* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Mass versus molar doses, similarities and
Discovery and development of proton pump inhibitors wikipedia , lookup
Discovery and development of neuraminidase inhibitors wikipedia , lookup
Drug design wikipedia , lookup
Neuropharmacology wikipedia , lookup
Tablet (pharmacy) wikipedia , lookup
Pharmacogenomics wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Prescription costs wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Polysubstance dependence wikipedia , lookup
Drug discovery wikipedia , lookup
Prescription drug prices in the United States wikipedia , lookup
Drug interaction wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Psychopharmacology wikipedia , lookup
Pharmacognosy wikipedia , lookup
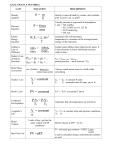
ORIGINAL ARTICLES Faculty of Pharmacy, Medical University of Gdańsk, Poland Mass versus molar doses, similarities and differences A. Chmielewska, H. Lamparczyk Received April 7, 2008, accepted June 11, 2008 Dr. Aleksandra Chmielewska, Medical University of Gdansk, Faculty of Pharmacy, Hallera 107, 80 416 Gdansk, Poland [email protected] or [email protected] Pharmazie 63: 843–848 (2008) doi: 10.1691/ph.2008.8578 Generally, they are two systems expressing the amounts of active substance in a given drug product, i.e. mass and molar dose. Currently, the dose system based on the mass is widely used in which doses are expressed in grams or milligrams. On the other hand, the molar dose system is in direct relation to the number of molecules. Hence, the objective of this work was to compare both systems in order to find their advantages and disadvantages. Active substances belonging to the groups of antibiotics, nootropic agents, b-blockers, vitamins, GABA-analog, COX-2 inhibitors, calcium channel antagonists, benzodiazepine receptor agonists, lipid-modifying agents (fibrates), non-steroidal anti-inflammatory drugs (profens), estrogens, neuroleptics, analgesics and benzodiazepines were considered. Moreover, products containing two active substances were also taken into account. These are mixtures of hydrochlorothiazide with active substances influencing the renin-angiotensin system and combined oral contraceptives. For each active substance, belonging to the groups mentioned above molar doses were calculated from mass doses and molar mass. Hence, groups of drugs with a single active substance, drugs with similar pharmacological activities, pharmaceutical alternatives, and drugs with a single active ingredient manufactured in different doses were compared in order to find which dose system describes more adequately differences between and within the groups mentioned above. Comparisons were supported by a number of equations, which theoretically justify the data, and relationships derived from calculations. 1. Introduction The problem of choosing a dose system is probably as old as the beginning of modern Medicinal Chemistry. It reached the time of Paul Ehrlich and his dilemmas how to dose the famous “magic bullet” substances. He also envisioned the dance of molecules in and around cells as the key to the chemistry of disease and its treatment. Remarkably, he mentioned molecules but not masses. There are two possibilities to express the amount of active substance in a drug. Firstly as a mass and secondly as molar dose. Currently widely accepted are mass doses expressed in grams or milligrams. Mass doses are considered to be comprehensive, uniform, and easier to understand. They fully conform to SI (Système International). Metric weight and volume measurements are universally used, easier to be explained, safer to apply, and are generally used in textbooks and clinical practice. Any attempt to change the mass to the molar system provoked discussion among medical professionals. A typical example is the change by major drug manufacturers with regard to the labelling and strength of calcium chloride solutions from weight per volume (w/v) to millimoles. The new solutions were found as not equivalent to familiar standard 10% and 20% w/v (Chamberlain et al. 1976). In response Roden and Mander (1976) argued that molar dosing is more