AP Bio Test Questions
... carbon, hydrogen, oxygen, sulfur, nitrogen, phosphorous carbon, hydrogen, oxygen in a 1:2:1 format mainly oxygen with carbon and hydrogen (hydrocarbons) hydrogen, oxygen, carbon, occasionally sulfur ...
... carbon, hydrogen, oxygen, sulfur, nitrogen, phosphorous carbon, hydrogen, oxygen in a 1:2:1 format mainly oxygen with carbon and hydrogen (hydrocarbons) hydrogen, oxygen, carbon, occasionally sulfur ...
Quiz #3 - San Diego Mesa College
... A) the combustion of wood B) the combustion of gasoline in a car engine C) the metabolism of glucose in a living cell D) the build-up of glucose from carbon dioxide and water E) the breakdown of brown fat in babies to generate heat Q. 4: An ATP molecule is made of following molecular components A) t ...
... A) the combustion of wood B) the combustion of gasoline in a car engine C) the metabolism of glucose in a living cell D) the build-up of glucose from carbon dioxide and water E) the breakdown of brown fat in babies to generate heat Q. 4: An ATP molecule is made of following molecular components A) t ...
Naming Ionic Compounds with Transition Metals
... *The charge of the metal is given in the name as a Roman numeral. ...
... *The charge of the metal is given in the name as a Roman numeral. ...
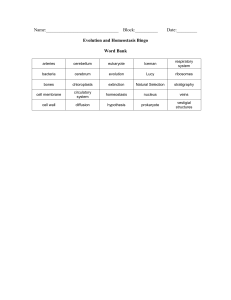
Bingo definitions
... 4. The part of an organism that becomes a fossil. 5. The first life on earth. 6. The oldest preserved human found. 7. The first hominid fossil found. 8. Organisms change over time. 9. All members of a species die forever. 10. Structures that have no function such as an appendix in humans. 11. Techni ...
... 4. The part of an organism that becomes a fossil. 5. The first life on earth. 6. The oldest preserved human found. 7. The first hominid fossil found. 8. Organisms change over time. 9. All members of a species die forever. 10. Structures that have no function such as an appendix in humans. 11. Techni ...
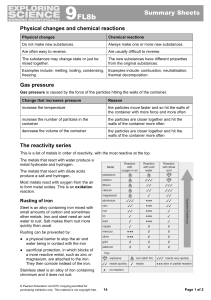
9F Reactivity - Parrs Wood High School
... These substances contain hydrogen and carbon only. They burn in a plentiful supply of air to form carbon dioxide and water: hydrocarbon + oxygen → carbon dioxide + water The test for oxygen is that it relights a glowing splint. An input of energy from a flame or spark is needed to start the combusti ...
... These substances contain hydrogen and carbon only. They burn in a plentiful supply of air to form carbon dioxide and water: hydrocarbon + oxygen → carbon dioxide + water The test for oxygen is that it relights a glowing splint. An input of energy from a flame or spark is needed to start the combusti ...
Gas Exchange
... • Organisms that live in aquatic, marine, or even moist terrestrial environments and which have all tissues within 1 mm of the moist integument, do not have specialized gas exchange structures nor do they require a circulatory system to transport oxygen. In some, such as nemertean worms a circulator ...
... • Organisms that live in aquatic, marine, or even moist terrestrial environments and which have all tissues within 1 mm of the moist integument, do not have specialized gas exchange structures nor do they require a circulatory system to transport oxygen. In some, such as nemertean worms a circulator ...
Write on the role of metalloenzymes in biological systems.
... Moreover, metal coordination can impose changes to protein structure and dynamics, which, in some cases, result in the modulation of function (Robert, 2011). This mechanism is indeed exploited by the cellular machinery, in which Ca2+ acts as a physiological and cellular signal carrier. One of the mo ...
... Moreover, metal coordination can impose changes to protein structure and dynamics, which, in some cases, result in the modulation of function (Robert, 2011). This mechanism is indeed exploited by the cellular machinery, in which Ca2+ acts as a physiological and cellular signal carrier. One of the mo ...
Unit 2 Section B Supplement b
... Identify three questions that should be answered prior to mining a metallic ore at a given location. ...
... Identify three questions that should be answered prior to mining a metallic ore at a given location. ...
Metabolism
... and glutamine can all be interconverted. – this is the source of the amino group of amino acids and amino sugars. ...
... and glutamine can all be interconverted. – this is the source of the amino group of amino acids and amino sugars. ...
Cellular Respiration (CR
... eukaryotic cells (location and names of processes involved)? How many ATP’s are produced from each cell type? --------------------------------------------Define: The process of releasing energy (ATP) from food (glucose). Both consumers AND producers need to go through cellular respiration to turn th ...
... eukaryotic cells (location and names of processes involved)? How many ATP’s are produced from each cell type? --------------------------------------------Define: The process of releasing energy (ATP) from food (glucose). Both consumers AND producers need to go through cellular respiration to turn th ...
Document
... system, which involves a series of proteins that can carry out the energy transfer reactions. Note the role of atmospheric oxygen in this! ...
... system, which involves a series of proteins that can carry out the energy transfer reactions. Note the role of atmospheric oxygen in this! ...
