homework assignment 2 - the Petersen Home Page
... 1. A 15.40-g sample of a finely-divided mixture of only Fe2S3 and FeS was reacted with excess H2 at elevated temperatures. If the weight percent of Fe2S3 in this mixture is 57.4%, then calculate the total mass in grams of Fe that can be produced. Assume the only other product of these reactions is H ...
... 1. A 15.40-g sample of a finely-divided mixture of only Fe2S3 and FeS was reacted with excess H2 at elevated temperatures. If the weight percent of Fe2S3 in this mixture is 57.4%, then calculate the total mass in grams of Fe that can be produced. Assume the only other product of these reactions is H ...
Molecular Mass of a gas
... Use R = 62.4 L·torr·mol-1·K-1 in your calculations. Use the proper number of significant digits throughout the calculations. Show the mathematical setup in the ...
... Use R = 62.4 L·torr·mol-1·K-1 in your calculations. Use the proper number of significant digits throughout the calculations. Show the mathematical setup in the ...
Chemistry 1- Final Exam Review
... c. H b. F d. I ____ 68. At constant temperature and pressure, gas volume is directly proportional to the a. molar mass of the gas. c. density of the gas at STP. b. number of moles of gas. d. pressure of the gas ____ 69. Calculate the approximate temperature of a 0.50 mol sample of gas at 750 mm Hg a ...
... c. H b. F d. I ____ 68. At constant temperature and pressure, gas volume is directly proportional to the a. molar mass of the gas. c. density of the gas at STP. b. number of moles of gas. d. pressure of the gas ____ 69. Calculate the approximate temperature of a 0.50 mol sample of gas at 750 mm Hg a ...
Unit 3: Properties and States of Matter
... • You and your lab partner must go around to each station and identify whether the property being demonstrated is a physical or chemical property and state why. ...
... • You and your lab partner must go around to each station and identify whether the property being demonstrated is a physical or chemical property and state why. ...
Science 9
... Matter exists in five states or phases: Solid Liquid Gas Plasma Bose-Einstein Condensation Phase describes a PHYSICAL state of matter. Matter “moves” from one phase to another by physical forces such as temperature and pressure. If energy is added (e.g., increased temperature) or taken awa ...
... Matter exists in five states or phases: Solid Liquid Gas Plasma Bose-Einstein Condensation Phase describes a PHYSICAL state of matter. Matter “moves” from one phase to another by physical forces such as temperature and pressure. If energy is added (e.g., increased temperature) or taken awa ...
CHEM 1405 Practice Exam 3 (2015)
... 9) Assuming similar conditions, how many liters of water vapor, H2O, react to produce 1L of hydrogen gas? C(s) + H2O(g) CO(g) + H2(g) A) 1 L ...
... 9) Assuming similar conditions, how many liters of water vapor, H2O, react to produce 1L of hydrogen gas? C(s) + H2O(g) CO(g) + H2(g) A) 1 L ...
Matter Test Review Sheet
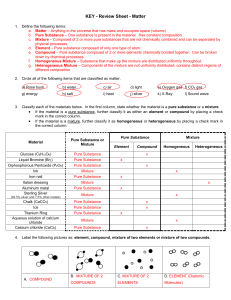
... f. Homogeneous Mixture – Substance that make up the mixture are distributed uniformly throughout. g. Heterogeneous Mixture – Components of the mixture are not uniformly distributed, contains distinct regions of different composition. 2. Circle all of the following items that are classified as matter ...
... f. Homogeneous Mixture – Substance that make up the mixture are distributed uniformly throughout. g. Heterogeneous Mixture – Components of the mixture are not uniformly distributed, contains distinct regions of different composition. 2. Circle all of the following items that are classified as matter ...
1 - gcisd
... 30. Water has several unique properties such as high boiling point, high surface tension and low vapor pressure. What are the main causes for these properties? 31. What happens at the triple point on a phase diagram? 32. Like dissolves like refers to the molecule’s _________________________. 33. Wha ...
... 30. Water has several unique properties such as high boiling point, high surface tension and low vapor pressure. What are the main causes for these properties? 31. What happens at the triple point on a phase diagram? 32. Like dissolves like refers to the molecule’s _________________________. 33. Wha ...
Unit 9 – Behavior of Gases
... 25. If 4.50 g of methane gas (CH4) is introduced to an empty 2.02 L container at 35.3oC, what is the pressure in the container? 26. What is the volume of NO gas at STP that contains 2.2 x 1023 molecules? 27. A breathing mixture used by deep-sea divers contains helium, oxygen and carbon dioxide gase ...
