as a PDF
... or monograph. The organometallic chemistry is confined to cr-bonded compounds in normal oxidation states; compounds with 7r-bonding ligands are generally excluded. Their inclusion would have increased the length of the book considerably and, moreover, their recent chemistry has been extensively and ...
... or monograph. The organometallic chemistry is confined to cr-bonded compounds in normal oxidation states; compounds with 7r-bonding ligands are generally excluded. Their inclusion would have increased the length of the book considerably and, moreover, their recent chemistry has been extensively and ...
Copyright 2010 Scott R
... Na(H3BNMe2BH3), in tetrahydrofuran produces the new complex Th(H3BNMe2BH3)4. The thorium center forms bonds with fifteen hydrogen atoms; accordingly, this is the first example of a fifteen-coordinate atom of any kind. As determined by both single crystal X-ray and single crystal neutron diffraction ...
... Na(H3BNMe2BH3), in tetrahydrofuran produces the new complex Th(H3BNMe2BH3)4. The thorium center forms bonds with fifteen hydrogen atoms; accordingly, this is the first example of a fifteen-coordinate atom of any kind. As determined by both single crystal X-ray and single crystal neutron diffraction ...
CHM 423 Coordination Chemistry
... donors. Thus a Lewis acid must have empty suitable orbitals to accommodate the donated electron pairs. The presence of empty suitable orbitals in transition metals (Cu, Co, Fe etc) and some compounds (BF3, BeCl2 with empty p-orbital) and ions (H+) of main block elements makes them to act as Lewis ac ...
... donors. Thus a Lewis acid must have empty suitable orbitals to accommodate the donated electron pairs. The presence of empty suitable orbitals in transition metals (Cu, Co, Fe etc) and some compounds (BF3, BeCl2 with empty p-orbital) and ions (H+) of main block elements makes them to act as Lewis ac ...
13- and 14-membered macrocyclic ligands containing
... same experimental conditions, and the corresponding values are also collected in Tables 1 and 2. The literature values for dota and dotp with the same metal ions are also listed for comparison reasons, however recommended values for these two ligands do not exist [36]. Indeed, the very high values o ...
... same experimental conditions, and the corresponding values are also collected in Tables 1 and 2. The literature values for dota and dotp with the same metal ions are also listed for comparison reasons, however recommended values for these two ligands do not exist [36]. Indeed, the very high values o ...
c00kieee - Ritter Illustration
... f-block elements, actinides (5f ) and lanthanides (4f ) are separated from the other elements. This modern placement as well as their name is attributed to Prof. Glenn T. Seaborg, who in the 1930s proposed the actinide theory. As a result of this concept, the actinides were removed from their origin ...
... f-block elements, actinides (5f ) and lanthanides (4f ) are separated from the other elements. This modern placement as well as their name is attributed to Prof. Glenn T. Seaborg, who in the 1930s proposed the actinide theory. As a result of this concept, the actinides were removed from their origin ...
The d-Block Elements
... next element—Ce—is 6s25d04f2. From this point through element 71, added electrons enter the 4f subshell, giving rise to the 14 elements known as the lanthanides. After the 4f subshell is filled, the 5d subshell is populated, producing the third row of the transition metals. Next comes the seventh pe ...
... next element—Ce—is 6s25d04f2. From this point through element 71, added electrons enter the 4f subshell, giving rise to the 14 elements known as the lanthanides. After the 4f subshell is filled, the 5d subshell is populated, producing the third row of the transition metals. Next comes the seventh pe ...
Amidine: Structure, Reactivity and Complexation Behaviour
... parent substituted molecules and hence there is a decrease in the N-H bond dissociation enthalpy when compared with the computed values for benzamidine. Electron withdrawing group stabilizes the neutral substituted molecule and therefore, the N-H BDE increases with respect to benzamidine solvent eff ...
... parent substituted molecules and hence there is a decrease in the N-H bond dissociation enthalpy when compared with the computed values for benzamidine. Electron withdrawing group stabilizes the neutral substituted molecule and therefore, the N-H BDE increases with respect to benzamidine solvent eff ...
Efficient Phosphodiester Hydrolysis by
... distamycin with plasmid DNA suggested that 1 and 2 are regio-specific, interacting with the minor groove of DNA. These complexes displayed luminescent properties. Complex 1 showed higher emission intensity than 2 due to a more efficient energy transfer between triplet and emission levels of terbium ...
... distamycin with plasmid DNA suggested that 1 and 2 are regio-specific, interacting with the minor groove of DNA. These complexes displayed luminescent properties. Complex 1 showed higher emission intensity than 2 due to a more efficient energy transfer between triplet and emission levels of terbium ...
Inorganometallic Chemistry
... TM complexes containing borylene ligands (BR) can adopt two different coordination modes: either terminal (BR group terminally bonded to one metal atom and boron assuming a coordination number 2) or bridging (borylene group adopting bridging position between two metal centers with a threefold-coordi ...
... TM complexes containing borylene ligands (BR) can adopt two different coordination modes: either terminal (BR group terminally bonded to one metal atom and boron assuming a coordination number 2) or bridging (borylene group adopting bridging position between two metal centers with a threefold-coordi ...
