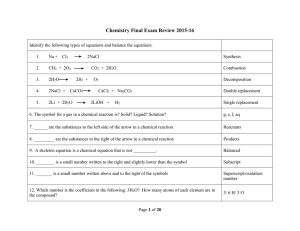
Second Semester Extra Review
... 1. What is a reversible reaction? 2. Define equilibrium. 3. What two substances do you NOT write in an equilibrium expression? 4. Find the equilibrium constant for 2 CO + O2 2 CO2 if the concentration of CO is 0.500M, O2 is 1.50M and CO2 is 0.250M. 5. Calculate the Ka of HF if [HF] = 0.500 M and the ...
... 1. What is a reversible reaction? 2. Define equilibrium. 3. What two substances do you NOT write in an equilibrium expression? 4. Find the equilibrium constant for 2 CO + O2 2 CO2 if the concentration of CO is 0.500M, O2 is 1.50M and CO2 is 0.250M. 5. Calculate the Ka of HF if [HF] = 0.500 M and the ...
Review 3
... 1. How many double covalent bonds are in an alkane? An alkene? An alkyne? 2. How many valence electrons does a carbon atom have? 3. Alkanes are hydrocarbons that contain what type of bonds? 4. Hydrocarbons containing a saturated carbon ring are called ____. 5. Be able to recognize a condensed struct ...
... 1. How many double covalent bonds are in an alkane? An alkene? An alkyne? 2. How many valence electrons does a carbon atom have? 3. Alkanes are hydrocarbons that contain what type of bonds? 4. Hydrocarbons containing a saturated carbon ring are called ____. 5. Be able to recognize a condensed struct ...
CHEMICAL REACTION
... side of the equation in a format that can be revised. Revise the tally with each change. • Use coefficients in front of the elements and compounds to balance the number of atoms of each element. • RIGHT: 2NaNO3 • WRONG:Na2NO3 ...
... side of the equation in a format that can be revised. Revise the tally with each change. • Use coefficients in front of the elements and compounds to balance the number of atoms of each element. • RIGHT: 2NaNO3 • WRONG:Na2NO3 ...
Chemistry Midterm Review 2006
... and gases in regards to density, compressibility, particle size, shape, volume, kinetic energy, attractive forces and movement 3. Explain how a solid melts into a liquid using kinetic energy in your explanation. 4. Define boiling point? 5. What is the difference between normal boiling point and boil ...
... and gases in regards to density, compressibility, particle size, shape, volume, kinetic energy, attractive forces and movement 3. Explain how a solid melts into a liquid using kinetic energy in your explanation. 4. Define boiling point? 5. What is the difference between normal boiling point and boil ...
Chemistry Midterm Review 2006
... gases in regards to density, compressibility, particle size, shape, volume, kinetic energy, attractive forces and movement 3. Explain how a solid melts into a liquid using kinetic energy in your explanation. 4. Define boiling point? 5. What is the difference between normal boiling point and boiling ...
... gases in regards to density, compressibility, particle size, shape, volume, kinetic energy, attractive forces and movement 3. Explain how a solid melts into a liquid using kinetic energy in your explanation. 4. Define boiling point? 5. What is the difference between normal boiling point and boiling ...
Name__________________________________ Block______
... 1. Water is matter because it has mass and takes up space. 2. Ideas are matter because they have mass. 3. Making ice cubes is a chemical change. 4. Boiling point and freezing point are physical properties. 5. Sodium chloride is an element. 6. The composition of a mixture never varies. ...
... 1. Water is matter because it has mass and takes up space. 2. Ideas are matter because they have mass. 3. Making ice cubes is a chemical change. 4. Boiling point and freezing point are physical properties. 5. Sodium chloride is an element. 6. The composition of a mixture never varies. ...
AP Exam One Retake Qualifying Assignment
... takes the shape and volume of its container gaseous state of matter at a temperature less than its boiling point rusting of metal NaCl in the reaction between sodium metal and chlorine gas ...
