1-3_studying_life
... 3. Circle the letter of each sentence that is TRUE about cells. a. A cell is the smallest unit of an organism that is considered alive. b. A multicellular organism may contain trillions of cells. c. A living thing that consists of a single cell is a multicellular organism. d. Organisms are made up o ...
... 3. Circle the letter of each sentence that is TRUE about cells. a. A cell is the smallest unit of an organism that is considered alive. b. A multicellular organism may contain trillions of cells. c. A living thing that consists of a single cell is a multicellular organism. d. Organisms are made up o ...
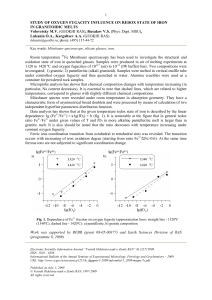
Study of oxygen fugacity influence on redox state of iron in
... Data analysis has shown that at the given temperature redox state of iron is described by the linear dependence: lg (Fe3+/Fe2+) = a·lg(fO2) + b (fig. 1). It is noticeable at the figure that in general redox ratio Fe3+/Fe2+ under given values of T and fO2 in more alkaline pantelleritic melt is larger ...
... Data analysis has shown that at the given temperature redox state of iron is described by the linear dependence: lg (Fe3+/Fe2+) = a·lg(fO2) + b (fig. 1). It is noticeable at the figure that in general redox ratio Fe3+/Fe2+ under given values of T and fO2 in more alkaline pantelleritic melt is larger ...
Magnesium chloride hexahydrate Product Number - Sigma
... Magnesium chloride is a widely used reagent in chemistry and molecular biology as a source of magnesium ion. Magnesium has a variety of biological roles in enzymology, cell membrane and wall structural integrity, muscle cell physiology, and nucleic acid structure.1,2 Magnesium is an essential co-fac ...
... Magnesium chloride is a widely used reagent in chemistry and molecular biology as a source of magnesium ion. Magnesium has a variety of biological roles in enzymology, cell membrane and wall structural integrity, muscle cell physiology, and nucleic acid structure.1,2 Magnesium is an essential co-fac ...
magnesium chloride TDS
... Magnesium chloride is a widely used reagent in chemistry and molecular biology as a source of magnesium ion. Magnesium has a variety of biological roles in enzymology, cell membrane and wall structural integrity, muscle cell physiology, and ...
... Magnesium chloride is a widely used reagent in chemistry and molecular biology as a source of magnesium ion. Magnesium has a variety of biological roles in enzymology, cell membrane and wall structural integrity, muscle cell physiology, and ...
Ch 12 Electrolysis in water
... 2 Cl- Cl2+ 2 e Water may be oxidized to oxygen 2 H2O O2 + 4 H+ + 4 e Chlorine, bromine and iodine will oxidize before oxygen. That is it. ...
... 2 Cl- Cl2+ 2 e Water may be oxidized to oxygen 2 H2O O2 + 4 H+ + 4 e Chlorine, bromine and iodine will oxidize before oxygen. That is it. ...
tetrahedral site
... Magnetite, Fe3O4 crystallizes with the spinel structure. The large oxygen ions are close packed in a cubic arrangement and the smaller Fe ions fill in the gaps. The gaps come in two flavors: tetrahedral site: Fe ion is surrounded by four oxygens octahedral site: Fe ion is surrounded by six o ...
... Magnetite, Fe3O4 crystallizes with the spinel structure. The large oxygen ions are close packed in a cubic arrangement and the smaller Fe ions fill in the gaps. The gaps come in two flavors: tetrahedral site: Fe ion is surrounded by four oxygens octahedral site: Fe ion is surrounded by six o ...
Facts to Remember to help you pass the NYS Science Assessment
... 6.) Most variety of organisms are found in kingdoms 7.) Least variety are found among species. 8.) The endocrine system produces hormones. 9.) Sex cells contain half the normal number of chromosomes found in body cells. 10.) The normal number of chromosomes in a human body cell is 46. 11.) The flowe ...