The d- and f- Block Element Block Elements The d- and f
... The d orbitals of the transition elements project to the periphery of an atom more than the other orbitals (i.e., s and p), hence, they are more influenced by the surroundings as well as affecting the atoms or molecules n surrounding them. In some respects, ions of a given d configuration (n = 1 – 9 ...
... The d orbitals of the transition elements project to the periphery of an atom more than the other orbitals (i.e., s and p), hence, they are more influenced by the surroundings as well as affecting the atoms or molecules n surrounding them. In some respects, ions of a given d configuration (n = 1 – 9 ...
Transition metals and coordination chemistry
... metals. Detergents: complexation of Ca2+ and Mg2+ (reduction of water hardness). Photography: use of Fe(III)EDTA as oxidizing agent. Pulp and paper industry: complexation of heavy metals during chlorine-free bleaching, stabilization of hydrogen peroxide. Textile industry: complexation of heavy metal ...
... metals. Detergents: complexation of Ca2+ and Mg2+ (reduction of water hardness). Photography: use of Fe(III)EDTA as oxidizing agent. Pulp and paper industry: complexation of heavy metals during chlorine-free bleaching, stabilization of hydrogen peroxide. Textile industry: complexation of heavy metal ...
template - Communications in Inorganic Synthesis
... oxygen donors, four from two carboxylate groups and the fifth from an ether group. It may bind to metal ions in mono-, bi-, tri- and multidentate coordination modes. A number of oda complexes with lanthanide ions have interesting physical and chemical properties as well as fascinating structures [1] ...
... oxygen donors, four from two carboxylate groups and the fifth from an ether group. It may bind to metal ions in mono-, bi-, tri- and multidentate coordination modes. A number of oda complexes with lanthanide ions have interesting physical and chemical properties as well as fascinating structures [1] ...
Document
... series, the atomic radii actually ____________again. At the beginning of the series, the increase in __________ _______________ with atomic number pulls in the electron cloud, resulting in a reduction of atomic size. Since electrons are added to an inner d subshell across the series, this adds to th ...
... series, the atomic radii actually ____________again. At the beginning of the series, the increase in __________ _______________ with atomic number pulls in the electron cloud, resulting in a reduction of atomic size. Since electrons are added to an inner d subshell across the series, this adds to th ...
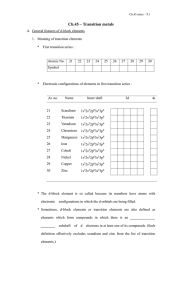
DCY1B - Manonmaniam Sundaranar University
... the decrease in atomic radii is much less compared to P-block elements. This is because, addition of electrons to the inner (n-1) d orbital effectively screen the outer ns electron from the added nuclear charge (primary screening effect). Further, the decrease in the atomic radii is minimum for grou ...
... the decrease in atomic radii is much less compared to P-block elements. This is because, addition of electrons to the inner (n-1) d orbital effectively screen the outer ns electron from the added nuclear charge (primary screening effect). Further, the decrease in the atomic radii is minimum for grou ...
KUT 203/2 - Chemistry Practical III (Inorganic Chemistry)
... • Determine the composition of isomers thus isolated through titration. • Substantiate the optical behaviour of these isomers. • Understand the concept on lingkage isomerism in coordination compounds and the technique used in the synthesis and isolation of these complex ions i.e. [(NH3)5CoNO2]Cl2 an ...
... • Determine the composition of isomers thus isolated through titration. • Substantiate the optical behaviour of these isomers. • Understand the concept on lingkage isomerism in coordination compounds and the technique used in the synthesis and isolation of these complex ions i.e. [(NH3)5CoNO2]Cl2 an ...
Elements – (Metals)
... Malleable – pounded into plate 4) Form positive ions Metals readily lose one or more electrons since electron configuration is such that many inner electrons shield full charge of nucleus and so outer electrons are not as strongly held. For example K forms K+ but Cl is apt to gain electron to become ...
... Malleable – pounded into plate 4) Form positive ions Metals readily lose one or more electrons since electron configuration is such that many inner electrons shield full charge of nucleus and so outer electrons are not as strongly held. For example K forms K+ but Cl is apt to gain electron to become ...
Scandium and Yttrium - Mercyhurst University
... in a Ytterby quarry.2 The rock looked like coal but was quite dense and so was considered to contain tungsten (the densest element known at that time).1 Upon careful chemical analysis by many chemists (see Table) this mineral yielded the ‘earth’ of all of the lanthanides as well as scandium and ytt ...
... in a Ytterby quarry.2 The rock looked like coal but was quite dense and so was considered to contain tungsten (the densest element known at that time).1 Upon careful chemical analysis by many chemists (see Table) this mineral yielded the ‘earth’ of all of the lanthanides as well as scandium and ytt ...
Biologically Important Inorganic Elements Occurrence and Availability
... • Despite the high abundance of Si, Al and Ti (the 2nd, 3rd and 10th most abundant elements on earth). Why are they are not utilized biologically? • Because of the insolubility of their naturally occurring oxides (SiO2, Al2O3, TiO2) under physiological conditions. A lower oxidation state is unavaila ...
