Chem 430 - Fall 1994
... for the process of compressing 2.00 mole of an ideal monatomic gas from 3.00 x 10-3 m3 at 360.2 K to 1.00 x 103 m3 adiabatically by a constant pressure of 1.00 MPa? ...
... for the process of compressing 2.00 mole of an ideal monatomic gas from 3.00 x 10-3 m3 at 360.2 K to 1.00 x 103 m3 adiabatically by a constant pressure of 1.00 MPa? ...
Chemistry 1: Second Semester Practice Exam Read each question
... C. An increase in temperature D. An increase in pressure 35. AgCl (s) Ù Ag 1+ (aq) + Cl 1- (aq). Consider this reaction at equilibrium. As chloride ions are added to this system and the temperature is kept constant, the value of the equilibrium constant A. Decreases B. Increases C. Remains the same ...
... C. An increase in temperature D. An increase in pressure 35. AgCl (s) Ù Ag 1+ (aq) + Cl 1- (aq). Consider this reaction at equilibrium. As chloride ions are added to this system and the temperature is kept constant, the value of the equilibrium constant A. Decreases B. Increases C. Remains the same ...
Chemistry B1A - Bakersfield College
... Some iron wire weighing 5.6 g is placed in a beaker and covered with 15.1 g of hydrochloric acid. The acid reacts with the metal and gives off hydrogen gas, which escapes into the surrounding air. After reaction the contents of the beaker weighs 20.4 g. What is the mass of hydrogen produced? Write t ...
... Some iron wire weighing 5.6 g is placed in a beaker and covered with 15.1 g of hydrochloric acid. The acid reacts with the metal and gives off hydrogen gas, which escapes into the surrounding air. After reaction the contents of the beaker weighs 20.4 g. What is the mass of hydrogen produced? Write t ...
Chem 152 Chapter 4
... Particles in the gas phase are moving the fastest. Liquid phase particles move with less energy than the gas phase. Solid phase particles are the slowest but they also have movement. (vibrations). ...
... Particles in the gas phase are moving the fastest. Liquid phase particles move with less energy than the gas phase. Solid phase particles are the slowest but they also have movement. (vibrations). ...
Tutorial 1
... 12. Which of the following can form hydrogen bonds with water? CH 3OCH3, CH4, HF, CHOOH 13. Which of the following species are capable of hydrogen bonding among themselves? H 2S, C6H6, CH3OH 14. Diethyl ether has a boiling point of 34.5 C and 1-butanol has a boiling point of 117 C: Diethyl ether ...
... 12. Which of the following can form hydrogen bonds with water? CH 3OCH3, CH4, HF, CHOOH 13. Which of the following species are capable of hydrogen bonding among themselves? H 2S, C6H6, CH3OH 14. Diethyl ether has a boiling point of 34.5 C and 1-butanol has a boiling point of 117 C: Diethyl ether ...
Fall Final 2009
... d. All molecules move randomly in zigzag directions. e. All the molecules have the same velocity. ...
... d. All molecules move randomly in zigzag directions. e. All the molecules have the same velocity. ...
1. All matter is made up of
... 10. A substance made up of two or more elements that have been physcially combined is a ____________ ...
... 10. A substance made up of two or more elements that have been physcially combined is a ____________ ...
NAME…………… - Kcse Online
... (i) Write your name and index number in the spaces provided above. (ii) Sign and write the date of examination in the spaces provided above. (iii) Answer ALL the questions in the spaces provided. (iv) Mathematical tables and silent electronic calculators may be used. (v) All working must be clearly ...
... (i) Write your name and index number in the spaces provided above. (ii) Sign and write the date of examination in the spaces provided above. (iii) Answer ALL the questions in the spaces provided. (iv) Mathematical tables and silent electronic calculators may be used. (v) All working must be clearly ...
~The different types of gases~ Oxygen (O2) The most common
... main constituents of biogas are methane and carbon dioxide. Considerable amount of biogas is produced by sludge digestion in the tanks of sewage treatment plants, which can be referred to as sewage gas. The anaerobic fermentation of organic residues and agricultural waste and garbage tips produce bi ...
... main constituents of biogas are methane and carbon dioxide. Considerable amount of biogas is produced by sludge digestion in the tanks of sewage treatment plants, which can be referred to as sewage gas. The anaerobic fermentation of organic residues and agricultural waste and garbage tips produce bi ...
Tutorial 1
... 12. Which of the following can form hydrogen bonds with water? CH3OCH3, CH4, HF, CHOOH 13. Which of the following species are capable of hydrogen bonding among themselves? H2S, C6H6, CH3OH 14. Diethyl ether has a boiling point of 34.5 C and 1-butanol has a boiling point of 117 C: Diethyl ether ...