adequate. The problem of choosing mass or molar dosing system was also considered by the major drug registration agenPharmazie 63 (2008) 11 cies. According to FDA, the rate and extent of absorption of the test drug do not show a significant difference from the rate and extent of absorption of the reference drug when administrated at the same molar dose of therapeutic ingredient under similar experimental conditions in either a single dose or multiple doses [Federal Food, Drug and Cosmetic Administration Act, section 505(j)(7)(B)]. The need of molar dosing in bioequivalence studies is also emphasised by CPMP (Note for guidance on the investigation of bioavailability and bioequivalence, (CPMP/EWP/ QWP/1401/98, London 26 July 2001.). Despite this, it is difficult to find examples where molar doses are used in experiments performed on human beings. Sometimes the pharmacokinetic parameters are expressed in moles while the initial doses in mass units (Richter et al. 2005). Molar dosage is often used in preclinical studies, performed on experimental animals (D’Cruz and Uckun 2001; Mamidi et al. 2002). 2. Investigations and results 2.1. Molar doses The molar dose system has great appeal in logic, because number of active substance molecules (n) is directly proportional to molar dose (MD). Eqs. (1) and (2) NA ¼ 6:02 1023 ð1Þ 843 ORIGINAL ARTICLES 2.2.2. Drug products with one active substance of the same pharmacologic class Fibrates belong chemically to the group of amphipathic carboxylic acids, which are antagonists of peroxisome proliferator-activated receptor (PPAR-alpha). Pharmacologically, fibrates belongs to of lipid-modifying agents. Treatment with fibrates results in a substantial decrease in plasma levels of triglycerides and is usually associated with a moderate decrease in LDL cholesterol and an increase in HDL cholesterol concentrations (Staels et al. 1998). Fibrates also appear to have a beneficial effect on the insulin resistance featured by the metabolic syndrome (Wysocki et al. 2004). Because of similarities in chemical structure and pharmacological action, they are often subject to substitution. Clinically, different individual response or tolerance of particular chemical molecules might justify this. Nevertheless, besides therapeutic reasons, the number of active molecules expressed by molar dose should also be taken into account. Despite usually applied mass doses, the molar dose is highest for gemfibrozil and lowest for ciprofibrate. It is noteworthy, that the mass doses ratio for gemfibrozil and ciprofibrate is 6, whilst the molar doses ratio is 6.93. It proves that in this case mass and molar doses are incomparable. Moreover, considering only molar doses ratio it means, that in case of gemfibrozil the number of potentially active molecules is 14% higher in comparison to ciprofibrate. Even more illustrative is the comparison of mass and molar doses in case of the probably most commonly used fibrates i.e. gemfibrozil and bezafibrate. The mass and molar dose ratios are 3 and 4.33 respectively. Thus, replacing bezafibrate 200 mg by gemfibrozil 600 mg the doses are changed not three times, but in fact 4.33 times. Profens belong to the family of non-steroidal anti-inflammatory drugs. Chemically, they are derivatives of 2-arylpropionic acid and they are all chiral. However, with the exception of S-naproxen, the profens have been recently used in clinical practice as racemic mixture (Evans 1996). Widely applied members of these active substances are listed in Table 2. Remarks concerning profens might be similar to those concerning fibrates. They possess closely related chemical structures and their pharmacological activities are roughly the same. In many countries, ibuprofen is sold without prescription, and therefore it is frequently used. Replacement of one profen by the other is normal clinical practice. Taking into account profens listed in Table 2 the molar dose is highest for tiaprofenic acid and lowest for ketoprofen. The ratio of mass to molar dose for tiaprofenic acid 300 mg and ketoprofen 100 mg is 3 and 2.9 respectively, thus negligible small, on the other hand in the case of ibuprofen and ketoprofen is 1 and 