... • Despite the high abundance of Si, Al and Ti (the 2nd, 3rd and 10th most abundant elements on earth). Why are they are not utilized biologically? • Because of the insolubility of their naturally occurring oxides (SiO2, Al2O3, TiO2) under physiological conditions. A lower oxidation state is unavaila ...
Preview Sample 1
... 35. The systematic (IUPAC) name of NaClO4 is: A. sodium perchlorate B. sodium chlorate C. sodium hypochlorate D. sodium chloride tetraoxide ...
... 35. The systematic (IUPAC) name of NaClO4 is: A. sodium perchlorate B. sodium chlorate C. sodium hypochlorate D. sodium chloride tetraoxide ...
Boron Group Compounds Oxidation States Boron
... Indium is more commonly found in the +1 oxidation state, while thallium is only found in this state (e.g. TlBr) This behavior is also seen in other p-block groupings, and is explained by the inert pair effect (results from the ionization energies of the 2nd and 3rd electrons in period 4 and heavier ...
... Indium is more commonly found in the +1 oxidation state, while thallium is only found in this state (e.g. TlBr) This behavior is also seen in other p-block groupings, and is explained by the inert pair effect (results from the ionization energies of the 2nd and 3rd electrons in period 4 and heavier ...
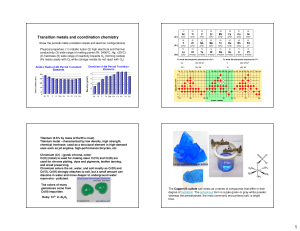
TRANSITION ELEMENTS
... Transition elements refer to those in the d-block of the Periodic Table. The sequence from scandium (Z = 21) to zinc (Z = 30) form what is generally regarded as the first transition series. Virtually ALL the properties of transition elements are related to their electronic structures, in particular ...
... Transition elements refer to those in the d-block of the Periodic Table. The sequence from scandium (Z = 21) to zinc (Z = 30) form what is generally regarded as the first transition series. Virtually ALL the properties of transition elements are related to their electronic structures, in particular ...
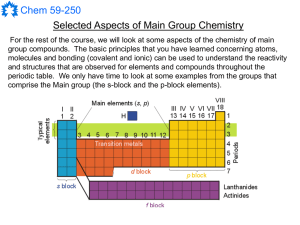
The d-block elements are commonly known as transition
... Transition metal compounds are paramagnetic when they have one or more unpaired d electrons. Some compounds are diamagnetic. These include octahedral, lowspin, d6 and square-planar d8complexes. In these cases, crystal field splitting is such that all the electrons are paired up. Ferromagnetism occu ...
... Transition metal compounds are paramagnetic when they have one or more unpaired d electrons. Some compounds are diamagnetic. These include octahedral, lowspin, d6 and square-planar d8complexes. In these cases, crystal field splitting is such that all the electrons are paired up. Ferromagnetism occu ...
as a PDF
... chemistry of transition series. Irregularities due to inter-electronic repulsion are most obvious in the lanthanide series where ligand field effects are very small. For the first century of lanthanide chemistry, talk of irregularities would have seemed ridiculous. The laborious discovery and separa ...
... chemistry of transition series. Irregularities due to inter-electronic repulsion are most obvious in the lanthanide series where ligand field effects are very small. For the first century of lanthanide chemistry, talk of irregularities would have seemed ridiculous. The laborious discovery and separa ...
Lanthanide

The lanthanide /ˈlænθənaɪd/ or lanthanoid /ˈlænθənɔɪd/ series of chemical elements comprises the fifteen metallic chemical elements with atomic numbers 57 through 71, from lanthanum through lutetium. These fifteen lanthanide elements, along with the chemically similar elements scandium and yttrium, are often collectively known as the rare earth elements.The informal chemical symbol Ln is used in general discussions of lanthanide chemistry to refer to any lanthanide. All but one of the lanthanides are f-block elements, corresponding to the filling of the 4f electron shell; lutetium, a d-block element, is also generally considered to be a lanthanide due to its chemical similarities with the other fourteen. All lanthanide elements form trivalent cations, Ln3+, whose chemistry is largely determined by the ionic radius, which decreases steadily from lanthanum to lutetium.They are termed as lanthanides because the lighter elements in the series are chemically similar to lanthanum. Strictly speaking, both lanthanum and lutetium have been labeled as group 3 elements, because they both have a single valence electron in the d shell. However, both elements are often included in any general discussion of the chemistry of the lanthanide elements.In presentations of the periodic table, the lanthanides and the actinides are customarily shown as two additional rows below the main body of the table, with placeholders or else a selected single element of each series (either lanthanum and actinium, or lutetium and lawrencium) shown in a single cell of the main table, between barium and hafnium, and radium and rutherfordium, respectively. This convention is entirely a matter of aesthetics and formatting practicality; a rarely used wide-formatted periodic table inserts the lanthanide and actinide series in their proper places, as parts of the table's sixth and seventh rows (periods).