* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download all work must be shown to receive full credit
Survey
Document related concepts
Transcript
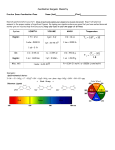
Dr. Woodward Chemistry 123 Sample Quiz 1 Spring 2007 Quiz 5 20 Minutes Name _____________________________________ Lab Inst. ___________ Two-Digit Section Number ___________ ALL WORK MUST BE SHOWN TO RECEIVE FULL CREDIT 1. The absorption maximum for the complex ion [Co(NH3)6]3+ occurs at 470 nm. (a) [2 points] Predict the color of this complex. Orange (b) [2 points] If the cobalt ion adopts a low spin configuration draw the crystal-field energy-level diagram and show the placement of the delectrons? dx2-y2 dxy dxz dz2 dyz (c) [4 points] Calculate the crystal field splitting energy, Δ, in eV. E = hc/λ = (6.626 × 10–34 J-s)(3.00 × 108 m/s)/(470 × 10–9 m) = 4.23 × 10–19 J E = (4.23 × 10–19 J) × (1 eV/1.602 × 10–19 J) = 2.64 eV (d) [2 points] Would you expect the absorption maximum of [Co(H2O)6]3+ to fall at a longer or at a shorter wavelength (assume low spin configuration is maintained)? According to the spectrochemical series H2O is a weaker field ligand that NH3. Therefore, the crystal field splitting Δ will be smaller and the absorption maximum will be shifted to longer wavelengths. 2. [6 points] The following pictures show two molecular orbitals of the many that exist for a MoF6 molecule, which has octahedral geometry. For each MO, identify the atomic orbitals involved in forming the molecular orbital (Mo 5s, 5p, 4d; F 2s, 2p), and classify the molecular orbital as bonding, antibonding or non-bonding. [The Mo atom is at the center of the molecule, it is surrounded by six fluorine atoms] Error! Mo 4d – O 2p antibonding O 2p nonbonding Q5-R 11:30 1 3. Draw all stereoisomers (geometric and/or optical) for the following complexes: (a) [4 points] octahedral [Co(NH)3Cl3]+ Two geometric isomers (fac- and mer-) as shown in problem 24.4 in the homework exercises (b) [4 points] square planar [PdBr2Cl2]2– Two geometric isomers, cis- and trans- 4. [3 points] The figure below shows the absorption spectra for a transition metal complex [M(NH3)6]2+ (where M is a transition metal ion). What would be the color of this complex? How would the absorption peak at ~610 nm shift if the NH3 ligands were replaced with H2O? 2.0 1.8 1.6 absorbance 1.4 1.2 1.0 0.8 0.6 0.4 0.2 0.0 200 300 400 500 600 700 800 900 wavelength (nm) This compound absorbs strongly yellow, orange and red light. Therefore it will appear to the middle color of the remaining colors (violet, blue, green), so it’s color will be blue [this is the [Cu(NH3)6]2+ discussed in the lab manual] If the NH3 ligands are replaced with H2O the crystal field splitting will get smaller and the absorption maximum will shift to longer wavelengths [see spectrum in the lab manual] Q5-R 11:30 2