... 12. Which of the following can form hydrogen bonds with water? CH3OCH3, CH4, HF, CHOOH 13. Which of the following species are capable of hydrogen bonding among themselves? H2S, C6H6, CH3OH 14. Diethyl ether has a boiling point of 34.5 C and 1-butanol has a boiling point of 117 C: Diethyl ether ...
AP Chemistry Unit 3 Test Review Topics Covered: Gases Liquids
... equilibrium water vapor pressure is higher due to the higher atmospheric pressure equilibrium water vapor pressure is lower due to the higher atmospheric pressure equilibrium water vapor pressure equals the atmospheric pressure at a lower temperature water molecules have a higher average kinetic ene ...
... equilibrium water vapor pressure is higher due to the higher atmospheric pressure equilibrium water vapor pressure is lower due to the higher atmospheric pressure equilibrium water vapor pressure equals the atmospheric pressure at a lower temperature water molecules have a higher average kinetic ene ...
R= 8.31 J/mol K = 0.0821 L atm/mol K = 62.4 L torr/mol K PV = nRT
... ______16. The pressure and kelvin temperature of a gas with a volume of 10.00 L are both doubled. The new volume of the gas is A) 10.00 L B) 2.500 L C) 40.00 L D) 5.000 L E) 20.00 L ______17. 20.00 mL of Ne gas is heated from 127°̊ C to 327°̊ C, with the pressure constant. The new volume of the gas ...
... ______16. The pressure and kelvin temperature of a gas with a volume of 10.00 L are both doubled. The new volume of the gas is A) 10.00 L B) 2.500 L C) 40.00 L D) 5.000 L E) 20.00 L ______17. 20.00 mL of Ne gas is heated from 127°̊ C to 327°̊ C, with the pressure constant. The new volume of the gas ...
1. a) 25% b)86% 2. For my opinion, I think the way to make
... It is used to determine the absorption light from a sample and it can be used as a detector of HPLC. The concentration of analyst in solution can be determined by measuring the absorbance of single wavelength and applying the Beer-Lambert Law. First, place the sample in the Uv-Vis beam and record th ...
... It is used to determine the absorption light from a sample and it can be used as a detector of HPLC. The concentration of analyst in solution can be determined by measuring the absorbance of single wavelength and applying the Beer-Lambert Law. First, place the sample in the Uv-Vis beam and record th ...
File
... _______27. The ratio of the densities of the gas in vessel A to the gas in vessel B. _______28. The ratio of the pressures of the gas in vessel A to the gas in vessel B. _______29. The ratio of the average kinetic energies of the gas in vessel A to the gas in vessel B. _______30. 36.0 kJ of heat are ...
... _______27. The ratio of the densities of the gas in vessel A to the gas in vessel B. _______28. The ratio of the pressures of the gas in vessel A to the gas in vessel B. _______29. The ratio of the average kinetic energies of the gas in vessel A to the gas in vessel B. _______30. 36.0 kJ of heat are ...
PDF(343KB)
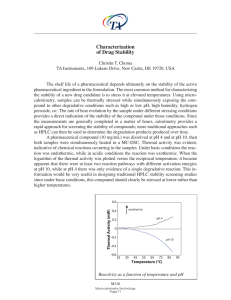
... the stability of a new drug candidate is to stress it at elevated temperatures. Using microcalorimetry, samples can be thermally stressed while simultaneously exposing the compound to other degradative conditions such as high or low pH, high humidity, hydrogen peroxide, etc. The rate of heat evoluti ...
... the stability of a new drug candidate is to stress it at elevated temperatures. Using microcalorimetry, samples can be thermally stressed while simultaneously exposing the compound to other degradative conditions such as high or low pH, high humidity, hydrogen peroxide, etc. The rate of heat evoluti ...
What is Matter PowerPoint
... particles of a liquid can easily slide past one another. Liquids have definite volume, but take the shape of their container. Liquids are hard to compress because their particles are close together. ...
... particles of a liquid can easily slide past one another. Liquids have definite volume, but take the shape of their container. Liquids are hard to compress because their particles are close together. ...
Solute
... Solid – definite volume and shape; particles packed in fixed positions. Liquid – definite volume but indefinite shape; particles close together but not in fixed positions Gas – neither definite volume nor definite shape; particles are at great distances from one another Plasma – high temperature, io ...
... Solid – definite volume and shape; particles packed in fixed positions. Liquid – definite volume but indefinite shape; particles close together but not in fixed positions Gas – neither definite volume nor definite shape; particles are at great distances from one another Plasma – high temperature, io ...
42.89 KB
... 9. How much energy is required to transform 14.0 g ice at 0.0oC to steam at 110.0 oC? Given the following information: Specific heat capacities: ice, 2.1 J/g.oC; liquid, 4.2 J/g.oC; steam, 2.0 J/g.oC; ...
