variation in both groups was observed in patients whose images were compromised by banding artifact than in those whose images were not. Because a heart rate of less than 65–70 bpm is recommended for optimal image quality in both prospective and retrospective ECG gating, E-blocker medication may be administered at the time of study. Study Limitations This study was not a prospective, randomized evaluation. Comparable patient groups were studied at different time intervals. Two different types of contrast material were used in both patient groups, although the distribution of isoosmolar and low-osmolar contrast agents was comparable between the two groups. The study was also limited to evaluating the thoracic aorta and did not evaluate the coronary arteries, which may be reconstructed AJR:193, October 2009 ECG Gating for Thoracic CT Angiography with equal fidelity using either a prospective or retrospective ECG-gated technique. Recently published studies have attested to the value of prospective cardiac gating in decreasing radiation dose for coronary artery CT angiography examinations while maintaining adequate image quality [12]. Retrospective cardiac gating may be used for quantitative analysis of left ventricular function and for studies of aortic valve motion. Thus, more complete patient information may be derived from a retrospective ECG-gated study. Nevertheless, a relatively low-dose prospective ECG-gated CT angiographic examination of the coronary arteries and thoracic aorta may be combined with either echocardiography, for functional evaluation of the left ventricle and aortic valve, or nuclear scintigraphy, for perfusion evaluation of the left ventricle. In conclusion, prospective ECG-gated thoracic CT angiography is a robust technique allowing high-quality diagnostic studies to be obtained with equivalent contrast material load but with a significantly reduced radiation dose compared with retrospective ECG-gated imaging. Optimum image quality for either technique is obtained with a relatively slow heart rate, which may require E-blocker medication. References 1. Hayter RG, Rhea JT, Small A, Tafazoli FS, Novelline RA. Suspected aortic dissection and other aortic disorders: multi-detector row CT in 373 cases in the emergency setting. Radiology 2006; 238:841–852 2. Bhalla S, West OC. CT of nontraumatic thoracic aortic emergencies. Semin Ultrasound CT MR 2005; 26:281–304 3. Yoshida S, Akiba H, Tamakawa M, et al. Thoracic involvement of type A aortic dissection and intramural hematoma: diagnostic accuracy—comparison of emergency helical CT and surgical findings. Radiology 2003; 228:430–435 4. Yamada I, Nakagawa T, Himeno Y, Numano F, Shibuya H. Takayasu arteritis: evaluation of the thoracic aorta with CT angiography. Radiology 1998; 209:103–109 AJR:193, October 2009 5. Matalanis G, Durairaj M, Brooks M. A hybrid technique of aortic arch branch transposition and antegrade stent graft deployment for complete arch repair without cardiopulmonary bypass. Eur J Cardiothorac Surg 2006; 29:611–612 6. Rubin GD, Armerding MD, Dake MD, Napel S. Cost identification of abdominal aortic aneurysm imaging by using time and motion analyses. Radiology 2000; 215:63–70 7. Vrtiska TJ, Fletcher JG, McCollough CH. Stateof-the-art imaging with 64-channel multidetector CT angiography. Perspect Vasc Surg Endovasc Ther 2005; 17:3–8 8. Roos JE, Willmann JK, Weishaupt D, Lachat M, Marincek B, Hilfiker PR. Thoracic aorta: motion artifact reduction with retrospective and prospective electrocardiography-assisted multi-detector row CT. Radiology 2002; 222:271–277 9. Bi X, Deshpande V, Simonetti O, et al. Three-dimensional breathhold SSFP coronary MRA: a comparison between 1.5T and 3.0T. J Magn Reson Imaging 2005; 22:206–212 10. Weber OM, Pujadas S, Martin AJ, et al. Free-breathing, three-dimensional coronary artery magnetic resonance angiography: comparison of sequences. J Magn Reson Imaging 2004; 20:395–402 11. Pereles FS, McCarthy RM, Baskaran V, et al. Thoracic aortic dissection and aneurysm: evaluation with non-enhanced true FISP MR angiography in less than 4 minutes. Radiology 2002; 223: 270–274 12. Earls JP, Berman EL, Urban BA, et al. Prospectively gated transverse coronary CT angiography versus retrospectively gated helical technique: improved image quality and reduced radiation dose. Radiology 2008; 246:742–753 13. Morin RL, Gerber TC, McCollough CH. Radiation dose and computed tomography of the heart. Circulation 2003; 107:917–922 14. White CS, Kuo D, Kelemen M, et al. Chest pain evaluation in the emergency department. AJR 2005; 185:533–540 15. Shrimpton PC, Edyrean S. CT scanner dosimetry. Br J Radiol 1998; 71:1–3 16. Mettler FA Jr, Wiest PW, Locken JA, Kelsey CA. CT scanning: patterns of use and dose. J Radiol Prot 2000; 20:353–359 17. Baker SR. Musings at the beginning of the hyperCT era. Abdom Imaging 2003; 28:110–114 18. Linton OW, Mettler FA Jr; National Council on Radiation Protection and Measurements. National conference on dose reduction in CT, with an emphasis on pediatric patients. AJR 2003; 181:321– 329 19. Paterson A, Frush D. Dose reduction in pediatric MDCT: general principles. Clin Radiol 2007; 62: 507–517 20. Leschka S, Alkadhi H, Plass A, et al. Accuracy of MSCT coronary angiography with 64-slice technology: first experience. Eur Heart J 2005; 26:1482–1487 21. Mollet NR, Cademartiri F, van Mieghem CA, et al. High-resolution spiral computed tomography coronary angiography in patients referred for diagnostic conventional coronary angiography. Circulation 2005; 112:2318–2323 22. Paul JF, Abada HT. Strategies for reduction of radiation dose in cardiac multislice CT. Eur Radiol 2007; 17:2028–2037 23. Bae KT. Peak contrast enhancement in CT and MR angiography: when does it occur and why? Pharmacokinetic study in a porcine mode. Radiology 2003; 227:809–816 24. Fleischmann D, Rubin GD, Paik DS, et al. Stairstep artifacts with single versus multiple detectorrow helical CT. Radiology 2000; 216:185–196 25. Qanadli SD, El Hajjam M, Mesurolle B, et al. Motion artifacts of the aorta simulating aortic dissection on spiral CT. J Comput Assist Tomogr 1999; 23:1–6 26. Loubeyre P, Angelie E, Grozel F, Abidi H, Minh VA. Spiral CT artifact that simulates aortic dissection: imaging reconstruction with use of 180° and 360° linear-interpolation algorithms. Radiology 1997; 205:153–157 27. Duvernoy O, Coulden R, Ytterberg C. Aortic motion: a potential pitfall in CT imaging of dissection in the ascending aorta. J Comput Assist Tomogr 1995; 19:569–572 28. Thomas S, Thomas E, Simon W, et al. Comparison of retrospectively ECG-gated and non-gated MDCT of the chest in an emergency setting regarding workflow, image quality, and diagnostic certainty. Emerg Radiol 2005; 12:19–29 29. Hsieh J. Advanced CT applications. In: Hsieh J. Computed tomography: principles, design, artifacts, and recent advances. Bellingham, WA: SPIE Optical Engineering Press, 2003:341–387 963