Facts you need to know to pass the Living Environment
... 48.If the gametes each have 23 chromosomes , then what does their zygote have?_______________ 49._____________________- the process that transforms developing cells into specialized cells with different structures and functions takes place in the ____________________ in mammals. 50.Reproduction fema ...
... 48.If the gametes each have 23 chromosomes , then what does their zygote have?_______________ 49._____________________- the process that transforms developing cells into specialized cells with different structures and functions takes place in the ____________________ in mammals. 50.Reproduction fema ...
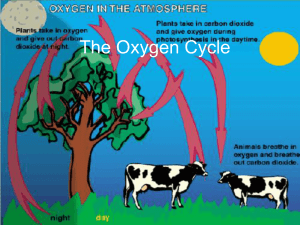
The Oxygen Cycle
... cycle begins with carbon dioxide in the atmosphere. • Animals breathe that oxygen and both plants and animals use the sugars for energy. ...
... cycle begins with carbon dioxide in the atmosphere. • Animals breathe that oxygen and both plants and animals use the sugars for energy. ...
Guided Notes sheet
... Cells can specialize and work together to form ____________________, organs, and organ systems ...
... Cells can specialize and work together to form ____________________, organs, and organ systems ...
Redox Review 9.1
... Determine the oxidation number (charge) of the ions and make sure there are enough of each to make a neutral compound ...
... Determine the oxidation number (charge) of the ions and make sure there are enough of each to make a neutral compound ...
Metabolism - California Science Teacher
... Learn the whole diagram of the control of cellular respiration , with glucose that is stimulated by AMP regulates into Fructose-6-phosphate which inhibits into pyruvate. ATP occurs which combines with Acetyl CoA , which goes in the citric acid cycle, later is the function of Oxidation phophorilation ...
... Learn the whole diagram of the control of cellular respiration , with glucose that is stimulated by AMP regulates into Fructose-6-phosphate which inhibits into pyruvate. ATP occurs which combines with Acetyl CoA , which goes in the citric acid cycle, later is the function of Oxidation phophorilation ...
Lecture Resource ()
... In each of these transformations, one of the bonds to the a-carbon of the amino acid substrate is broken in the first step of the reaction ...
... In each of these transformations, one of the bonds to the a-carbon of the amino acid substrate is broken in the first step of the reaction ...
3 limiting factors: Photosynthesis Carbon dioxide + Water à Glucose
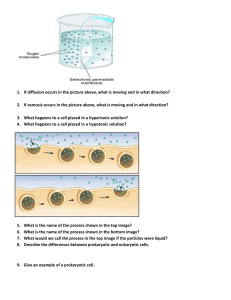
... Diffusion: Net movement from high concentration to a low concentration. ...
... Diffusion: Net movement from high concentration to a low concentration. ...
Functions of metal ions in biological systems
... charge, promote catalysis through charge stabilization or shielding negative charges. Metal ions are often much more effective catalysts than protons because can be present in high concentrations at neutral pH, and can have charges > +1. Metal ions are therefore called „superacids”. ...
... charge, promote catalysis through charge stabilization or shielding negative charges. Metal ions are often much more effective catalysts than protons because can be present in high concentrations at neutral pH, and can have charges > +1. Metal ions are therefore called „superacids”. ...
Name: #: Cellular Respiration Review 2 Process Where does it
... instead of words: 6O2 + C6H12O6 6H2O + 6CO2 + 36ATP 7. Why do we say there is a ‘net’ gain of 2 ATP at the end of glycolysis? Glycolysis produces 4ATP but since it needs 2 ATP to start, the cell only increases its amount of ATP by 2 ...
... instead of words: 6O2 + C6H12O6 6H2O + 6CO2 + 36ATP 7. Why do we say there is a ‘net’ gain of 2 ATP at the end of glycolysis? Glycolysis produces 4ATP but since it needs 2 ATP to start, the cell only increases its amount of ATP by 2 ...
Cell Unit Test Review Answer Sheet
... Attach to cell – Injection of genetic material – replication of DNA/RNA – Assembly of Proteins – Reproduce viruses – Destroys host cell – cycle repeats 13. What is the monomer of DNA? Nucleic acid 14. The enzyme catalase breaks down hydrogen peroxide into water and oxygen. During this reaction, the ...
... Attach to cell – Injection of genetic material – replication of DNA/RNA – Assembly of Proteins – Reproduce viruses – Destroys host cell – cycle repeats 13. What is the monomer of DNA? Nucleic acid 14. The enzyme catalase breaks down hydrogen peroxide into water and oxygen. During this reaction, the ...
organisms - Lyndhurst Schools
... repair damage, and reproduce Autotrophic- organisms can make their own food Heterotrophicorganisms cannot make their own food ...
... repair damage, and reproduce Autotrophic- organisms can make their own food Heterotrophicorganisms cannot make their own food ...
APES-Cycles-of
... Phosphorus is an important building block of DNA and ATP; all living things must have phosphorus P is found in rocks, and enters food webs when plants take up phosphorus found in soil Only one that doesn’t cycle through the atmosphere! (no gaseous form) P is a limiting factor for primary productivit ...
