* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Quantum Chemistry II: Lecture Notes
Electromagnetism wikipedia , lookup
Quantum entanglement wikipedia , lookup
Elementary particle wikipedia , lookup
Quantum vacuum thruster wikipedia , lookup
Introduction to gauge theory wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Neutron magnetic moment wikipedia , lookup
EPR paradox wikipedia , lookup
Old quantum theory wikipedia , lookup
Condensed matter physics wikipedia , lookup
Accretion disk wikipedia , lookup
Angular momentum wikipedia , lookup
Bell's theorem wikipedia , lookup
Nuclear physics wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Hydrogen atom wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Angular momentum operator wikipedia , lookup
Geometrical frustration wikipedia , lookup
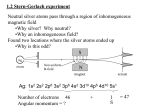
2. Electron Spin Contents Electron spin angular momentum operator, eigenvalues and eigenfunctions; hydrogen atom wavefunctions with electron spin; magnetic moment; magnetic resonance. Central Concepts Spin (operator) Difficult Points Spin eigenfunctions which depend on no external parameters, but can be written in an explicit way. Notes 1. Spin Operators: Eigenvalues The most important property for an angular momentum operator is [Ŝi, Ŝ j]=iεijkħŜ k i,j,k=x,y,z. (1.1) (See eq.(2.6) of Lecture 1) from which almost all other properties can be derived. For the electron, the spin is just a form of angular momentum, but it is intrinsic meaning it cannot be expressed into the form like eqs.(2.1-3) or eqs.(2.15,22) of Lecture 1. However, we can still find out the eigenvalues of the spin angular momentum operators of the electron. For convenience, we denote spin operators with S such as: Ŝ2=Ŝ x2+Ŝ y2+Ŝ z2 (1.2) Eq.(1) is just a compact form of the following equations: [Ŝ x,Ŝ y]=iħŜ z, [Ŝ y, Ŝ z]=iħŜx, [Ŝz, Ŝ x]=iħŜ y, [Ŝ y, Ŝ x]=-iħŜ z, etc. (1.1') Similar to orbital angular momentum operators, we can easily obtain the following equations: [Ŝ2, Ŝ x]=0, [Ŝ2, Ŝ y]=0, [Ŝ2, Ŝ z]=0 Or compactly, [Ŝ2, Ŝ i]=0, i=x,y,z (1.3) Introducing ladder operators, we can find out the eigenvalues of . Ŝ2 and Ŝi (i=x,y,z) just by replacing L with S and repeating the procedure of Section 5.4 of the textbook. The result is: the eigenvalues of Ŝ2 are s(s+1) ħ2, s=0,1/2,1,3/2….(1.4) and the eigenvalues of Ŝz are msħ, ms=-s,-s+1,…,s-1,s. (1.5) Figure: Illustration of the angular momentum vector component Lz. for a tilted loop and its z [If we change Ĺ into Ĵ, we get the same diagram for ANY angular momentum.] Generally, if the z-component of spin angular momentum is measured to be mJħ, the orientation of the total angular momentum relative to z-direction is clearly θ(Ĵ. Ĵz)= cos-1(mJ/[J(J+1)]1/2) (1.6) Therefore, if the z-component of spin angular momentum is measured to be msħ, the orientation of the total angular momentum relative to z-direction is θ(Ŝ, Ŝz)=cos-1(ms/[s(s+1)]1/2) (1.6’) 2. Electron Spin Operators: Eigenvalues and Eigenfunctions For electrons, experiment shows that the value of s is 1/2.[thus the procedure in Section 5.4 can be made much simpler for electrons; see textbook pp. 300-301: Because there are only two possible spin states, we clearly have Ŝ+α=0, Ŝ+β=ħα,, Ŝ-α=ħβ, Ŝ-β=0 (2.1) ⇛ Ŝxα=( Ŝ++Ŝ-)α/2= ħβ/2, Ŝxβ=( Ŝ++Ŝ-)β/2=ħα/2 (2.2) and Ŝyα=( Ŝ+-Ŝ-)α/2i= iħβ/2, Ŝyβ=( Ŝ+-Ŝ-)β/2i=-iħα/2 (2.3) These equations are usually expressed into matrix form as αŜxα=0, βŜxα=ħ/2, αŜxβ=ħ/2, βŜxβ=0, (2.4a) αŜyα=0, βŜyα=iħ/2, αŜyβ=-iħ/2, βŜyβ=0 (2.4b) αŜzα=ħ/2, βŜzα=0, αŜzβ=0, βŜzβ=-ħ/2 (2.4c) Can you write down the matrices Ŝx, Ŝy Ŝz? ] Protons and neutrons also have s=1/2. Photons, the light quantum have spin 1 (but there is no s=0 photons!!! That’s because of relativistic effect for photons travel at the speed of light in the vacuum.) The total angular momentum of an electron is therefore [s(s+1)]1/2 ħ= [1/2(1/2+1)]1/2 ħ=√3/2 ħ (2.5) Applying eq.