* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Transition metals and coordination chemistry
Metastable inner-shell molecular state wikipedia , lookup
List of phenyltropanes wikipedia , lookup
Organic chemistry wikipedia , lookup
History of chemistry wikipedia , lookup
History of electrochemistry wikipedia , lookup
Computational chemistry wikipedia , lookup
Oxidation state wikipedia , lookup
Resonance (chemistry) wikipedia , lookup
Molecular orbital diagram wikipedia , lookup
Atomic theory wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Geochemistry wikipedia , lookup
Thermoelectric materials wikipedia , lookup
Ligand binding assay wikipedia , lookup
Bond valence method wikipedia , lookup
Electrochemistry wikipedia , lookup
Jahn–Teller effect wikipedia , lookup
Homoaromaticity wikipedia , lookup
Surface properties of transition metal oxides wikipedia , lookup
History of molecular theory wikipedia , lookup
Electron configuration wikipedia , lookup
Metal carbonyl wikipedia , lookup
Electrical resistivity and conductivity wikipedia , lookup
Chemical bond wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Spin crossover wikipedia , lookup
Metallic bonding wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Stability constants of complexes wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
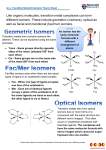
21 Transition metals and coordination chemistry Physical properties: (1) metallic luster (2) high electrical and thermal conductivity (3) wide range of melting points (W: 3400oC, Hg: <25oC) (4) hardness (5) wide range of reactivity towards O2 (forming oxides) (Fe reacts easily with O2 while coinage metals do not react with O2) 23 24 25 26 27 28 Ti V Cr Mn Fe Co Ni 3d14s2 3d24s2 3d34s2 3d54s1 3d54s2 3d64s2 3d74s2 3d84s2 45 45 Y Zr Nb Mo Tc Ru Rh Pd [Kr] 4d15s2 4d25s2 4d45s1 4d55s1 4d55s2 4d75s1 4d85s1 4d10 La Hf Ta W Re Os Ir Pt [Xe] 5d16s2 4f145d26s2 4f145d36s2 4f145d46s2 4f145d56s2 4f145d66s2 4f145d76s2 4f145d96s1 39 Show the periodic table (oxidation states and electron configurations) 22 Sc [Ar] 57 40 41 72 42 73 74 To write the electronic structure for Co2+: Co Co2+ [Ar] 3d74s2 [Ar] 3d7 43 44 75 76 77 78 To write the electronic structure for V3+: V [Ar] 3d34s2 V3+ [Ar] 3d2 Titanium (0.6% by mass of Earth's crust) Titanium metal - characterized by low density, high strength, chemical inertness- used as a structural element in high-demand uses such as jet engines, high-performance bicycles, etc. Chromium (Cr) - (greek chroma, color) Cr(0) (metal) is used for making steel. Cr(VI) and Cr(III) are used for chrome plating, dyes and pigments, leather tanning, and wood preserving. Chromium enters the air, water, and soil mostly as Cr(III) and Cr(VI). Cr(III) strongly attaches to soil, but a small amount can dissolve in water and move deeper to underground water reservoirs - pollutant. The colors of many gemstones come from Cr(III) impurities Ruby: Cr3+ in Al2 O3 Large crystals of copper sulfate The Copper(II) sulfate salt exists as a series of compounds that differ in their degree of hydration. The anhydrous form is a pale green or gray-white powder, whereas the pentahydrate, the most commonly encountered salt, is bright blue. 1 Coordination Chemistry The Nobel Prize in Chemistry 1913 Alfred Werner (University of Zurich, Switzerland) "in recognition of his work on the linkage of atoms in molecules by which he has thrown new light on earlier investigations and opened up new fields of research especially in inorganic chemistry“ Prior to Werner's work, it was not known how the atoms in a molecule of the chemical formula [Pt(NH3)2Cl2] were connected. The theories at the time predicted such molecules to be connected as linear chains: [Pt-NH3-NH3-Cl]Cl or Cl-NH3-Pt-NH3-Cl Compound Color