1.23 re- As examples of active substances belonging to these group, fibrates and profens were chosen, as listed in Table 2. Table 2: Fibrates and profenes as examples for compounds, which display similar pharmacological activities Table 1: Examples of single active drug substances Active substance NA denote Avogadro number 23 n ¼ 6:02 10 MD ð2Þ The molar dose can easily be calculated from mass dose (m) and molar mass (M): Eq. (3). m MD ¼ ð3Þ M Generally, for drugs with different active ingredients molar doses are not equal to mass doses. Although, the mass dose can be calculated from the molar dose when molar mass of the active substance is known. Thus, molar dose is more informative because it includes knowledge about molar mass. The problem is not only theoretical but also practical. For example, the commonly used beta-blocker propranolol is usually applied as a salt propranolol hydrochloride. Molecular weights of the base and salt are 259.3 and 294.7 respectively. Thus for the widely used 40 mg mass dose, molar dose will differ from 0.154 mmoles for base to 0.135 mmoles for salt. Therefore, the relation to base is completely unrealistic so only the last value includes true information about the number of propranolol molecules in the 400 mg mass dose. This will be discussed in more detail in the section concerning pharmaceutical alternatives. 2.2. Calculations 2.2.1. Drugs with single active substance Compounds of this class are listed in Table 1 according to their molar masses in increasing order. It can be seen the range of molar masses is very narrow. In Table 1 examples of active substances possessing various molar masses from 122 to 862 g/mol are listed. Active ingredients usually do not extend a molar mass of 500 g/mol, because substances with higher M values have low absorption rate when administrated orally. Molar doses include also information on mass dose, which is chosen to achieve a predicted therapeutic effect. Recently, the strength of many drugs is changed due to the application of nanotechnology in active ingredient manufacture. These include micronisation, use of fine emulsions, carbon nanomaterials such as fullerenes, cyclodextrins, calixarenes, crown ethers and so on. Exploration of this concept leads directly to nanopharmacology in strictly scientific sense (Emerich and Thanos 2006; Medina et al. 2007). Active substance M (g/mol) m (mg) Nicotinic acid (niacin) 122.1 500 Piracetam 142.2 1200 Gabapentin 171.2 600 Atenolol 266.3 25 Etodolac 287.4 400 Temazepam 300.7 15 Felodipine 384.3 5 Zopiclone 388.8 7.5 Alfacalcidol 400.6 0.25 Erythromycin 862.1 200 844 MD mmol Number of molecules (n) 4.095 8.439 3.505 0.0940 1.394 0.0498 0.013 0.0192 0.000624 0.232 2.465 1021 5.057 1021 2.124 1021 5.659 1019 8.392 1020 2.998 1019 7.83 1018 1.156 1019 3.750 1017 1.397 1020 Fibrates Clofibrate Gemfibrozil Ciprofibrate Fenofibrate Bezafibrate Profens Ibuprofen Naproxen Flurbiprofen Ketoprofen Tiaprofenic acid M (g/mol) m (mg) MD mmol Number of molecules (n) 242.7 250.3 289.2 360.8 361.8 500 600 100 160 200 2.060 2.397 0.346 0.443 0.553 1.240 1021 1.443 1021 2.083 1020 2.667 1020 3.329 1020 206.3 230.2 244.3 254.3 260.3 100 250 100 100 300 0.485 1.086 0.409 0.393 1.152 2.920 1020 6.538 1020 2.462 1020 2.366 1020 6.935 1020 Pharmazie 63 (2008) 11 ORIGINAL ARTICLES Table 3: Mass and molar doses of pharmaceutical alternatives Active substance M (g/mol) m (mg) MD mmol Number of molecules (n) Estradiol Estradiol valerate Pridinol hydrochloride Pridinol mesylate Amlodipine Amlodipine besilate Isosorbide mononitrate Isosorbide dinitrate Amantadine hydrochloride Amantadine sulfate Quetiapine Quetiapine fumarate 272.4 356.5 331.5 391.5 409.1 547.6 191.1 236.1 187.7 400.5 383.5 883.1 0.5 2 2 5 4.18 10 10 10 10 100 100 100 115.1 0.073 0.056 0.150 0.106 0.0244 0.0176 0.523 0.424 5.33 2.50 0.260 0.1300 0.439 1019 0.337 1019 0.903 1019 0.638 1019 0.147 1019 0.106 1019 31.48 1018 25.50 1018 32.07 1019 15.02 1019 15.65 1020 7.826 1020 spectively, what means 23% difference. This should be always considered as one of the important factors when decision of replacement is taken or combination therapy is applied. 