* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download fluid_pr
Drag (physics) wikipedia , lookup
Sir George Stokes, 1st Baronet wikipedia , lookup
Lattice Boltzmann methods wikipedia , lookup
Boundary layer wikipedia , lookup
Coandă effect wikipedia , lookup
Compressible flow wikipedia , lookup
Hemorheology wikipedia , lookup
Accretion disk wikipedia , lookup
Hydraulic machinery wikipedia , lookup
Stokes wave wikipedia , lookup
Magnetohydrodynamics wikipedia , lookup
Flow measurement wikipedia , lookup
Flow conditioning wikipedia , lookup
Airy wave theory wikipedia , lookup
Computational fluid dynamics wikipedia , lookup
Navier–Stokes equations wikipedia , lookup
Bernoulli's principle wikipedia , lookup
Aerodynamics wikipedia , lookup
Fluid thread breakup wikipedia , lookup
Derivation of the Navier–Stokes equations wikipedia , lookup
Reynolds number wikipedia , lookup
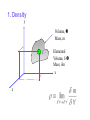
1. Density y Volume, Mass, m C z Elemental Volume, Mass, m x 2. Fluid Shear M y M’ Fluid Element at time, t P y P’ Fluid Element at time, t+t x N y O Fx d Fx du yx lim A 0 A d Ay d y y y Force, Fx Velocity u 3. Viscosity In some sense measures fluidity of a fluid. Actually it is the resistance offered by a layer of fluid to the motion of an adjacent one. Consider the two-plate experiment. In case of a fluid in between them, we know that the upper plate moves with a speed U whereas the lower plate does not move. This sets up a velocity gradient in a direction normal to flow. 4. Newtonian Fluids du yx dy • For a Newtonian fluid In general ie., yx du dy u y is called •absolute or dynamic viscosity. •Its dimensions are ML-1 T-1 •Air and water are common examples Kinematic viscosity (n) is defined as ( /r) . Its dimensions are M L-3 5. Non-Newtonian Fluids • Shear stress not proportional to deformation rate •Toothpaste, paint are common examples du k dy Deformation rate du dy n 6. Temperature Effect It is observed that viscosity of a liquid decreases with temperature where as that of a gas increases with temperature. Find out why. Values of viscosity and kinematic viscosity n for various fluids are tabulated in handbooks and textbooks. For air viscosity may be calculated using 1.5 T 1.458 106 T 110.4 the Sutherland formula, where T is in Kelvin and is in kg/s m. 7. Velocity Field •Velocity at a point may be defined as the instantaneous velocity of a fluid particle passing through that point. V V(x, y, z, t) or V uiˆ vˆj wkˆ For a steady flow the properties do not change with time - V 0 or V V(x, y, z) t If S is any property, S 0 or S S(x, y, z) t 8. 1, 2, 3 Dimensional Flows One, Two, Three Dimensional Flows -- One, Two, Three Space Coordinates required to specify Velocity Field r u R One Dimensional flow. u = u(r) x umax y x u=u(x,y) u=u(x,y) Two Dimensional flow. u = u(x,y) 9. Surface Tension It is the apparent interfacial stress that acts when a liquid has a density interface like liquid-gas, liquid-solid, liquid-liquid 2R 2R q<90 0 q Dh q>90 0 2σ cosθ Δh λR Dh q 10. Surface Shapes water water soap Wetting wax Non-wetting 11. Forces on half a fluid drop pπ R 2 s 2R 2 p R 12. Continuum Flow For most engineering applications we consider fluid to be continuous. But we do know that matter consists of molecules. To be considered continuous a fluid must have a large number of molecules in a tiny place which is small compared to the body dimensions. Under ordinary conditions this is true. For eg., A cubic metre of Air at STP contains 2.5 x 10 25 molecules. Its mean free path is like 6.6 x 10-8 m. 13. Rarefied Flows At great heights from the sea level it is not possible to consider air to be continuous. The molecular mean free path may be of the same order of magnitude as the body dimensions. Eg., at an altitude of 130 km the mean free path of air is 10.2m. Then it becomes important to consider individual or groups of molecules. This leads to the discipline of Rarefied Gas Dynamics. 14. Bulk Modulus of Elasticity Compressibility of a fluid may be expressed in terms of Bulk Modulus of Elasticity. dp k dv v For air k is equal to g (adiabatic conditions) and p (isothermal) For water, k =2.2 Gpa, meaning that when a pressure of 0.1Mpa acts upon a cubic metre of water, the change in volume resulting is 1/22000 m 3.