* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Quantum Mechanics Bohr`s model: - one of the first ones to use idea
Perturbation theory (quantum mechanics) wikipedia , lookup
EPR paradox wikipedia , lookup
Quantum state wikipedia , lookup
Wheeler's delayed choice experiment wikipedia , lookup
Coupled cluster wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Renormalization wikipedia , lookup
Coherent states wikipedia , lookup
Ensemble interpretation wikipedia , lookup
Aharonov–Bohm effect wikipedia , lookup
Density matrix wikipedia , lookup
Hidden variable theory wikipedia , lookup
Canonical quantization wikipedia , lookup
Lattice Boltzmann methods wikipedia , lookup
Elementary particle wikipedia , lookup
Identical particles wikipedia , lookup
Copenhagen interpretation wikipedia , lookup
Renormalization group wikipedia , lookup
Atomic theory wikipedia , lookup
Hydrogen atom wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Double-slit experiment wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Path integral formulation wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
Erwin Schrödinger wikipedia , lookup
Probability amplitude wikipedia , lookup
Dirac equation wikipedia , lookup
Schrödinger equation wikipedia , lookup
Particle in a box wikipedia , lookup
Wave function wikipedia , lookup
Wave–particle duality wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Matter wave wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
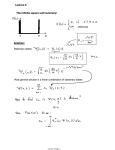
Quantum Mechanics Bohr's model: - one of the first ones to use idea of matter waves to solve a problem - gives good explanation of spectrum of single electron atoms, like hydrogen BUT: - does not explain even simple many electron systems, like Helium - does not explain intensity of spectral lines - does not help to explain structure of atoms, their binding to other atoms, or any physical of chemical properties of interacting atoms In 1925-26: - another more general approach to explain properties of microscopic system such as atoms is developed by Erwin Schrödinger, Werner Heisenberg, Max Born, Paul Dirac and others - it is called quantum mechanics by 1930s: - a vast number of experiments in physics and chemistry is successfully explained using this new theory - up to now the theory of quantum mechanics has been proven to be correct by many experiments - quantum mechanics is a very successful theory phys4.4 Page 1 Goal: - develop a theory that describes the wave properties of particles correctly steps: - use a complex valued wave function ψ to describe the particle - the modulus squared of ψ evaluated at a specific coordinate and time is proportional to the probability density P to find a particle at that coordinate and at that time - ψ∗ is the complex conjugate wave function to ψ properties of ψ: - the wave function is normalized - if ψ describes the particle and |ψ|2 its probability density then the total probability of finding the particle somewhere has to be unity (=1) for the wave function to be a valid description of the particle phys4.4 Page 2 properties of ψ: - single valued and continuous - single valued and continuous derivatives probability - to find a particle in a region of space (for simplicity in one dimension): Schrödinger equation: - fundamental equation of quantum mechanics - the wave equation for matter waves - is postulated and cannot be derived (like other fundamental equations, laws of thermodynamics, Newton's equation of motion, …) exercise: - work out some examples of wave equations to remember some of their properties and get some familiarity working with these phys4.4 Page 3 time-dependent Schrödinger equation: wave function: - for a free particle described by a plane wave along the xdirection with velocity v = λ ν - express ν and λ in terms of energy and momentum - thus - with the reduced Planck constant - This wave function describes a free particle. How to find the wave function describing a particle in the presence of external constraints, e.g. a potential resulting in some force acting on the particle? goal: - Find a fundamental differential equation the solution to which is the wave function describing the properties of the particle. phys4.4 Page 4 Schrödinger equation: - a strict derivation from first principles is not possible - let's develop some intuition trying to set up a wave equation for ψ by differentiating it with respect to x - and with respect to t - for v < c the total energy of a particle is given by - multiplying with ψ phys4.4 Page 5 - substitute expressions for E and p - time-dependent Schrödinger equation (1D) - in three dimensions (3D) - the potential function U contains all the external forces acting on the particle - solving the Schrödinger equation for ψ the probability density for the particle can be found for any coordinate and time - again: Schrödinger's equation is a postulate, there is no formal justification in the considerations presented above - but the physical world as probed in exquisite detail in experiments is very well described by this equation phys4.4 Page 6 Linearity: - the Schrödinger equation is linear in the wave function ψ - it contains no terms that involve higher powers of ψ or it's derivatives Superposition: - as a result if ψ1 and ψ2 are solutions