... takes the shape and volume of its container gaseous state of matter at a temperature less than its boiling point rusting of metal NaCl in the reaction between sodium metal and chlorine gas ...
Type Of Chemical Reaction
... 36. The average kinetic energy of particles of a substance is directly proportional to the ____________ of a substance. 37. Water could be made to boil at a temperature higher than 100 °C by: ...
... 36. The average kinetic energy of particles of a substance is directly proportional to the ____________ of a substance. 37. Water could be made to boil at a temperature higher than 100 °C by: ...
General Chemistry Sample Exam 2 and Outline
... it. The reaction produces sodium sulfate, carbon dioxide and water. i) Write the molecular, ionic and net ionic equation and balance the equation (include the phases). ii) What is the limiting reagent and how much remains if 35 ml of 6.0 M sulfuric acid is spilled and 50 grams of sodium bicarbonate ...
... it. The reaction produces sodium sulfate, carbon dioxide and water. i) Write the molecular, ionic and net ionic equation and balance the equation (include the phases). ii) What is the limiting reagent and how much remains if 35 ml of 6.0 M sulfuric acid is spilled and 50 grams of sodium bicarbonate ...
Chapter 14…Kinetic Theory
... Which substance is least soluble at 10C? Which two substances have the same solubility at 72C? 80 grams of KBr placed in 60C creates a (saturated/unsaturated/supersaturated) solution. When Be(NO3)2 (aq) and NaOH (aq) are mixed together, the resulting precipitate is: Placing a solute in a solvent ...
... Which substance is least soluble at 10C? Which two substances have the same solubility at 72C? 80 grams of KBr placed in 60C creates a (saturated/unsaturated/supersaturated) solution. When Be(NO3)2 (aq) and NaOH (aq) are mixed together, the resulting precipitate is: Placing a solute in a solvent ...
Chapter 4: Properties of Gases
... C6H12O6(s) in a bomb calorimeter (constant volume) is 47.0 kJ. Then for one mole of glucose, the quantity of heat evolved (in kJ) is (according to the equation): C6H12O6 (s) + 6O2 (g) 6CO2 (g) + 6H2O (ℓ); H = ? (a) 2.82x103 ...
... C6H12O6(s) in a bomb calorimeter (constant volume) is 47.0 kJ. Then for one mole of glucose, the quantity of heat evolved (in kJ) is (according to the equation): C6H12O6 (s) + 6O2 (g) 6CO2 (g) + 6H2O (ℓ); H = ? (a) 2.82x103 ...
Chemical Reactions - Mr. Brown`s Science Town
... Example: Chromium (III) sulfite Cr+3SO3-2 = Cr2(SO3)3 d. Molecular Compounds – Prefixes = Subscripts ...
... Example: Chromium (III) sulfite Cr+3SO3-2 = Cr2(SO3)3 d. Molecular Compounds – Prefixes = Subscripts ...
Name__________________________ Period_______ Word
... example of a reaction condition is the heat symbol (∆ ), which indicates that the reactants were heated. An equation can also indicate the physical state of the reactants and products. The physical states of matter symbols are listed below. ...
... example of a reaction condition is the heat symbol (∆ ), which indicates that the reactants were heated. An equation can also indicate the physical state of the reactants and products. The physical states of matter symbols are listed below. ...
Ionic Compounds 1. What is the formula for aluminum phosphate
... Solutions – Like Dissolves Like, Molarity, Reaction Types, Solubility 1. An unknown substance dissolves in water but not in benzene (a nonpolar solvent). Molecules of what type are present in the substance? 2. A 87.2-g sample of SrCl2 is dissolved in 112.5 mL of solution. Calculate the molarity of ...
... Solutions – Like Dissolves Like, Molarity, Reaction Types, Solubility 1. An unknown substance dissolves in water but not in benzene (a nonpolar solvent). Molecules of what type are present in the substance? 2. A 87.2-g sample of SrCl2 is dissolved in 112.5 mL of solution. Calculate the molarity of ...
