Bonding Notes
... 2) what types of bonds hold these atoms together (ionic or covalent) Why do atoms form bonds? This is a topic that we will expand on greatly as we progress through grade 11 and 12 chemistry. For our purposes, we can understand bonding as a consequence of the maximum stability that atoms have when th ...
... 2) what types of bonds hold these atoms together (ionic or covalent) Why do atoms form bonds? This is a topic that we will expand on greatly as we progress through grade 11 and 12 chemistry. For our purposes, we can understand bonding as a consequence of the maximum stability that atoms have when th ...
- Department of Chemistry, York University
... 3b. Synthesis of benzene, PAHs, and … C+ + C3H C4+ + H C4+ + H2 C4H2+ + H C4H2+ + H C4H3+ + h C4H3+ + C2H2 (or + C2H3) C6H5+ + h (or + H) C6H5+ + H2 C6H7+ + h C6H7+ + e C6H6 + H Fe(C2H2)2+ + C2H2 FeC6H6+ + h ...
... 3b. Synthesis of benzene, PAHs, and … C+ + C3H C4+ + H C4+ + H2 C4H2+ + H C4H2+ + H C4H3+ + h C4H3+ + C2H2 (or + C2H3) C6H5+ + h (or + H) C6H5+ + H2 C6H7+ + h C6H7+ + e C6H6 + H Fe(C2H2)2+ + C2H2 FeC6H6+ + h ...
Chapter 2 cont’
... called anions (Cl-) when atoms lose electrons, they become positively charged ions, called cations (Na+) ions behave much differently than the neutral atom ◦ e.g., The metal sodium, made of neutral Na atoms, is highly reactive and quite unstable. However, the sodium cations, Na+, found in table salt ...
... called anions (Cl-) when atoms lose electrons, they become positively charged ions, called cations (Na+) ions behave much differently than the neutral atom ◦ e.g., The metal sodium, made of neutral Na atoms, is highly reactive and quite unstable. However, the sodium cations, Na+, found in table salt ...
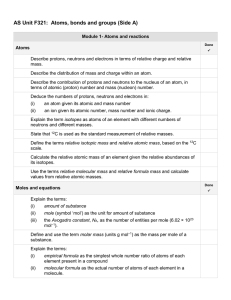
AS Unit F321 Unit 1 Side A check list
... ease of forming negative ions, in terms of atomic size, shielding and nuclear attraction. This may have been met in the other half (check) Describe the term disproportionation as a reaction in which a substance is simultaneously oxidised and reduced, illustrated by: (i) ...
... ease of forming negative ions, in terms of atomic size, shielding and nuclear attraction. This may have been met in the other half (check) Describe the term disproportionation as a reaction in which a substance is simultaneously oxidised and reduced, illustrated by: (i) ...
mass Spectrometry (mS)
... NOTE: Because the positive ion formed has an unpaired electron it is sometimes shown with a dot indicating that it is a free radical, e.g. CH3+• The positive ions are then ACCELERATED by an electric field and focused into a fine beam by passing through a series of slits with increasing negative pote ...
... NOTE: Because the positive ion formed has an unpaired electron it is sometimes shown with a dot indicating that it is a free radical, e.g. CH3+• The positive ions are then ACCELERATED by an electric field and focused into a fine beam by passing through a series of slits with increasing negative pote ...
Name
... (b)Given the photoelectron spectra below for phosphorus, P, and sulfur, S, which of the following best explains why the 2p peak for S is further to the left than the 2p peak for P, but the 3p peak for S is further to the right than the 3p peak for P? Circle your answer. I. S has a greater effective ...
... (b)Given the photoelectron spectra below for phosphorus, P, and sulfur, S, which of the following best explains why the 2p peak for S is further to the left than the 2p peak for P, but the 3p peak for S is further to the right than the 3p peak for P? Circle your answer. I. S has a greater effective ...