of a S.E. the superposition of both is also a solution - a1 and a2 are complex coefficients - the superposition principle applies to matter waves in similar ways as to any other waves - the superposition principle gives rise to interference effects in quantum mechanics single solution terms two-solution terms phys4.4 Page 7 Expectation values: - all information about the particle is contained in the wave function ψ - we know already how to calculate the probability to find a particle at a certain coordinate and time - to extract other experimentally accessible quantities expectation values are calculated position expectation values: - classical center of mass for a collection of N particles with Ni particles at position xi - for a single particle with probability pi at position xi - in the continuous limit phys4.4 Page 8 general: - expectation values of quantity G(x) (that is not an operator!) - What about the expectation values of the momentum p and the energy E? - differentiate the free particle wave function ψ with respect to x and t - correspondence phys4.4 Page 9 Operators: - momentum operator - energy operator - total energy operator: - multiply with ψ to get back to the Schrödinger equation - expectation values of an operator O - for example for the momentum phys4.4 Page 10 example: - expectation value of the momentum p of a free particle time independent (steady state) Schrödinger equation - in many problems the potential U in the Schrödinger equation does not depend on time explicitly - in this case the Schrödinger equation can be simplified - note for the free particle wave function - for all time-independent problems the wave function can be separated into these two parts phys4.4 Page 11 time-independent (steady-state) Schrödinger equation: - in three dimensions - this steady-state Schrödinger equation may have one or more solutions ψi - the energy Ei corresponding to the different solutions ψi naturally explain the energy quantization observed in any stable timeindependent system phys4.4 Page 12 Eigenfunctions and eigenvalues of operators: - consider an operator O - if this equation holds ψn is an eigenfunction of the operator O with eigenvalue on - the equation above is also called an eigenvalue equation measurement postulate: - if a physical observable O corresponding to the operator O is measured the only possible measurement outcomes are any of the on phys4.4 Page 13 Hamilton operator: - the total energy operator E of a system can be written in terms of the momentum and potential energy operators p, U in - with the Hamilton operator H - the Schrödinger equation thus is a eingenvalue equation for the Hamiltonian H - the En are the eigenenergies of a system described by the Hamiltonian H, they are corresponding to the quantized energies of the system operators: position momentum potential energy kinetic energy phys4.4 Page 14 total energy Particle in a box: - a single particle trapped in a box with hard walls - one of the simplest possible examples of solving a Schrödinger equation - let's treat the box as one-dimensional (1D) - the particle is confined along the x-direction at x = 0 and at x=L - in the region 0 < x < L the potential is U = 0, elsewhere U=∞ - plot of potential U phys4.4 Page 15 Particle in a box: - task: find the wave function ψ describing the particle in the box by solving the Schrödinger equation - 2nd order ordinary differential equation for ψ - general solution - boundary condition ψ = 0 for x < 0 and x > L phys4.4 Page 16 boundary conditions: - continued - resulting allowed particle energies En - these are the allowed energy levels of a particle in a 1D box wave function: - the corresponding (not yet normalized) wave functions - normalization condition normalized wave function: phys4.4 Page 17 plots for different n: - wave functions ψn note: - probability densities |ψn|2 - the probability densities vary a lot with n - n = 1: largest probability in the center (x = L/2) - n = 2: zero probability in the center - classically probability is expected to be the same everywhere phys4.4 Page 18 expectation values: - position <x> - momentum <p> - the expectation value of the momentum vanishes! Why is that? - expectation for momentum from energy E = p2/2m - average momentum phys4.4 Page 19 momentum eigenfunction: - momentum eigenvalue equation - energy eigenfunctions are not momentum eigenfunctions - momentum eigenfunctions momentum eigenvalues phys4.4 Page 20 Finite Potential Well: - in a 1D potential well where U = 0 for 0 < x < L and U = U0 < ∞ the particle can penetrate into the wall - all natural potential wells have finite depth - consider a particle with energy E < U0 less than the depth of the potential - three regions in the problem - region II: U = 0 - region I and III: U0 > E phys4.4 Page 21 general solution: - in regions I and III - otherwise ψ would diverge - in region II - with boundary conditions continuity of wave function continuity of momentum phys4.4 Page 22 finite potential well wave functions: some notes: probability densities: - the particle is trapped forever - how does the particles ever get into the potential well? - how does it leave? - physical examples of a particle in a box phys4.4 Page 23 phys4.4 Page 24 phys4.4 Page 25 phys4.4 Page 26 phys4.4 Page 27