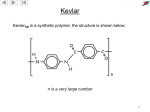
* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download CHAPTER TWENTY-TWO: CARBOXYLIC ACIDS
Proteolysis wikipedia , lookup
Catalytic triad wikipedia , lookup
Metalloprotein wikipedia , lookup
Genetic code wikipedia , lookup
Peptide synthesis wikipedia , lookup
Citric acid cycle wikipedia , lookup
15-Hydroxyeicosatetraenoic acid wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Nucleic acid analogue wikipedia , lookup
Specialized pro-resolving mediators wikipedia , lookup
Butyric acid wikipedia , lookup
Biosynthesis wikipedia , lookup
Fatty acid synthesis wikipedia , lookup
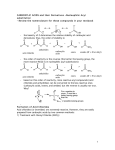
OUTLINE CHAPTER 21 KLEIN: CARBOXYLIC ACIDS AND THEIR DERIVATIVES 1. Structure and Properties of Carboxylic Acids a. Bp/mp/solubility b. Acidity Rel acidity of substituted acids (ARIO stabilization of conjugate base) Comparison to Phenols and other weak acids Amino Acids and Zwitterions 2. Preparation of Carboxylic Acids a. Via oxidation of primary alcohols and aldehydes b. Via oxidation of benzylic CH with KMnO4 or H2CrO4 c. Via Grignard with CO2 3. Reactivity of Carboxylic Acids a. Rxn w SOCl2 to form acid chlorides b. Fischer Esterification i. Reversible ii. Equilibrium Constant Keq iii. Hydrolysis of Ester under basic conditions for quantitative conversion c. Reaction with amines and DCC to form amides d. Reduction i. To alcohols with LiAlH4 ii. To aldehydes via acid chlorides with LiAlH(Ot-bu)3 iii. Selective Reduction: o NaBH4 doesn’t reduce RCOOH o RCOOH less reactive than ketones/aldehydes/alkenes with H2/Pt e. Rxn with alkyl lithium R’Li to form dianion then hydrolysis to Ketone RCOR’ 4. Relative Reactivity and Structure of Carboxylic Acid Derivatives a. Acid chlorides>Anhydrides>Esters, Acids>Amides, nitriles>carboxylates b. Leaving group ability and base strength (weak bases are better LG) 5. Interconversion of Derivatives via addition-elimination Acid chloridesAnhydridesEsters, AcidsAmidescarboxylates 6. Acid Chloride Reactivity i. With carboxylates, water, alcohols, and amines (Addn-Elim) ii. With LiAlH4 to form alcohols iii. With LiAlH(tBuO)3 to form aldehydes iv. With Grignards to form 3° alcohols (mixtures/ poor yields common) v. With Gilman (organocuprate) reagents (note: this reaction occurs in high yield only with acid chlorides) vi. With Benzene and AlCl3 in Friedel-Crafts Acylation 7. Acid Anhydrides a. Preparation from acid chlorides with carboxylates b. Reactivity With water, alcohols, and amines (addn-Elim) 8. Esters a. Preparation (review) i. From acid chlorides or acid anhydrides with alcohols ii. From carboxylic acids and acid catalyst: Fischer Esterification iii. Lactones: Cyclic Ethers via intramolecular Acyl substitution iv. From acids with diazomethane v. Transesterification: One ester to another b. Reactivity i. with amines; with water in acid or base; with alcohols in acid or base (Addn-Elim) ii. with LAH to form 3° alcohols (2° alcohols from formate esters) iii. with DIBAL-H (Al(CH2CH(CH3)2H) to form aldehydes at low T iv. With Grignards and organolithium reagents to form alcohols (double addn) 9. Amides a. Preparation i. From carboxylic acids with amines using DCC (review) ii. From acid chlorides, acid anhydrides or esters with amines (AddnElim) b. Reactivity i. Hydrolysis with water and acid or base ii. Reduction with LAH to amines iii. Reduction with DIBAL-H to aldehydes 10. Nitriles a. Preparation via SN2 displacement on alkyl halide b. Reactivity i. With water in acid or base to amide and then carboxylic acid ii. With LAH to 1° amine iii. With DIBAL-H to aldehyde c. With Grignard to form ketone (after hydrolysis of imine product) 11. Special Spectroscopic Features a. IR patterns: anhydrides, carboxylic acids b. 13C NMR chemical shifts: the C=O signal c. The 1H NMR spectra of amides: hindered rotation and slow exchange OH– H3O+ NH3 RCOCl RCOOCOR’ RCOOR’ RCOO– RCOO– RCOO– RCOOH RCOOH RCOOH RCONH2 RCONH2 RCONH2 RCOOH RCOO– ------- Poor yield RCONHR’ RCOO– RCOOH No RCN RCOO– RCOOH no SUMMARY R’OH R’Li or R’MgBr then H+ RCOOR’ RCR’2OH RCOOR’ RCR’2OH RCOOR’ RCR’2OH (H+ or OH– cat) RCOOR’ R’H + (H+ or RCOO– – OH cat) no R’H + RCNR’– no RCR’ (via hydrolysis of RR’C=N) R’2CuLi LAH DIBAL-H RCOR’ no no RCH2OH RCH2OH RCH2OH Not useful Not useful RCHO R’H RCH2OH RCOO– R’H RCH2NH2 no RCH2NH2 RCHO and RCH2NH2 RCHO LEARNING OUTCOMES: Understand the structural features that affect relative reactivity in acyl transfer reactions (interconversion of carboxylic acid derivatives via addn- elimination reactions) Predict the missing product and/or reactant in a relevant chemical reaction involving carboxylic acids and carboxylic acid derivatives. Use curved-arrow formalism to depict the known mechanism of an organic reaction. Propose a mechanism for an unfamiliar chemical transformation by analogy to known reaction pathways. Understand the structural features that lead to product selectivity in organic reactions. Develop strategies to avoid undesirable side reactions in a multi-step synthesis. Devise multi-step syntheses of molecular targets using comprehensive knowledge of functional group transformations. SAMPLE EXAM PROBLEMS: 1. Propose a sequence of steps to effect the following molecular transformation: 2. Rank the acidity of the following from 1= most acidic to 5= least acidic 3. Use curved-arrow formalism to depict the mechanism of the following chemical transformation. Show every step in sequence including proton transfer steps and include all unbonded electrons and formal charges in your drawings. 4. a) Explain why the following reaction does not take place in high yield. Include in your explanation any alternative products formed. b) Propose an alternative synthesis of the product beginning with the same starting material.