2.2.3. Pharmaceutical alternatives Drug products are considered pharmaceutical alternatives if they contain the same therapeutic moiety, but are different salts, esters, or complexes of that moiety. FDA document includes also drugs in different forms e.g. tablets vs. capsules (GCP Module: FDA Regulations for Clinical Research). Nevertheless in many cases it was found that the bioavailabilities of active substances from tablets and capsules are equal (Schall et al. 1995; Chen et al. 2005; Peres et al. 2006). On the other hand some investigations show differences. Tianidazine tablets and capsules were found bioequivalent in the fasting state. Administration of these drug forms in the fasting state decreased bioavailability to an higher extent from tablets (Shah et al. 2006) Changing in pharmacokinetic profiles were also observed in comparative studies of tramadol slow release (SR) tablets and capsules (Cnota et al. 2005). Therefore, in this work discussion was restricted only to the first part of FDA definition, because choice of dosage system precede the choice of the drug form and is in part independent. In Table 3 examples of pharmaceutical alternatives were listed. Large differences between mass and molar doses are observed in case of esters. Estradiol in 2 mg mass dose is manufactured and marketed in form of pure steroid and its valerate ester. The difference between these two forms expressed as molar doses is 23.3%. Under supervision of registration agencies most of the drugs, which are salts now are properly labelled as equivalents of active moiety or to other commonly accepted salt. This can be done by simple calculation using the ratio of molar masses. For example quetiapine is present in tablets as fumarate salt in which two molecules of quetiapine base are connected to one molecule of fumaric acid. The molar mass for the salt and base is 883.1 and 383.5 g/mol respectively. The ratio of molar masses is given by Eq. (4). MS 883:1 ¼ ¼ 1:15 MB 2 383:5 2.2.4. Drugs with single active ingredient manufactured in different doses Currently, it is common practice, to manufacture drug products in a wide range of doses. Examples of such drugs are given in Table 4. For a given active substance ratios of mass and molar doses are equal. This is confirmed by Eqs. (4) and (5) MD1 m1 m2 m1 ¼ ¼ ð4Þ MD2 M M m2 MD1 m1 m3 m1 ¼ ð5Þ ¼ MD3 M M m3 for the same drug with doses 1, 2 and 3. In practice this means, that a dose of 30 mg atenolol is two times higher than a dose of 15 mg, both in sense of mass and molar dosage. 2.2.5 Drug products with two active substances ð4Þ Using molar mass coefficient it can be easy to calculate the amount of the base, which should be used to obtain tablets of a predicted mass dose. For 100, 150 and Pharmazie 63 (2008) 11 300 mg tables it will be 115, 172 and 344 mg respectively. Nevertheless currently three systems coexists, recently imposed by all major registration agencies (e.g. FDA, EMEA) in which molar mass coefficients are applied and doses are expressed as mass doses in relation to the active moiety. In most pharmacopoeias the active substance is accurately described and molar masses are related to salts, although in some cases recalculation to the active moiety is also given. In textbooks, in which the name of salt or base is mentioned, it might be assumed that the mass dose is related to salt and base respectively. However, it strongly depends upon the quality of a particular textbook. For example, amlodipine in some textbooks is mentioned as amlodipine besilate, in others as amlodipine, although the difference between mass and molar dose is substantial. However, all above mentioned systems are well justified, and it should be emphasised, that they are not univocal and confusing. Very frequently drugs are composed of two or more active substances. Typical examples are oral contraceptives, mixtures of diuretics with substances used for the treatment of hypertension, acetylsalicylic acid with vitamin C, paracetamol with codeine and many others. In this case ratios of 845 ORIGINAL ARTICLES Table 4: Examples of drugs manufactured in different doses Active substance M (g/mol) m (mg) MD mmol Pindolol 248.3 Atenolol 266.3 