... 6.) Most variety of organisms are found in kingdoms 7.) Least variety are found among species. 8.) The endocrine system produces hormones. 9.) Sex cells contain half the normal number of chromosomes found in body cells. 10.) The normal number of chromosomes in a human body cell is 46. 11.) The flowe ...
Oxidation-Reduction (Redox) Reactions
... One of these is a redox reaction, and the other is not. How can you tell which is which? You need to determine which species, if any, has lost electrons and which, if any, has gained electrons. We do this by assigning oxidation numbers. ...
... One of these is a redox reaction, and the other is not. How can you tell which is which? You need to determine which species, if any, has lost electrons and which, if any, has gained electrons. We do this by assigning oxidation numbers. ...
PowerPoint Overview for Introduction
... Sodium (0.15%) is another electrolyte that is vital for electrical signaling in nerves. It also regulates the amount of water in the body. ...
... Sodium (0.15%) is another electrolyte that is vital for electrical signaling in nerves. It also regulates the amount of water in the body. ...
Directions: For each organelle you need to, draw a
... Directions: For each organelle you need to describe the function of the cell and draw a reminder picture. ...
... Directions: For each organelle you need to describe the function of the cell and draw a reminder picture. ...
Review Sheet
... Charles Darwin- species of life have descended over time from common ancestry, proposed the scientific theory of evolution resulted from a process that he called natural selection. ...
... Charles Darwin- species of life have descended over time from common ancestry, proposed the scientific theory of evolution resulted from a process that he called natural selection. ...
Concepts in Transition Metal Chemistry – Questions
... Use the information in the Appendix and the value Eө[S2O82−|2SO42−] = 2.00 V to find out which of the following reactions are thermodynamically favourable: (a) oxidation of Cr2+(aq) by Mn3+(aq); (b) oxidation of Mn2+(aq) by Co3+(aq); (c) oxidation of Mn2+(aq) by persulfate; (d) reduction of persulfa ...
... Use the information in the Appendix and the value Eө[S2O82−|2SO42−] = 2.00 V to find out which of the following reactions are thermodynamically favourable: (a) oxidation of Cr2+(aq) by Mn3+(aq); (b) oxidation of Mn2+(aq) by Co3+(aq); (c) oxidation of Mn2+(aq) by persulfate; (d) reduction of persulfa ...
word
... What are some of the model organisms and to what properties of the cell biology do they contribute? What are some advantages and disadvantages of model organisms for research? What are the scientific names of some of the model organisms? How are fluorescent-labeled antibodies viewed binding to a spe ...
... What are some of the model organisms and to what properties of the cell biology do they contribute? What are some advantages and disadvantages of model organisms for research? What are the scientific names of some of the model organisms? How are fluorescent-labeled antibodies viewed binding to a spe ...
metals and non metals
... Q7 In what forms are metals found in nature? With the help of examples,explain how metals react with oxygen and dilute acids. Also write chemical equation for the reactions. Q8 Explain how the following metals are obtained from their compounds by reduction process: (i)Metal X which is low in reactiv ...
... Q7 In what forms are metals found in nature? With the help of examples,explain how metals react with oxygen and dilute acids. Also write chemical equation for the reactions. Q8 Explain how the following metals are obtained from their compounds by reduction process: (i)Metal X which is low in reactiv ...
EFB325 Cell Physiology Welcome to Cell Physiology Course
... agencies, non-gov't organizations, and pharmaceutical companies understanding this material is increasingly important in our daily lives-health and human medicine, food production and safety, forensics, environmental science, biotechnology - Try to confer some awareness of the impact on our lives an ...
... agencies, non-gov't organizations, and pharmaceutical companies understanding this material is increasingly important in our daily lives-health and human medicine, food production and safety, forensics, environmental science, biotechnology - Try to confer some awareness of the impact on our lives an ...
Crossword Pazzle Across 4. the outer protein coat of a virus 5
... 7. an infectious particle made of protein rather than DNA or RNA 8. sphere-shaped bacterium 13. fype of infection in which the host cell bursts and is destroyed 14. single-celled microorganism that lacks a nucleus ...
