I CAN write Chemical formulas
... the oxidation numbers and write the oxidation number (without plus or minus) of one element as the subscript of the other element. 3. Reduce the subscripts (number of atoms) to their simplest form, if needed. WHAT IS THE CHEMICAL FORMULA FOR CALCIUM CHLORIDE? ...
... the oxidation numbers and write the oxidation number (without plus or minus) of one element as the subscript of the other element. 3. Reduce the subscripts (number of atoms) to their simplest form, if needed. WHAT IS THE CHEMICAL FORMULA FOR CALCIUM CHLORIDE? ...
Review Quiz 7 - ltcconline.net
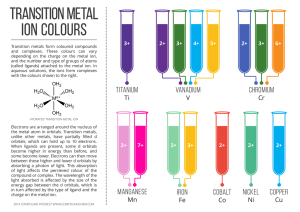
... Transition metal ions lose the s-orbital electrons before they lose the d orbital electrons. This happens because the energy of the (n-1)d orbital is significantly less than the ns electrons. Coordination compound: A complex ion and counterion with no net charge. Complex ion: Charged species consist ...
... Transition metal ions lose the s-orbital electrons before they lose the d orbital electrons. This happens because the energy of the (n-1)d orbital is significantly less than the ns electrons. Coordination compound: A complex ion and counterion with no net charge. Complex ion: Charged species consist ...
TABLE 3–1 Some Common Types of Enzymes
... catalyze the rearrangement of bonds within a single molecule. catalyze polymerization reactions such as the synthesis of DNA and RNA. catalyze the addition of phosphate groups to molecules. Protein kinases are an important group of kinases that attach phosphate groups to proteins. catalyze the hydro ...
... catalyze the rearrangement of bonds within a single molecule. catalyze polymerization reactions such as the synthesis of DNA and RNA. catalyze the addition of phosphate groups to molecules. Protein kinases are an important group of kinases that attach phosphate groups to proteins. catalyze the hydro ...
DEC 2016 BIO: some useful words File
... the site on an enzyme where the substrates bind. when a solute is dissolved in a solution – it mixes completely and is transparent The solid material that dissolves in a solvent A liquid that dissolves a solute when a solid becomes part of a solution a difference in concentration across a membrane t ...
... the site on an enzyme where the substrates bind. when a solute is dissolved in a solution – it mixes completely and is transparent The solid material that dissolves in a solvent A liquid that dissolves a solute when a solid becomes part of a solution a difference in concentration across a membrane t ...
transition metal - Compound Interest
... the metal atom in orbitals. Transition metals, unlike other metals, have partially filled d orbitals, which can hold up to 10 electrons. When ligands are present, some d orbitals become higher in energy than before, and some become lower. Electrons can then move between these higher and lower d orbi ...
... the metal atom in orbitals. Transition metals, unlike other metals, have partially filled d orbitals, which can hold up to 10 electrons. When ligands are present, some d orbitals become higher in energy than before, and some become lower. Electrons can then move between these higher and lower d orbi ...
29th Annual Meeting | American Society of Preventive Oncology
... Our laboratory is involved in the development of safe organo-metallic complexes using natural biological molecules as ligands. These are nutritional supplements as well as candidate chemotherapy agents. The initial phase of development of these materials is based on the electronic signaling properti ...
... Our laboratory is involved in the development of safe organo-metallic complexes using natural biological molecules as ligands. These are nutritional supplements as well as candidate chemotherapy agents. The initial phase of development of these materials is based on the electronic signaling properti ...
Metals in Redox Biology - University of Nebraska–Lincoln
... -HscA binds to a conserved stretch of amino acids (LPPVK) in IscU. The LPPVK motif is located near a highly conserved Cys (Cys106) residue in IscU, so IscU binding to HscAB and subsequent ATP hydrolysis might alter the interaction of this cysteine with clusters on IscU. ...
... -HscA binds to a conserved stretch of amino acids (LPPVK) in IscU. The LPPVK motif is located near a highly conserved Cys (Cys106) residue in IscU, so IscU binding to HscAB and subsequent ATP hydrolysis might alter the interaction of this cysteine with clusters on IscU. ...
Slide 1
... far more powerful than glycolysis at recovering energy from food molecules and is where the bulk of the energy used by eukaryotic cells is extracted. ...
... far more powerful than glycolysis at recovering energy from food molecules and is where the bulk of the energy used by eukaryotic cells is extracted. ...
Unit 7 Review
... Which of the following metals could be added to aqueous iron (II) oxide to produce solid iron metal? ...
... Which of the following metals could be added to aqueous iron (II) oxide to produce solid iron metal? ...
NAME: IDU DOREEN MATRIC NO: 14/SCI03/011 COURSE
... environment of the metal-binding site can be obtained by replacing the Zn2+ ion with a paramagnetic, chromophoric metal-ion probe. Co2+ readily replaces Zn2+ – they prefer similar coordination environments, have comparable ionic radii, and display similar reactivity patterns. The resultant cobalt-su ...
... environment of the metal-binding site can be obtained by replacing the Zn2+ ion with a paramagnetic, chromophoric metal-ion probe. Co2+ readily replaces Zn2+ – they prefer similar coordination environments, have comparable ionic radii, and display similar reactivity patterns. The resultant cobalt-su ...
Biological Molecules
... Lipids – Disolve in ethanol, then add mixture to water. A milky emulsion is a positive result. Reducing sugars- add Benedicts solution and heat. A brick red colour is a positive test. Protein- add to buirett solution. A dark purple colour is a positive result. Cellulose- Add iodine solution. ...