... Phosphorus is an important building block of DNA and ATP; all living things must have phosphorus P is found in rocks, and enters food webs when plants take up phosphorus found in soil Only one that doesn’t cycle through the atmosphere! (no gaseous form) P is a limiting factor for primary productivit ...
Ch2Packet - Cobb Learning
... 23. How do organisms with many cells grow? _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ 24. In addition to getting larger, what other things might happen ...
... 23. How do organisms with many cells grow? _______________________________________________________________ _______________________________________________________________ _______________________________________________________________ 24. In addition to getting larger, what other things might happen ...
Answers for Review Questions Exam 3
... 9. Electrolysis is the use of an electric current to bring about a chemical change. Reduction and oxidation both occur at the same place as in a galvanic cell, but they have different polarities, - and + respectively. It differs from a galvanic cell in that it is in the opposite direction of a galva ...
... 9. Electrolysis is the use of an electric current to bring about a chemical change. Reduction and oxidation both occur at the same place as in a galvanic cell, but they have different polarities, - and + respectively. It differs from a galvanic cell in that it is in the opposite direction of a galva ...
Answers for Review Questions Exam 3
... 10. Electrolysis is used as a source of elements from their ions. Ex. Na from Molten NaCl, Cl2 from a NaCl solution. 11. 0.1663 A current is needed. 12. First 2.47 Volts should be 2.47 Amperes. That gives 4.100g of Fe deposited. 13. Corrosion is the loss of metals to a solution of some form. The pro ...
... 10. Electrolysis is used as a source of elements from their ions. Ex. Na from Molten NaCl, Cl2 from a NaCl solution. 11. 0.1663 A current is needed. 12. First 2.47 Volts should be 2.47 Amperes. That gives 4.100g of Fe deposited. 13. Corrosion is the loss of metals to a solution of some form. The pro ...
EXAMINING THE IMPACT OF LIGAND BASICITY ON THE REACTIVITY OF TRANSITION METAL SYSTEMS THROUGH COMPUTATIONAL METHODS
... the properties and observed reactivity of transition metal complexes. Indeed, gaining the ability to “tune” the properties of metal complexes is a primary goal in inorganic and organometallic chemistry. Unfortunately, a detailed understanding of the fundamental impact of ligand basicity on a metal c ...
... the properties and observed reactivity of transition metal complexes. Indeed, gaining the ability to “tune” the properties of metal complexes is a primary goal in inorganic and organometallic chemistry. Unfortunately, a detailed understanding of the fundamental impact of ligand basicity on a metal c ...
HOMEWORK : CHAPTER 20
... the current is 37.8 A. How long (in hours) must the current run to dissolve this anode and electroplate it onto the cathode? 20.14 How would you obtain zinc from sphalerite (ZnS)? 20.16 A certain mine produces 2.0 X 108 kg of copper from chalcopyrite (CuFeS2) each year. The ore contains only 0.80 pe ...
... the current is 37.8 A. How long (in hours) must the current run to dissolve this anode and electroplate it onto the cathode? 20.14 How would you obtain zinc from sphalerite (ZnS)? 20.16 A certain mine produces 2.0 X 108 kg of copper from chalcopyrite (CuFeS2) each year. The ore contains only 0.80 pe ...
Cellular respiration is the of food
... oxygen is available, the pyruvate is broken down into ______________ or ________________ and carbon dioxide. This is called anaerobic respiration or _________________. The entire process occurs in the ...
... oxygen is available, the pyruvate is broken down into ______________ or ________________ and carbon dioxide. This is called anaerobic respiration or _________________. The entire process occurs in the ...
TAKS Review - Greenslime
... structures and functions of living organisms and their interdependence on each other and ...
... structures and functions of living organisms and their interdependence on each other and ...
Results in new DNA
... c. Natural selection selects for the homozygous individual for normal alleles. d. Natural selection selects against the heterozygous individual. ...
... c. Natural selection selects for the homozygous individual for normal alleles. d. Natural selection selects against the heterozygous individual. ...
Document
... 1. Determine if the compound is binary or ternary. 2. If the compound is binary, write out the symbols for the two elements present: metal then non-metal. * Elements with lower electronegativity values are usually listed first in a chemical formula. 3. Find and write out the oxidation number for the ...
... 1. Determine if the compound is binary or ternary. 2. If the compound is binary, write out the symbols for the two elements present: metal then non-metal. * Elements with lower electronegativity values are usually listed first in a chemical formula. 3. Find and write out the oxidation number for the ...
Evolution of metal ions in biological systems
Evolution of Metal Ions in Biological Systems refers to the incorporation of metallic ions into living organisms and how it has changed over time. Metal ions have been associated with biological systems for billions of years, but only in the last century have scientists began to truly appreciate the scale of their influence. Major (iron, manganese, magnesium and zinc) and minor (copper, cobalt, nickel, molybdenum, tungsten) metal ions have symbiotically aligned with living organisms and the associated complexes have evolved over time.