... 9. How much energy is required to transform 14.0 g ice at 0.0oC to steam at 110.0 oC? Given the following information: Specific heat capacities: ice, 2.1 J/g.oC; liquid, 4.2 J/g.oC; steam, 2.0 J/g.oC; ...
Midterm Review Date
... what occurs when the gas inside the cylinder is heated? A) The number of gas molecules increases. B) The number of collisions between gas molecules per unit time decreases. C) The average velocity of the gas molecules increases. D) The volume of the gas decreases. 54. Under which conditions of tempe ...
... what occurs when the gas inside the cylinder is heated? A) The number of gas molecules increases. B) The number of collisions between gas molecules per unit time decreases. C) The average velocity of the gas molecules increases. D) The volume of the gas decreases. 54. Under which conditions of tempe ...
1) Which of the following correctly lists the atoms in order of
... 3) An unknown liquid was vaporized in an experiment in order to find its molar mass. Calculate the molar mass of this substance from the following recorded data: Volume of the flask: ...
... 3) An unknown liquid was vaporized in an experiment in order to find its molar mass. Calculate the molar mass of this substance from the following recorded data: Volume of the flask: ...
Paper chromatography
... Mikhail Tswett, Russian, 1872-1919 In 1906 Tswett used to chromatography to separate plant pigments, He called the new technique chromatography because the result of the analysis was • Most materials in our surroundings are mixtures of two or more components. Mixtures are either homogeneous or het ...
... Mikhail Tswett, Russian, 1872-1919 In 1906 Tswett used to chromatography to separate plant pigments, He called the new technique chromatography because the result of the analysis was • Most materials in our surroundings are mixtures of two or more components. Mixtures are either homogeneous or het ...
Chem 30A, Test Review #2
... A 945-mL balloon contains 2.75 g of a gaseous substance 24oC and 1.00 atm. What is the density of the gas (in g/mL)? (b) If the density of air is 1.20 g/L under the same condition, will the balloon float in air or fall to the ground? Explain. ...
... A 945-mL balloon contains 2.75 g of a gaseous substance 24oC and 1.00 atm. What is the density of the gas (in g/mL)? (b) If the density of air is 1.20 g/L under the same condition, will the balloon float in air or fall to the ground? Explain. ...
Spring Benchmark Exam
... 30. A technician prepared a solution by heating 100 milliliters of distilled water while adding KCl crystals until no more KCl would dissolve. She then capped the clear solution and set it aside on the lab bench. After several hours she noticed the solution had become cloudy and some solid had settl ...
... 30. A technician prepared a solution by heating 100 milliliters of distilled water while adding KCl crystals until no more KCl would dissolve. She then capped the clear solution and set it aside on the lab bench. After several hours she noticed the solution had become cloudy and some solid had settl ...
lab-6-chrmatography
... THIS allowing to carry out qualitative and quantitative analysis of chemical components in complex mixtures. ...
... THIS allowing to carry out qualitative and quantitative analysis of chemical components in complex mixtures. ...
Gas chromatography

Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture (the relative amounts of such components can also be determined). In some situations, GC may help in identifying a compound. In preparative chromatography, GC can be used to prepare pure compounds from a mixture.In gas chromatography, the mobile phase (or ""moving phase"") is a carrier gas, usually an inert gas such as helium or an unreactive gas such as nitrogen. The stationary phase is a microscopic layer of liquid or polymer on an inert solid support, inside a piece of glass or metal tubing called a column (a homage to the fractionating column used in distillation). The instrument used to perform gas chromatography is called a gas chromatograph (or ""aerograph"", ""gas separator"").The gaseous compounds being analyzed interact with the walls of the column, which is coated with a stationary phase. This causes each compound to elute at a different time, known as the retention time of the compound. The comparison of retention times is what gives GC its analytical usefulness.Gas chromatography is in principle similar to column chromatography (as well as other forms of chromatography, such as HPLC, TLC), but has several notable differences. First, the process of separating the compounds in a mixture is carried out between a liquid stationary phase and a gas mobile phase, whereas in column chromatography the stationary phase is a solid and the mobile phase is a liquid. (Hence the full name of the procedure is ""Gas–liquid chromatography"", referring to the mobile and stationary phases, respectively.) Second, the column through which the gas phase passes is located in an oven where the temperature of the gas can be controlled, whereas column chromatography (typically) has no such temperature control. Finally, the concentration of a compound in the gas phase is solely a function of the vapor pressure of the gas.Gas chromatography is also similar to fractional distillation, since both processes separate the components of a mixture primarily based on boiling point (or vapor pressure) differences. However, fractional distillation is typically used to separate components of a mixture on a large scale, whereas GC can be used on a much smaller scale (i.e. microscale).Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). These alternative names, as well as their respective abbreviations, are frequently used in scientific literature. Strictly speaking, GLPC is the most correct terminology, and is thus preferred by many authors.