CHM2045 Final Exam Review, Spring 2017
... CHM2045 Final Exam Review, Spring 2017 1. The combustion of 40.10 g of a compound which contains only C, H, Cl and O yields 58.57 g of CO2 and 14.98 g of H2O. Another sample of the compound with a mass of 75.00 g is found to contain 22.06 g of Cl. What is the empirical formula of the compound? If th ...
... CHM2045 Final Exam Review, Spring 2017 1. The combustion of 40.10 g of a compound which contains only C, H, Cl and O yields 58.57 g of CO2 and 14.98 g of H2O. Another sample of the compound with a mass of 75.00 g is found to contain 22.06 g of Cl. What is the empirical formula of the compound? If th ...
Ch. 11: Gases
... independent of its chemical identity! • Gas behavior is markedly different than solid or liquid behavior. • We look at a theory that explains why gas behavior is universal and then at the origin of equations that allow us to do gas calculations. ...
... independent of its chemical identity! • Gas behavior is markedly different than solid or liquid behavior. • We look at a theory that explains why gas behavior is universal and then at the origin of equations that allow us to do gas calculations. ...
Oxygen Gas Safety
... Do not remove labels from cylinders or crates (except the return address cover). Do not lift cylinders by the valve. Use suitable cylinder handling and securing equipment. Do not use oil or grease on valves, fittings, or any other equipment. Always close valves using moderate force only, even when t ...
... Do not remove labels from cylinders or crates (except the return address cover). Do not lift cylinders by the valve. Use suitable cylinder handling and securing equipment. Do not use oil or grease on valves, fittings, or any other equipment. Always close valves using moderate force only, even when t ...
Chapter 13…States of Matter
... 7. Saturated solution: cannot hold any more of a given solute at a given temperature 8. Unsaturated solution: can hold more of a given solute at a given temperature 9. Supersaturated solution: can hold more of a given solute than it should theoretically hold (requires heating and stirring) 10. Preci ...
... 7. Saturated solution: cannot hold any more of a given solute at a given temperature 8. Unsaturated solution: can hold more of a given solute at a given temperature 9. Supersaturated solution: can hold more of a given solute than it should theoretically hold (requires heating and stirring) 10. Preci ...
Review Sheet Filled Out
... Can’t tell where an electron is at any moment in time – the uncertainty principle There’s more – the list could be long! ...
... Can’t tell where an electron is at any moment in time – the uncertainty principle There’s more – the list could be long! ...
Name - cloudfront.net
... How is the ideal gas law usually written? The combined gas law relates which items together? If a balloon is squeezed, what happens to the pressure of the gas inside the balloon? If the volume of a container of gas is reduced, what will happen to the pressure inside the container? In general, for a ...
... How is the ideal gas law usually written? The combined gas law relates which items together? If a balloon is squeezed, what happens to the pressure of the gas inside the balloon? If the volume of a container of gas is reduced, what will happen to the pressure inside the container? In general, for a ...
03 nanoparticles part 7 File - e-learning
... The technique consists in the chemical reaction of two or more compounds in gas phase and the successive deposition of the produced solid material. The technique results costly for those materials exhibiting low vapor pressure values (ceramics). In the other cases, the technique offers all advantage ...
... The technique consists in the chemical reaction of two or more compounds in gas phase and the successive deposition of the produced solid material. The technique results costly for those materials exhibiting low vapor pressure values (ceramics). In the other cases, the technique offers all advantage ...
1 - mvhs-fuhsd.org
... e. Physical property: a quality or condition of a substance that can be observed or measured without changing the substances composition. EX: has a green color f. Chemical property: the ability of a substance to undergo a change that produces matter with a different composition than the original mat ...