Historic name CoCl3 • 6 NH3 Yellow Luteo-complex CoCl3 • 5 NH3 purple Purpureo-complex CoCl3 • 4 NH3 Green Praseo-complex CoCl3 • 4 NH3 Violet Violeo-complex Werner studied the conductivities of solutions. Other factors being equal, the conductivity of an aqueous solution of an ionic salt is proportional to the number of ions produced when the salt is dissolved. Werner studied the following series: Pt(NH3)6Cl4 has relatively high conductivity (more ions) Pt(NH3)5Cl4 has decreasing conductivity Pt(NH3)4Cl4 has decreasing conductivity Pt(NH3)3Cl4 has decreasing conductivity Pt(NH3)2Cl4 has zero conductivity KPt(NH3)Cl5 has increasing conductivity K2PtCl6 has increasing conductivity These experiments led Werner to postulate the presence of 2 different types of bonding in inorganic coordination compounds: 1. Primary valences, fixed in number by charge balance; these were ionic (i.e. to ions only) and non-directional. 2. Secondary valences, determined by the nature of the central metal atom (usually 4 or 6); non-ionic, directional. Based on the postulations, Werner formulated the compounds [Pt(NH3)6]Cl4 [Pt(NH3)5Cl]Cl3 [Pt(NH3)4Cl2]Cl2 [Pt(NH3)3Cl3]Cl [Pt(NH3)2Cl4] K[Pt(NH3)Cl5] K2[PtCl6] has relatively high conductivity (more ions) has decreasing conductivity has decreasing conductivity has decreasing conductivity has zero conductivity has increasing conductivity has increasing conductivity (4-n)+ [Pt(NH3)6-nCln] 1- (Cl )4-n Coordination chemistry Transition metals can form a variety of coordination compounds Complex: A central metal ion (atom) bonded via coordinate bonds to one or more molecules or ions called ligands Complex ion – complex with net charge coordination number – number of coordinate bonds between metal ion and the ligand or ligands Coordination compound: (1) neutral substance made up of complex ion and another ion of opposite charge. (2) neutral complex Ligand: contains at least one nonbonding pair of electrons Some important aspects of transition metal ions (1) The valence electrons are in d orbitals (2) The d orbitals do not have large radial extension. They are therefore mostly nonbonding in complexes of transition metal ions 2 Valence bond description a ligand-to-metal dative bond metal center M metal empty hybridized orbital L ligand ligand orbital accomodating a lone pair of electrons Ligands Bidentate ligands (chelate ligands) Types of Ligands • Depending on the number of electron lone pairs available, ligands will bond to a metal through 1 (monodentate), 2 (bidentate), 3 (tridentate), ... atoms • Any polydentate ligand is also called a chelating ligand • Cl-, NH3, H2O are all monodentate ligands • Ethylenediamine (en) is a common bidentate ligand 3 Polydentate ligands ethylenediaminetetraacetic acid H4EDTA In 1999, the annual consumption of EDTA was equivalent to about 35,000 tons in Europe and 50,000 tons in the US. The most important uses are: Industrial cleaning: complexation of Ca2+ and Mg2+ ions, binding of heavy metals. Detergents: complexation of Ca2+ and Mg2+ (reduction of water hardness). Photography: use of Fe(III)EDTA as oxidizing agent. Pulp and paper industry: complexation of heavy metals during chlorine-free bleaching, stabilization of hydrogen peroxide. Textile industry: complexation of heavy metals, bleach stabilizer. EDTA is used in chelation therapy for acute hypercalcemia (急性的高血鈣症), mercury poisoning and lead poisoning Classify the following ligands as monodentate, bidentate, or tridentate. 