... 7. an infectious particle made of protein rather than DNA or RNA 8. sphere-shaped bacterium 13. fype of infection in which the host cell bursts and is destroyed 14. single-celled microorganism that lacks a nucleus ...
SB3. Students will derive the relationship between single
... Viruses are infectious particles made of a _____________ shell called a capsid. Viruses are considered _______living. Viruses need a ________ cell to exist. Viruses contains either __________ or __________. The genetic material is single-stranded or double-stranded depending on the kind of ...
... Viruses are infectious particles made of a _____________ shell called a capsid. Viruses are considered _______living. Viruses need a ________ cell to exist. Viruses contains either __________ or __________. The genetic material is single-stranded or double-stranded depending on the kind of ...
Lecture 11 - AP Chem Solutions
... a. 1.0 M Li2(CO3), 1.0 M NH3, or 1.0 M LiCl All solutions are 1.0M, meaning that they all contain 1 mole of the formula unit in question per litre of water. NH3 is a covalent compound that does not dissociate into positive and negative ions; and thus, it does not conduct electricity. Li2(CO3) and Li ...
... a. 1.0 M Li2(CO3), 1.0 M NH3, or 1.0 M LiCl All solutions are 1.0M, meaning that they all contain 1 mole of the formula unit in question per litre of water. NH3 is a covalent compound that does not dissociate into positive and negative ions; and thus, it does not conduct electricity. Li2(CO3) and Li ...
TOPIC 1: CELLS 1.Homeostasis is the ability of an organism to
... 5. Organelles are the small parts that make up a cell (each has at least one specific function) a. Vacuoles-- store waste and water (large in plant cells, small in animal cells) b. Ribosome – (very small and is often represented by a dot) located on the ER or in cytoplasm. Ribosomes are where protei ...
... 5. Organelles are the small parts that make up a cell (each has at least one specific function) a. Vacuoles-- store waste and water (large in plant cells, small in animal cells) b. Ribosome – (very small and is often represented by a dot) located on the ER or in cytoplasm. Ribosomes are where protei ...
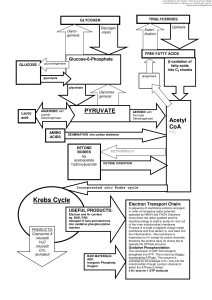
L4_bacterial metabolism7e
... • Precursor metabolites made from alphaketoglutarate and oxaloacetate ...
... • Precursor metabolites made from alphaketoglutarate and oxaloacetate ...
STUDY GUIDE –Intro to Cell Biology
... The process by which cells change to become different kinds of cells with different functions = DIFFERENTIATION The process by which organisms as a group change over time; Process by which modern organisms have descended from ancient organisms = EVOLUTION What do we call embryonic cells that have th ...
... The process by which cells change to become different kinds of cells with different functions = DIFFERENTIATION The process by which organisms as a group change over time; Process by which modern organisms have descended from ancient organisms = EVOLUTION What do we call embryonic cells that have th ...
HW_Ch1-Quiz.doc
... 6. The process of evolution involves __________. a. natural selection of organisms that produce more offspring in certain environmental conditions b. changes in a species due to mutations c. adaptation of an organism to its environment d. all of the above e. the first and third answers above ...
... 6. The process of evolution involves __________. a. natural selection of organisms that produce more offspring in certain environmental conditions b. changes in a species due to mutations c. adaptation of an organism to its environment d. all of the above e. the first and third answers above ...
Evolution of metal ions in biological systems
Evolution of Metal Ions in Biological Systems refers to the incorporation of metallic ions into living organisms and how it has changed over time. Metal ions have been associated with biological systems for billions of years, but only in the last century have scientists began to truly appreciate the scale of their influence. Major (iron, manganese, magnesium and zinc) and minor (copper, cobalt, nickel, molybdenum, tungsten) metal ions have symbiotically aligned with living organisms and the associated complexes have evolved over time.