Propranolol hydrochloride 295.8 Oxazepam 286.7 Thioridazine hydrochloride 407.0 5.0 10.0 25 50 100 10 20 40 60 80 90 10 15 30 10 15 25 50 100 150 200 0.0201 0.0403 0.094 0.188 0.376 0.0339 0.0676 0.136 0.203 0.270 0.304 0.0348 0.0523 0.105 0.0296 0.0443 0.0739 0.148 0.296 0.444 0.592 molar and mass doses are unequal. MDA mA 6¼ ð6Þ MDB mB were subscripts A, B denotes components of the mixture. The general relationship between mass and molar doses is described by Eq. (7). MDA mA mB MB mA ¼ ¼ ð7Þ MDB MA MB MA mB It is noteworthy, that ratio of molar masses MB/MA for a given active substance components is constant, thus; MB ¼ const ¼ A ð8Þ MA MDA mA ¼A ð9Þ MDB mB This means that the molar dose ratio can be calculated directly from the mass dose ratio when coefficient A (Eq. (9)) is known. The well known diuretic, hydrochlorothiazide, is frequently present as a component in combination therapy with various antihypertensive agents, particularly with those which influencing the renin angiotensin system. Table 5 lists mixtures of inhibitors the angiotensin converting enzyme (ACE) and substances influencing angiotensin II receptor with hydrochlorothiazide. Examples 1–12 are ACE inhibitors belonging to a subclass known as dicarboxylate-containig ACE inhibitors, possessing similar chemical structure. They are all chiral and are manufactured as a single stereoisomer. Most of them are prodrugs, for example enalapril is metabolised in vivo to its active form enalaprilat. Comparatively, all ACE inhibitors of this subclass have similar antihypertensive efficacy when equivalent doses are administered. Therefore they are often replaced by each other. Considering data in Table 5 it can be find that the mass doses differ from molar doses in each case for both components. This is a general rule concerning also drugs with one active substance. An important factor is also the ratio of active substances calculated on the basis of mass and molar doses. For various components they are different. These differences increased with coefficient A, and obviously with the mass dose ratio Eq. (9). Even when the Table 5: Complex drugs containing hydrochlorothiazide and active substances influencing renin-angiotensin system No 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 846 sub. Active substance MB/MA m (mg) MD (mmol) mA/mB MDA/MDB A B A B A B A B A B A B A B A B A B A B A B A B A B A B A B Hydrochlorothiazide Ramipril Hydrochlorothiazide Ramipril Hydrochlorothiazide Enalapril maleate hydrochlorothiazide Enalapril maleate Hydrochlorothiazide Cilazapril Hydrochlorothiazide Benazepril hydrochloride Hydrochlorothiazide Benazepril hydrochloride Hydrochlorothiazide Benazepril hydrochloride Hydrochlorothiazide Quinapril Hydrochlorothiazide Quinapril Hydrochlorothiazide Moexipril hydrochloride Hydrochlorothiazide Moexipril hydrochloride Hydrochlorothiazide Losartan potassium Hydrochlorothiazide Valsartan Hydrochlorothiazide Valsartan 1.399 25 12.5 25 5 25 10 12.5 10 12.5 5 6.25 5 12.5 10 25 20 12.5 10 12.5 20 12.5 7.5 25 15 12.5 50 12.5 80 12.5 160 0.0840 0.0300 0.0840 0.0120 0.0840 0.0203 0.0420 0.0203 0.0420 0.0119 0.0209 0.0108 0.0420 0.0216 0.0840 0.0433 0.0420 0.0228 0.0420 0.0456 0.0420 0.0140 0.0840 0.0280 0.0420 0.108 0.0420 0.184 0.0420 0.368 2 2.8 5 7.0 2.5 4.14 1.25 2.07 2.5 3.50 1.25 1.94 1.25 1.94 0.625 0.919 1.67 3.00 0.25 0.389 0.156 0.228 0.0781 0.114 1.654 1.402 1.548 1.473 1.797 1.548 1.463 Pharmazie 63 (2008) 11 ORIGINAL ARTICLES Table 6: Examples of various oral contraceptives No sub. Active substance MB/MA m (mg) MD (mmol) 1 A B A B A B A B A B A B A B Norgestimate Ethinylestradiol Desogestrel Ethinylestradiol Dienogest Ethinylestradiol Gestoden Ethinylestradiol Levonorgestrel Ethinylestradiol Levonorgestrel Ethinylestradiol Levonorgestrel Ethinylestradiol 0.802 250 35 150 30 2000 30 75 30 250 50 125 50 150 30 0.676 0.118 0.483 0.101 6.42 0.101 0.242 0.101 0.8 0.169 0.4 0.169 0.48 0.101 2 3 4 5 6 7 0.955 0.952 0.955 0.948 0.948 0.948 ratios of mass doses are equal the ratios of molar doses might differ. This is exemplified by composition nos. 3 and 5. Clinically, this means that great care should be taken when a compound replaces a