Determination of electrochemical equivalent of copper and
... If 0,8 < α < 1 the electrolyte is called strong and it means that almost all dissolved particles were dissociated. For middle electrolytes α = 0,5 and the number of particles dissociated and not dissociated is similar. When α < 0,01 the electrolyte is a weak one. In the practice we will use copper s ...
... If 0,8 < α < 1 the electrolyte is called strong and it means that almost all dissolved particles were dissociated. For middle electrolytes α = 0,5 and the number of particles dissociated and not dissociated is similar. When α < 0,01 the electrolyte is a weak one. In the practice we will use copper s ...
Intro to Soln Stoich
... Solutions with a large number of ions present in solution are excellent conductors of electricity ...
... Solutions with a large number of ions present in solution are excellent conductors of electricity ...
Vocabulary Notes
... A mixture of two or more metals or metals and non-metals that result in the mixture having different properties than their component elements. Ex. Stainless Steel. ...
... A mixture of two or more metals or metals and non-metals that result in the mixture having different properties than their component elements. Ex. Stainless Steel. ...
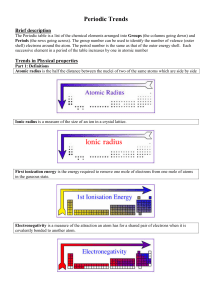
Topic 3 Periodicity notes SL - Chemical Minds
... i) there is a decrease in the electrostatic attraction between the positive protons in the nucleus and the negative electrons in the electron shells orbiting the nucleus and ii) the valence electrons are shielded by the inner electron shells Shielding means that there is an increase in electron-elec ...
... i) there is a decrease in the electrostatic attraction between the positive protons in the nucleus and the negative electrons in the electron shells orbiting the nucleus and ii) the valence electrons are shielded by the inner electron shells Shielding means that there is an increase in electron-elec ...
(activity) of hydrogen ions
... indicator added. (2-3) The acid is titrated with the alkali in the burette until the indicator turns green and the volume of alkali noted. (1-3) are repeated with both known volumes mixed together BUT without the contaminating indicator. (4) The solution is transferred to an evaporating dish and hea ...
... indicator added. (2-3) The acid is titrated with the alkali in the burette until the indicator turns green and the volume of alkali noted. (1-3) are repeated with both known volumes mixed together BUT without the contaminating indicator. (4) The solution is transferred to an evaporating dish and hea ...
Solution - ZOMUedu
... Molarity (M) = Moles of solute / Liters of solution ■ 1 M = 1 mol solute/ liter solution ○ Dilution = adding more solvent to a known solution ■ The moles of solute stay the same ■ M1V1 = M2V2 ○ Stock solution = a solution of known concentration that is used to make more dilute solutions Precipita ...
... Molarity (M) = Moles of solute / Liters of solution ■ 1 M = 1 mol solute/ liter solution ○ Dilution = adding more solvent to a known solution ■ The moles of solute stay the same ■ M1V1 = M2V2 ○ Stock solution = a solution of known concentration that is used to make more dilute solutions Precipita ...
Multiphoton ionization of inner-valence electrons and fragmentation
... For the MPI of ethylene the value of gcr is calculated to be ; 3.05. Our experimental result on the MPI of rare gas atoms w13x has shown that the criterion expressed by Eq. Ž8. is too strong and in practice the criterion g - gcr is good enough for real experiments. It is impossible to give a more pr ...
... For the MPI of ethylene the value of gcr is calculated to be ; 3.05. Our experimental result on the MPI of rare gas atoms w13x has shown that the criterion expressed by Eq. Ž8. is too strong and in practice the criterion g - gcr is good enough for real experiments. It is impossible to give a more pr ...
Learning Goals - Issaquah Connect
... Go to the list of Phet HTML5 Chemistry simulations. Click on the Build an Atom simulation and start the sim. Once the simulation opens, click on “Atom”. a. Click on the X’s behind the Net Charge and Mass Number titles to display the graphics. Add protons, neutrons & electrons to the model until you ...