... e. Physical property: a quality or condition of a substance that can be observed or measured without changing the substances composition. EX: has a green color f. Chemical property: the ability of a substance to undergo a change that produces matter with a different composition than the original mat ...
Fundamentals of Physical Chemistry
... measured at 764 mm Hg and 150 oC. Write the formula of the compound. 4. Nitrogen dioxide can not be obtained in a pure form in the gas phase because it exists as a mixture of NO2 and N2O4. At 25 oC and 0.98 atm, the density of this gas mixture is 2.7 gL-1. What is the partial pressure of each gas. 5 ...
... measured at 764 mm Hg and 150 oC. Write the formula of the compound. 4. Nitrogen dioxide can not be obtained in a pure form in the gas phase because it exists as a mixture of NO2 and N2O4. At 25 oC and 0.98 atm, the density of this gas mixture is 2.7 gL-1. What is the partial pressure of each gas. 5 ...
Element - the simplest form of matter that can exist under normal
... Elements are the building blocks for all other substances There are now 117 known elements (as of 2006). All elements after uranium on the periodic table are man-made. A compound is a chemical combination of two or more different elements joined together in a fixed proportion. Every compound has its ...
... Elements are the building blocks for all other substances There are now 117 known elements (as of 2006). All elements after uranium on the periodic table are man-made. A compound is a chemical combination of two or more different elements joined together in a fixed proportion. Every compound has its ...
2nd Semester Exam Review
... • Gasses consist of small particles that take up little volume relative to the volume of empty space around them – Gas molecules are very far apart and therefore don’t experience attractive or repulsive forces. ...
... • Gasses consist of small particles that take up little volume relative to the volume of empty space around them – Gas molecules are very far apart and therefore don’t experience attractive or repulsive forces. ...
Gas chromatography

Gas chromatography (GC) is a common type of chromatography used in analytical chemistry for separating and analyzing compounds that can be vaporized without decomposition. Typical uses of GC include testing the purity of a particular substance, or separating the different components of a mixture (the relative amounts of such components can also be determined). In some situations, GC may help in identifying a compound. In preparative chromatography, GC can be used to prepare pure compounds from a mixture.In gas chromatography, the mobile phase (or ""moving phase"") is a carrier gas, usually an inert gas such as helium or an unreactive gas such as nitrogen. The stationary phase is a microscopic layer of liquid or polymer on an inert solid support, inside a piece of glass or metal tubing called a column (a homage to the fractionating column used in distillation). The instrument used to perform gas chromatography is called a gas chromatograph (or ""aerograph"", ""gas separator"").The gaseous compounds being analyzed interact with the walls of the column, which is coated with a stationary phase. This causes each compound to elute at a different time, known as the retention time of the compound. The comparison of retention times is what gives GC its analytical usefulness.Gas chromatography is in principle similar to column chromatography (as well as other forms of chromatography, such as HPLC, TLC), but has several notable differences. First, the process of separating the compounds in a mixture is carried out between a liquid stationary phase and a gas mobile phase, whereas in column chromatography the stationary phase is a solid and the mobile phase is a liquid. (Hence the full name of the procedure is ""Gas–liquid chromatography"", referring to the mobile and stationary phases, respectively.) Second, the column through which the gas phase passes is located in an oven where the temperature of the gas can be controlled, whereas column chromatography (typically) has no such temperature control. Finally, the concentration of a compound in the gas phase is solely a function of the vapor pressure of the gas.Gas chromatography is also similar to fractional distillation, since both processes separate the components of a mixture primarily based on boiling point (or vapor pressure) differences. However, fractional distillation is typically used to separate components of a mixture on a large scale, whereas GC can be used on a much smaller scale (i.e. microscale).Gas chromatography is also sometimes known as vapor-phase chromatography (VPC), or gas–liquid partition chromatography (GLPC). These alternative names, as well as their respective abbreviations, are frequently used in scientific literature. Strictly speaking, GLPC is the most correct terminology, and is thus preferred by many authors.