previously chosen preparation. On the other hand, when the components of a complex drug and mass doses ratio are the same changes of doses do not affect both mass and molar doses ratios. This is exemplified by combined oral contraceptives (COCs). COCs usually contain two groups of hormones i.e. estrogens and progestins. As estrogen usually ethinylestradiol is used whereas progestins are various. COCs suppress ovulation, thicken the cervical mucus, change the endometrium making implantation less likely and reduce sperm transport in the upper genital tract. They are considered to be safe particularly when used in low doses (Stone 1993; Bachmann et al. 2004). COCs are also used in treatment of premenstrual dysphoric disorders (Kahn and Halbreich 2001; Yonkers et al. 2005). In Table 6 examples of various oral contraceptives are listed. Inspection of the data presented in Table 6 leads to the conclusion that the ratios of MB to MA are nearly equal to unity, except preparation 1. Therefore, for the other preparations listed in Table 6, the relationship described by Eq. (9) is valid. This is expressed by Eq. (10). MB MDA mA ffi1 ffi MA MDB mB ð10Þ Concluding, under these circumstances, the molar dose and mass dose ratios are nearly equal. 3. Discussion The starting point of this work was the Avogadro number and its relation to the number of molecules (n) in one mole or its fraction. Assuming that active substances influence the substructures of any living organisms, including human, on the molecular level number n should be the basic factor, which justifies the dose of a given active substance. Only the molar dose system is directly related to n. In this work n was calculated for many active ingredients belonging to different chemical and pharmacological classes. Interestingly, number n, in actually marketed drugs, is in the very narrow range from 1021 to 1017. Bearing this in mind a question rose what happened with such huge amount of molecules, if they are unnecessarily applied. A number of answers may be given. The theory of a molecular level of interactions between substructures of Pharmazie 63 (2008) 11 mA/mB MDA/MDB 7.14 5.73 5.0 4.78 66.67 63.56 2.5 2.4 5.0 4.73 2.5 2.37 5.0 4.75 the organism and the active substance does not work. There are some suggestions, that the active ingredients react on a supramolecular level (Lamparczyk et al. 1994; Zarzycki et al. 1997), however in such a case the number of reacting molecules should be lower. Therefore, it still should be assumed that not all of the active ingredients reach the receptor site. In such a case a new problem is becoming important, i.e. how to improve the drug delivery from the intestinal tract to the receptor site. Some answers concerning this problem are given on the ground of nanotechnology, or more precisely, nanotechnology-based drugs (Emerich 2006; Medina et al. 2007). These new drug products improve the distribution of active substance and therefore, the number of acting molecules might be smaller. The main problem concerning the new drugs or active drug substances is their not fully predicted toxicity (Clancy 2002; Cui 2005; Kipen and Laskin 2005; Oberdorster 2004). Moreover it is assumed that nanotechnology-based drugs are also acting on a molecular level. This confirms the importance for full acceptance of the molar dosing system. Finally, a limited series of complex drugs with two active substances were considered. In this case the ratio between molar masses MB to MA plays an important role. When it approaches unity the ratios between molar and mass doses of two active substances A and B are starting to be equal. References Bachmann G, Sulak PJ, Sampson-Landers C, Benda N, Marr J (2004) Efficacy and safety of a low-dose 24-day combined oral contraceptive containing 20 micrograms ethinylestradiol and 3 mg drospirone. Contraception 70: 191–198. Chamberlain DA, Clark AJ, Sheldon J (1976) Letter to the Editor. Br Med J 30 October: p 1068. Chen CY, Lu CL, Luo JC, Chang FY, Lee SD, Lai YL (2005) Esomeprazole tablet vs omeprazole capsule in treating erosive esophagitis. World J Gastroenterol 11: 3112–3117. Clancy L, Goodman P, Sinclar H, Dockery DW (2002) Effect