Metals and non-metals III IMPORTANT POINTS Non-metals
... 1. a. Magnesium, chromium and sodium are all metals, hence, they react with oxygen to form basic oxides b. Chromium, as it is a transition metal. Metals have high density and coloured compounds are formed by transition metals. c. Bromine - the formula is Br2, that is, two atoms of bromine. d. Bromin ...
... 1. a. Magnesium, chromium and sodium are all metals, hence, they react with oxygen to form basic oxides b. Chromium, as it is a transition metal. Metals have high density and coloured compounds are formed by transition metals. c. Bromine - the formula is Br2, that is, two atoms of bromine. d. Bromin ...
snc 2do unit: chemistry unit test review questions
... c) What type of reaction is this? 5. Identify the type of reaction, and write a balanced chemical equation for: A) zinc + iron (III) nitrate -------> ________ + ______________ B) potassium + oxygen ------> _________________ C) magnesium carbonate -----> magnesium oxide + carbon dioxide D) __________ ...
... c) What type of reaction is this? 5. Identify the type of reaction, and write a balanced chemical equation for: A) zinc + iron (III) nitrate -------> ________ + ______________ B) potassium + oxygen ------> _________________ C) magnesium carbonate -----> magnesium oxide + carbon dioxide D) __________ ...
Name: Date: Concept Check Questions Chapter 9 Cellular
... 1. During the redox reaction in glycolysis (step 6 in figure 9.9 in the orange book), which molecule acts as the oxidizing agent? The reducing agent? 9.3 The citric acid cycle completes the energy-yielding oxidation of organic molecules ...
... 1. During the redox reaction in glycolysis (step 6 in figure 9.9 in the orange book), which molecule acts as the oxidizing agent? The reducing agent? 9.3 The citric acid cycle completes the energy-yielding oxidation of organic molecules ...
13-4 Applications of Genetic Engineering
... Genes from other organisms can work together and still function (ex. luciferase and tobacco plants) Transgenic—contain genes from other organisms A gene from one organism can be inserted into cells from another organism. Genetic engineering has spurred the growth of biotechnology, a new indust ...
... Genes from other organisms can work together and still function (ex. luciferase and tobacco plants) Transgenic—contain genes from other organisms A gene from one organism can be inserted into cells from another organism. Genetic engineering has spurred the growth of biotechnology, a new indust ...
25 Elements
... an electron to form Li+. It is the first element within the second period. • Lithium is mixed (alloyed) with Aluminium and magnesium for light-weight alloys, and is also used in batteries, some greases, some glasses, and in medicine. ...
... an electron to form Li+. It is the first element within the second period. • Lithium is mixed (alloyed) with Aluminium and magnesium for light-weight alloys, and is also used in batteries, some greases, some glasses, and in medicine. ...
Cells “The Building Blocks of Life”
... – Based on limited information or knowledge – Verified of falsified through experimentation ...
... – Based on limited information or knowledge – Verified of falsified through experimentation ...
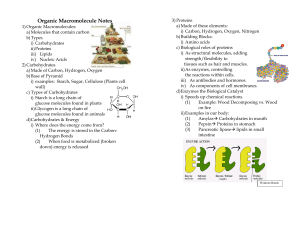
Organic Macromolecule Notes
... a) Made of these elements: i) Carbon, Hydrogen, Oxygen, Nitrogen b) Building Blocks: i) Amino acids c) Biological roles of proteins i) As structural molecules, adding strength/flexibility to tissues such as hair and muscles. ii) As enzymes, controlling the reactions within cells. iii) As antibodies ...
... a) Made of these elements: i) Carbon, Hydrogen, Oxygen, Nitrogen b) Building Blocks: i) Amino acids c) Biological roles of proteins i) As structural molecules, adding strength/flexibility to tissues such as hair and muscles. ii) As enzymes, controlling the reactions within cells. iii) As antibodies ...
BIOCHEM MID SEM EXAM 2014 The Foundations of Biochemistry
... Qu. What is Biochemistry? Provide a brief definition in 30 words or less. The study of biomolecules inside & outside of cells from living systems and how they interact to maintain & perpetuate life via universal physical & chemical laws. Qu. What are the cellular foundations of biochemistry? - Cells ...
... Qu. What is Biochemistry? Provide a brief definition in 30 words or less. The study of biomolecules inside & outside of cells from living systems and how they interact to maintain & perpetuate life via universal physical & chemical laws. Qu. What are the cellular foundations of biochemistry? - Cells ...
Student worksheet for prokaryotic, animal and plant cells
... Student—please print this worksheet and complete it as you interact with the tutorial. The completed worksheet should be turned in to your assigned teacher. Tutorial: Comparison of prokaryotic, animal and plant cells 1. Plants and animals have eukaryotic cells. What is the other type of cell in this ...
... Student—please print this worksheet and complete it as you interact with the tutorial. The completed worksheet should be turned in to your assigned teacher. Tutorial: Comparison of prokaryotic, animal and plant cells 1. Plants and animals have eukaryotic cells. What is the other type of cell in this ...
Evolution of metal ions in biological systems
Evolution of Metal Ions in Biological Systems refers to the incorporation of metallic ions into living organisms and how it has changed over time. Metal ions have been associated with biological systems for billions of years, but only in the last century have scientists began to truly appreciate the scale of their influence. Major (iron, manganese, magnesium and zinc) and minor (copper, cobalt, nickel, molybdenum, tungsten) metal ions have symbiotically aligned with living organisms and the associated complexes have evolved over time.