(1.5) to s=1/2 we have Ŝzα=ħ/2 α, Ŝzβ=-ħ/2 β (2.6) Since Ŝ2 commutes with Ŝz, the eigenfunctions of Ŝz can also be that of Ŝ2 and we have Ŝ2α=3ħ2/4α, Ŝ2β=3ħ2/4β (2.7) Since there are only two possible spin states for electrons and they depend no external variables as orbital angular momentum eigenfunctions. However, we may write down the electron spin eigenfunctions as follows α=α(ms)=δms,1/2 , β=β(ms) δms,-1/2 (2.8) [remember Dirac’s delta-functionδms,1/2=1 if ms=1/2 or 0 otherwise] and the normalization conditions can be readily obtained Σms=-1/2,ms=1/2|α(ms)|2=1, Σms=-1/2,ms=1/2|β(ms)|2=1 [Proof: e.g., Σms=-1/2,ms=1/2|α(ms)|2=|0|2+|1|2=1] and the orthogonality condition is (2.9) Σms=-1/2,ms=1/2α(ms)* β(ms)=0 [proof: Σms=-1/2, ms=1/2 (2.10 α(ms) β(ms)=0*1+1*0=0] * When both the space and spin variables are included, we have the following normalization condition: Σms=-1/2,ms=1/2∫∫∫|ψ(x,y,z,ms)|2dxdydz=1 (2.11 or equivalently Σms=-1/2,ms=1/2∫∫∫|ψ(r,θ,φ,ms)|2 r2sinθdrdθdφ=1 (2.12 [Note sometimes, the summation over ms is omitted and the above equations become ∫∫∫|ψ(x,y,z,ms)|2dxdydz=1 and ∫∫∫|ψ(r,θ,φ,ms)|2 r2sinθdrdθdφ=1,respectively, but one should remember this summation should always be performed whenever the normalization is carried out.] 3. Hydrogen Atom with Electron Spin When the electron spin is considered, to a good approximation, the Hamiltonian of hydrogen atom will be the same if there is no magnetic field applied to it. Therefore the energy eigenequation will be the same except the eigenfunctions are modified so that the spin wavefunctions are taken into account: ψ nlm(r,θφ)→ψ nlm(r,θφ) g(ms) (3.1) where g(ms) is the spin state which is generally a linear combination of α and β: g(ms)=c1α+c2β. There are two independent choices for c1 and c2. Therefore, the degeneracy of hydrogen atom eigenstate is changed into 2n2. 4. Magnetic Resonance Associated with every spin S is a magnetic moment m: m=gsqS/2m (4.1) where m is the mass and q the charge of the spin and gs is g-factor. For electrons, ge≈2, q=-e, we have m=-eS/me►|m|=-e|S|/me =-√3eħ/2me Similarly, for protons, we have m=geeS/2mp=√3eħ/2mp (4.2) (4.3) When a spin is placed in a magnetic field, the energy is given by E=-m.B (4.4) Above equation is the basis of all magnetic resonance spectroscopies such as ESR (electron spin), NMR (nuclear spins), μSR(muon spin) etc. Suppose the magnetic field is along z-axis, we have E=gsqmsħB0/2m=γmsħB0 (4.5) where γ =gsq/2m is called gyromagnetic ratio which can either be positive or negative depending on the sign of gs and q. The resonance frequencies then are found to be given by ν=ΔE=γħB0(ms2-ms1) (4.6) For single electrons and protons, there is only one frequency (γħB0). Because the mass of proton is about 1840 times heavier than that of electron and the g factor of proton is 5.5857/2 times larger than that of electron, the proton NMR requency is 1840*2/5.5857≈700 times lower. Summary The electrons have an intrinsic angular momentum called spin. The spin operator satisfies all the properties imposed on a quantum angular momentum operator and its spin quantum number is 1/2 and it has the total angular momentum of √3ħ/2. The wavefunction of an atom/molecule must take the spin variable into consideration. The spin is always related to a magnetic moment, which is the basis of ESR (electron spin), NMR (nuclear spin), μSR (muon spin) etc. The resonance frequency is determined by the energy difference of the spins at different orientations in spin space(eq.(4.6)) Questions See assignment Problems See assignment References Textbook pp.282-285, pp.299-301. Assignment Back to lecture note index Back to quantum chemistry II home Questions? Back to Ding’s home