... Lipids – Disolve in ethanol, then add mixture to water. A milky emulsion is a positive result. Reducing sugars- add Benedicts solution and heat. A brick red colour is a positive test. Protein- add to buirett solution. A dark purple colour is a positive result. Cellulose- Add iodine solution. ...
effective oxidation states applied to endohedral - IQCC
... The concept of oxidation state (OS) is widespread in transition metal chemistry and in the study of redox and catalytic reactions. The reactivity, spin-state, spectroscopic and geometrical features of transition metal (TM) complexes are often rationalized on the basis of the OS of the metal center. ...
... The concept of oxidation state (OS) is widespread in transition metal chemistry and in the study of redox and catalytic reactions. The reactivity, spin-state, spectroscopic and geometrical features of transition metal (TM) complexes are often rationalized on the basis of the OS of the metal center. ...
REACTIVITY OF METALS Extension 1. A new metal has been
... (iii) Magnesium and zinc sulphate solution In a displacement reaction, two metals of different reactivity ‘compete’ for oxygen. In such a reaction, oxidation and reduction occur simultaneously. ...
... (iii) Magnesium and zinc sulphate solution In a displacement reaction, two metals of different reactivity ‘compete’ for oxygen. In such a reaction, oxidation and reduction occur simultaneously. ...
Living Environment Regents Review
... Plants absorb carbon dioxide from the air. Too much carbon dioxide chloroplasts will cause the Earth to heat up Animals can (the greenhouse effect). eat the sugar made to use as energy ...
... Plants absorb carbon dioxide from the air. Too much carbon dioxide chloroplasts will cause the Earth to heat up Animals can (the greenhouse effect). eat the sugar made to use as energy ...
Quizon ch5-6-7-8new.doc
... a. the reactants have more potential energy than the products b. energy is released c. a net input of energy is not required d. all of the above e. none of the above 7. In most land plants, photosynthesis occurs in cells of the __________ of the leaves, because these cells contain the largest number ...
... a. the reactants have more potential energy than the products b. energy is released c. a net input of energy is not required d. all of the above e. none of the above 7. In most land plants, photosynthesis occurs in cells of the __________ of the leaves, because these cells contain the largest number ...
1.5.16(Chem) - mrcarlsonschemistryclass
... Compounds • Atoms bonded together with an IONIC bond are called ionic compounds. • An ionic bond is a METAL bonded with a NONMETAL. • Draw the crystal lattice structure for sodium chloride: ...
... Compounds • Atoms bonded together with an IONIC bond are called ionic compounds. • An ionic bond is a METAL bonded with a NONMETAL. • Draw the crystal lattice structure for sodium chloride: ...
Oxidation Number Rules
... 1. Any uncombined atom, or any atom in a molecule of an element, is assigned an oxidation number of zero. Examples: N2, Cl2, C, Sn, S8 2. The oxidation number of a simple, monatomic ion is the same as the charge on the ion. Examples: Na+ is +1, Cu+2 is +2, Cu+ is +1, F¯ is -1. 3. The oxidation numbe ...
... 1. Any uncombined atom, or any atom in a molecule of an element, is assigned an oxidation number of zero. Examples: N2, Cl2, C, Sn, S8 2. The oxidation number of a simple, monatomic ion is the same as the charge on the ion. Examples: Na+ is +1, Cu+2 is +2, Cu+ is +1, F¯ is -1. 3. The oxidation numbe ...
Redox Reactions Test Review
... 9. Define spectator ion. 10. In the equation Ni + 2 HCl NiCl2 + H2 label the following a. Oxidized: b. Reduced: c. Spectator Ion: 11. In the equation Ca2+ + 2 Li Ca + 2 Li+ label the following a. Oxidized: b. Reduced: ...
... 9. Define spectator ion. 10. In the equation Ni + 2 HCl NiCl2 + H2 label the following a. Oxidized: b. Reduced: c. Spectator Ion: 11. In the equation Ca2+ + 2 Li Ca + 2 Li+ label the following a. Oxidized: b. Reduced: ...
1-2.02 test study guide
... 13.What is the main source of energy for all living things? 14.What are the functions of proteins? 15.Who was the first person to see the cell? 16.What did schleiden and Schwann tell us? 17.What are the 3 principals of the cell theory 18.What does the cell theory apply to? 19.What is the difference ...
... 13.What is the main source of energy for all living things? 14.What are the functions of proteins? 15.Who was the first person to see the cell? 16.What did schleiden and Schwann tell us? 17.What are the 3 principals of the cell theory 18.What does the cell theory apply to? 19.What is the difference ...
1-3 Studying Life
... 6. Plants, some bacteria, and most algae obtain their energy directly from ___________. 7. A _______________ is a signal to which an organism responds. 8. Give 2 examples of external stimuli: _______________________________________. 9. The process by which organisms maintain constant internal condit ...
... 6. Plants, some bacteria, and most algae obtain their energy directly from ___________. 7. A _______________ is a signal to which an organism responds. 8. Give 2 examples of external stimuli: _______________________________________. 9. The process by which organisms maintain constant internal condit ...
Evolution of metal ions in biological systems
Evolution of Metal Ions in Biological Systems refers to the incorporation of metallic ions into living organisms and how it has changed over time. Metal ions have been associated with biological systems for billions of years, but only in the last century have scientists began to truly appreciate the scale of their influence. Major (iron, manganese, magnesium and zinc) and minor (copper, cobalt, nickel, molybdenum, tungsten) metal ions have symbiotically aligned with living organisms and the associated complexes have evolved over time.