... 25. If 4.50 g of methane gas (CH4) is introduced to an empty 2.02 L container at 35.3oC, what is the pressure in the container? 26. What is the volume of NO gas at STP that contains 2.2 x 1023 molecules? 27. A breathing mixture used by deep-sea divers contains helium, oxygen and carbon dioxide gase ...
final exam practice test - Clayton State University
... a. The amount of heat absorbed by a system at constant volume is E for the process. b. The amount of heat absorbed by a system at constant pressure is H for the process. c. In the relationship E = w + q as applied to a typical chemical reaction, w is usually larger than q. d. At constant temperat ...
... a. The amount of heat absorbed by a system at constant volume is E for the process. b. The amount of heat absorbed by a system at constant pressure is H for the process. c. In the relationship E = w + q as applied to a typical chemical reaction, w is usually larger than q. d. At constant temperat ...
AP Ch 5 Gases . ppt
... 1. A gas is composed of molecules that are separated from each other by distances far greater than their own dimensions. The molecules can be considered to be points; that is, they possess mass but have negligible volume. 2. Gas molecules are in constant motion in random directions. Collisions among ...
... 1. A gas is composed of molecules that are separated from each other by distances far greater than their own dimensions. The molecules can be considered to be points; that is, they possess mass but have negligible volume. 2. Gas molecules are in constant motion in random directions. Collisions among ...
Chemistry Semester 2 Final Exam Chemistry Semester 2 Final Exam
... 1.) A gas has a pressure of 10.56 atm at 25 degrees Celcius. If gas is heated to 40 degrees C, what will the new pressure be? 10.0 atm 2.) A 350 mL air sample collected at 35 C has a pressure of 550. torr. What pressure will the air exert if it is allowed to expand to 425 mL at 57 C? 485 torr 3.) ...
... 1.) A gas has a pressure of 10.56 atm at 25 degrees Celcius. If gas is heated to 40 degrees C, what will the new pressure be? 10.0 atm 2.) A 350 mL air sample collected at 35 C has a pressure of 550. torr. What pressure will the air exert if it is allowed to expand to 425 mL at 57 C? 485 torr 3.) ...
ELAB: One of the Most Potent Amino Acid Analysis
... The accuracy of an analysis is largely determined by its reproducibility. In quantitative chromatographic analysis there must be a reproducible correlation between the amount of substance and the measured result. For the sake of simplicity, this relationship should be linear. Since most detectors do ...
... The accuracy of an analysis is largely determined by its reproducibility. In quantitative chromatographic analysis there must be a reproducible correlation between the amount of substance and the measured result. For the sake of simplicity, this relationship should be linear. Since most detectors do ...
Chemistry I
... 18. The maximum amount of product supposed to be formed from a chemical reaction. a. limiting reactant b. theoretical yield ...
... 18. The maximum amount of product supposed to be formed from a chemical reaction. a. limiting reactant b. theoretical yield ...
File
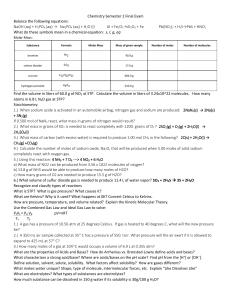
... 58. A 160. mg sample of NaOH, (MM = 40.0 g) is dissolved to prepare an aqueous solution with a volume of 200. mL. What is the molarity of sodium hydroxide in 40. mL of this solution? A) 0.00400 M B) 0.0160 M C) 0.0200 M D) 0.0800 M E) 0.100 M 59. The ionization constant, Kb, of the base HONH2 is 1. ...
... 58. A 160. mg sample of NaOH, (MM = 40.0 g) is dissolved to prepare an aqueous solution with a volume of 200. mL. What is the molarity of sodium hydroxide in 40. mL of this solution? A) 0.00400 M B) 0.0160 M C) 0.0200 M D) 0.0800 M E) 0.100 M 59. The ionization constant, Kb, of the base HONH2 is 1. ...
Chemical Reactions
... The ___________ of which a new substances forms is called the ___________ of __________________. Different chemical substances will produce _______________ at _________________ rates. Factor that affects rate of reaction Temperature ...
... The ___________ of which a new substances forms is called the ___________ of __________________. Different chemical substances will produce _______________ at _________________ rates. Factor that affects rate of reaction Temperature ...
quarter 4 final exam guide - District 196 e
... 40.0 grams of steam at 150. degrees Celcius is cooled, condensed to liquid water, and cooled to 20.0 degrees Celcius. How much energy is given off in this entire process? (Use a diagram “temperature versus time” to visualize what happens as energy is removed from the system – identify the potential ...