of air-pollution control on death rates in Dublin, Ireland: an intervention study. Lancet 360: 1210–1214. Cnota PJ, Nowak H., Tagarro I, Erb K, Schurer M, Schulz HU, Maus J (2005) Tramadol SR formulations: Pharmacokinetic comparison of a multiple units dose (capsule) versus a single unit dose (tablet). Clin Drug Investig 25: 435–443. Cui D, Tian F, Ozkan CS, Wang M, Gao H (2005) Effect of single wall carbon nanotubes on human HEK293 cells. Toxicol Lett 155: 73–85. D’Cruz OJ, Uckun FM (2001) Short-term (13-week) toxicity study of 5bromo-6-methoxy-5,6-dihydro-30 azidothymidine-50 -(p-bromophenyl)methoxyalanil phosphate (WHI-07), a novel anti-HIV and contraceptive agent, in B6C3F1 mice. Toxicological Sciences 60: 373–378. Emerich DF, Thanos CG (2006) The pinpoint promise of nanoparticlebased drug delivery and molecular diagnosis. Biomol Engin 23: 171– 184. 847 ORIGINAL ARTICLES Evans AM (1996) Pharmacodynamics an pharmacokinetics of the profens: enantioselectivity, clinical implications and, special reference to S(+) ibuprofen. J Clin Pharmacol 36: 7S-15S. Kahn LS, Halbreich U (2001) Oral contraceptives and mood. Expert Opin Pharmacother 2: 1367–1382. Kipen HM, Laskin DL (2005) Smaller is not always better: nanotechnology yields nanotoxicology. Am J Physiol Lung Cell Mol Physiol 289: L696-L697. Lamparczyk H, Zarzycki PK Nowakowska J (1994) Effect of temperature on separation of Norgestrel enantiomers by high-performance liquid chromatography. J Chromatogr A 668: 469–473. Mamidi RN, Mullangi R, Kota J, Bhamidipati R, Khan AA, Katneni K, Datla S, Singh SK, Rao KY, Rao CS, Srinivas NR, Rajagoplan R (2002) Pharmacological and pharmacokinetic evaluation of celecox prodrugs in rats. Biopharm Drug Disp 23: 273–282. Medina C, Santos-Martines MJ, Radomski A, Corrigan OI, Radomski MW(2007) Nanoparticles: pharmacological and toxicological significance. Br J Pharmacol 150: 552–558. Oberdorster E (2004) Manufactures nanomaterials (fullerenes, C60) induce oxidative stress in the brain of juvenile largemouth bass. Environ Health Perspect 112: 1058–1062. Peres M, Cardenas W, Ramires G, Peres M, Restrepo P ( 2006) A comparative, cross-over, double blind, randomized study for bioequivalence assessment between two formulations of valsartan capsules vs tablets. Colombia Med 37: 114–120. Richter K, Kanniess F, Biberger C, Nave R, Magnussen H (2005) Comparison of the oropharyngeal deposition of inhaled ciclesonide and fluticasone propionate in patient with asthma. J Clin Pharmacol 45: 146–152. 848 Roden SM, Mander GA (1976) Molar dosage of calcium chloride. Br Med. J 20 November: p. 1255. Shah J, Wesnes KA, Kovelesky RA, Henney HR (2006) Effects of food on the single-dose pharmacokinetics/pharmacodynamics of thiazanidine capsules and tablets in healthy volunteers. Clinical Therap 20: 1308–1317. Schall R, Muller FO, Duursema L, Groenewoud G, Hundt HK, Middle MV, Mogilnicka EM Swart KJ (1995) Relative bioavailability of rifampicin, izoniazid and ethambutol from a combination tablet vs. concomitant administration of a capsule containing rifampicin and a tablet containing isoniazid and ethambutol. Arzneimittelforschung 45: 1236–1239. Staels B, Dallongeville J, Auverx J, Schoonjans K, Leitersdorf E, Fruchart JC (1998) Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation 98: 2088–2093. Stone S (1993) Clinical review of a monophasic oral contraceptive containing desogestrel and ethinylestradiol. Int J Fertil Menopausal Stud 38 suppl: 117–121. Wysocki J, Belowski D, Kalina M, Kochanski L, Okopien B, Kalina Z (2004) Effects of micronized fenofibrate on insulin resistance in patients with metabolic syndrome. Int J Clin Pharmacol Ther 42: 212–217. Yonkers KA, Brown C, Pearlstein TB, Foegh M, Sampson-Landers C, Rapkin A (2005) Efficacy of a new low-dose oral contraceptive with drospirenone in premenstrual dysphoric disorder. Obstetr Gynecol 106: 492–501. Zarzycki PK, Nowakowska J, Chmielewska A, Wierzbowska M, Lamparczyk H (1997) Thermodynamic study of the retention behaviour of selected macrocycles using reversed phase high-performance thin-layer chromatography plates and metanol-water mobile phases. J Chromatogr A 787: 227–233. Pharmazie 63 (2008) 11