4 The Concept of Coordination Numbers The concept of coordination numbers was an important result of Werner's research. It clearly established the two most important aspects that are required to discuss chemical structure and change: oxidation state and coordination number. The concept of oxidation state implies that we have knowledge who loses electrons and who gains electrons in a compound. This cannot be done unambiguously unless generally accepted rules are applied. Coordination number -- 1 Complexes with coordination number 1 were erroneously claimed for copper and silver, but a genuine metal complex with CN= 1 (M = Indium) was recently reported by P. Power (S. T. Haubrich, P. Power, JACS 1998, 120, 2202-2203) i Pr i Pr i Pr In i Pr i Pr i Pr The concept of coordination number is a geometric concept and simply represents the number of next neighbors. It is important to realize that the concept of coordination numbers does not imply any specific type of bonding. Dative bonds, hydrogen bridge bonding, single, double or triple bonds indeed any type of bond (including those found in solids) is permissible. It is this generality of the concept that makes the concept so valuable. Stable compounds with coordination number 2 are restricted to the copper group (Cu, Ag, Au) and to mercury. Typical coordination numbers 5 Isomerism Isomers (same formula, different properties) Structural isomers (different bonds) Coordinationsphere isomers Linkage isomers Stereoisomers (same bonds, different arrangements) Geometric isomers Cis and trans isomers Structural isomers Optical isomers Mirror images Linkage isomers Linkage isomers are two or more coordination compounds in which the donor atom of at least one of the ligands is different (i.e., the connectivity between atoms is different) Two types of structural isomers are linkage isomerism and coordination-sphere isomers. Coordination sphere isomers: ratio of ligand:metal same, but ligands are attached to metal ions in different numbers – – – – [Cr(H2O)6]Cl3, violet [CrCl(H2O)5]Cl2.H2O, blue-green [CrCl2(H2O)4]Cl.2H2O, dark green [CrCl3(H2O)3].3H2O, yellow green 3+ OH2 H2 O Cr [CoIII(NH3)5Br]SO4 [CoIII(NH3)5SO4] Br 2+ Cl OH2 H2O OH2 H2 O violet - 3Cl H2 O Cr H2 O OH2 OH2 H2 O green 2Cl- Similarly, SCNcan coordinate through S or N. + Cl H2 O Cr H2 O OH2 OH2 Cl- trans-[RuCl(py)4(NO)]+ Cl Phys. Chem. Chem. Phys., 2007, 9, 3717 - 3724 green 6 Stereoisomers Geometric isomers Stereoisomers are isomeric molecules whose atomic connectivity is the same but whose atomic arrangement in space is different. Geometric isomers Geometric isomers are stereoisomers where the two forms are not mirror images of each other. Square planar complexes Pt(NH3)2Cl2 exists as cis and trans isomers Octahedral complexes fac (facial) mer (meridional) cis trans Tetrahedral complexes have all positions equivalent and adjacent to all other positions, so there are no geometric isomers 7 Optical isomers Non-identical mirror images with the same geometric arrangement of ligands. A set of two (mirror image) isomers are called enantiomers. Enantiomers differ only in optical properties – rotate plane-polarized light in opposite directions. Optical isomers are mirror images of one another that cannot be superimposed. Neither tetrahedral M(A-A)2 nor square planar M(A-A)2 has isomers Tetrahedral M(A-B)2 has optical isomers but not geometric isomers; square planar M(A-B)2 has geometric isomers but not optical isomers. 8 Crystal field theory (Ligand field theory) – Color and Magnetism Most transition metal complexes are colorful Color of a complex depends on the metal, its oxidation state, and its ligands Examples: [Cu(H2O)6]2+ pale blue; [Cu(NH3)6]2+ dark blue; d0 metal ions are usually colorless. 9 Crystal field theory was developed to explain the colors and magnetism of coordination compounds Major assumption: electrons on the metal ( d electrons) are repelled by the ligand electrons 10 dx2-y2, dz2 dxy, dxz, dyz dxy, dxz, dyz dx2-y2, dz2 11 [Ti(H2O)6]3+ The spectrochemical series Concept of high-spin and low-spin 12