... 40.0 grams of steam at 150. degrees Celcius is cooled, condensed to liquid water, and cooled to 20.0 degrees Celcius. How much energy is given off in this entire process? (Use a diagram “temperature versus time” to visualize what happens as energy is removed from the system – identify the potential ...
What are the four states of matter?
... Mass: amount of matter in an object Volume: amount of space an object occupies ...
... Mass: amount of matter in an object Volume: amount of space an object occupies ...
Final Exam review semester 1
... hydrogen in the Haber process. What will be the effect on the equilibrium if the temperature is increased and some of the ammonia is removed from the system? ____ ____ ...
... hydrogen in the Haber process. What will be the effect on the equilibrium if the temperature is increased and some of the ammonia is removed from the system? ____ ____ ...
semester two review sheet
... 2. Calculate the temperature of a gas if 6.5 moles of it occupies a volume of 1.5 L at 2.4 atm 3. There is a mixture of three gases in a closed container. Gas A exerts a pressure of 10 psi, Gas B a pressure of 5.5 psi, and the total pressure is 34.7 psi. What is the partial pressure of Gas C? 4. A g ...
... 2. Calculate the temperature of a gas if 6.5 moles of it occupies a volume of 1.5 L at 2.4 atm 3. There is a mixture of three gases in a closed container. Gas A exerts a pressure of 10 psi, Gas B a pressure of 5.5 psi, and the total pressure is 34.7 psi. What is the partial pressure of Gas C? 4. A g ...
Basic Concepts of the Gas Phase
... Gases appear to have no structure, size or shape. Different gases are always completely miscible and fill all space at their disposal, in contrast to liquids or solids, in which cohesive forces and surface tension restrain the thermal movement of molecules. Under the same conditions of temperature a ...
... Gases appear to have no structure, size or shape. Different gases are always completely miscible and fill all space at their disposal, in contrast to liquids or solids, in which cohesive forces and surface tension restrain the thermal movement of molecules. Under the same conditions of temperature a ...
Type Of Chemical Reaction
... 36. The average kinetic energy of particles of a substance is directly proportional to the ____________ of a substance. 37. Water could be made to boil at a temperature higher than 100 °C by: ...
... 36. The average kinetic energy of particles of a substance is directly proportional to the ____________ of a substance. 37. Water could be made to boil at a temperature higher than 100 °C by: ...
IB 1 CHEMISTRY
... Chemistry has a universal language. The International Union of Pure and Applied Chemistry (IUPAC) is an organization that develops a system of standardized nomenclature for chemical compounds. See the IUPAC gold book for chemical terminology: ...
... Chemistry has a universal language. The International Union of Pure and Applied Chemistry (IUPAC) is an organization that develops a system of standardized nomenclature for chemical compounds. See the IUPAC gold book for chemical terminology: ...
Gas chromatography

Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture (the relative amounts of such components can also be determined). In some situations, GC may help in identifying a compound. In preparative chromatography, GC can be used to prepare pure compounds from a mixture.In gas chromatography, the mobile phase (or ""moving phase"") is a carrier gas, usually an inert gas such as helium or an unreactive gas such as nitrogen. The stationary phase is a microscopic layer of liquid or polymer on an inert solid support, inside a piece of glass or metal tubing called a column (a homage to the fractionating column used in distillation). The instrument used to perform gas chromatography is called a gas chromatograph (or ""aerograph"", ""gas separator"").The gaseous compounds being analyzed interact with the walls of the column, which is coated with a stationary phase. This causes each compound to elute at a different time, known as the retention time of the compound. The comparison of retention times is what gives GC its analytical usefulness.Gas chromatography is in principle similar to column chromatography (as well as other forms of chromatography, such as HPLC, TLC), but has several notable differences. First, the process of separating the compounds in a mixture is carried out between a liquid stationary phase and a gas mobile phase, whereas in column chromatography the stationary phase is a solid and the mobile phase is a liquid. (Hence the full name of the procedure is ""Gas–liquid chromatography"", referring to the mobile and stationary phases, respectively.) Second, the column through which the gas phase passes is located in an oven where the temperature of the gas can be controlled, whereas column chromatography (typically) has no such temperature control. Finally, the concentration of a compound in the gas phase is solely a function of the vapor pressure of the gas.Gas chromatography is also similar to fractional distillation, since both processes separate the components of a mixture primarily based on boiling point (or vapor pressure) differences. However, fractional distillation is typically used to separate components of a mixture on a large scale, whereas GC can be used on a much smaller scale (i.e. microscale).Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). These alternative names, as well as their respective abbreviations, are frequently used in scientific literature. Strictly speaking, GLPC is the most correct terminology, and is thus preferred by many authors.