... Go to the list of Phet HTML5 Chemistry simulations. Click on the Build an Atom simulation and start the sim. Once the simulation opens, click on “Atom”. a. Click on the X’s behind the Net Charge and Mass Number titles to display the graphics. Add protons, neutrons & electrons to the model until you ...
Chemical Formulas
... single atom Na+ or S2- usually tell by column on periodic table, some elements have more than one oxidation number or charge Binary compounds- only 2 elements in the compound Na2S Polyatomic ions - ions formed from more than one type of atom covalently bonded together OHPO43- NH4+ ...
... single atom Na+ or S2- usually tell by column on periodic table, some elements have more than one oxidation number or charge Binary compounds- only 2 elements in the compound Na2S Polyatomic ions - ions formed from more than one type of atom covalently bonded together OHPO43- NH4+ ...
Chemical Bonding
... » Ionic Bonding – Metal with non-metal » Covalent Bonding – Non-metal with non-metal Weak Bonds » Dipole-dipole » Hydrogen bonding » Dispersion forces ...
... » Ionic Bonding – Metal with non-metal » Covalent Bonding – Non-metal with non-metal Weak Bonds » Dipole-dipole » Hydrogen bonding » Dispersion forces ...
Chemical Bonding
... » Ionic Bonding – Metal with non-metal » Covalent Bonding – Non-metal with non-metal Weak Bonds » Dipole-dipole » Hydrogen bonding » Dispersion forces ...
... » Ionic Bonding – Metal with non-metal » Covalent Bonding – Non-metal with non-metal Weak Bonds » Dipole-dipole » Hydrogen bonding » Dispersion forces ...
Formulas of Compounds
... 1. Now we can use names and formulas of cations and anions to write formulas of compounds. 2. In these formulas, the sum of the total cation and anion charges must be zero. 3. We use subscripts to balance the charges. 4. In most cases, we write the cation first and then the anion. (Positive first). ...
... 1. Now we can use names and formulas of cations and anions to write formulas of compounds. 2. In these formulas, the sum of the total cation and anion charges must be zero. 3. We use subscripts to balance the charges. 4. In most cases, we write the cation first and then the anion. (Positive first). ...
Bonding
... difference between sets of d orbitals is comparable to the energy of visible light. In zinc ions, all the d orbitals are paired and all the orbitals are degenerate. (d) Arsenic atoms have one more valence electron than silicon atoms and can lose an electron to form As+ ions which can occupy some of ...
... difference between sets of d orbitals is comparable to the energy of visible light. In zinc ions, all the d orbitals are paired and all the orbitals are degenerate. (d) Arsenic atoms have one more valence electron than silicon atoms and can lose an electron to form As+ ions which can occupy some of ...
Chem 1st Sem Rev Ch2
... c. father of the modern atomic theory, everything made of atoms d. planetary model of the atom, electrons move around the nucleus like planets around sun. e. plum pudding model of the atom: atom looks like chocolate chip cookie f. gold foil experiment – atoms have a dense core called nucleus g. he g ...
... c. father of the modern atomic theory, everything made of atoms d. planetary model of the atom, electrons move around the nucleus like planets around sun. e. plum pudding model of the atom: atom looks like chocolate chip cookie f. gold foil experiment – atoms have a dense core called nucleus g. he g ...
Slajd 1 - Uniwersytet Warszawski
... 2) using MS method applying ‘soft’ ionization techniques APCI and ESI, which allow samples to be analysed in a liquid mobile phase, samples of verapamil and calcium-verapamil complex solutions in methanol were studied. The following values were obtained: ∆H = -36.27 kcal/mol and ∆G = -32.42 kcal/mol ...
... 2) using MS method applying ‘soft’ ionization techniques APCI and ESI, which allow samples to be analysed in a liquid mobile phase, samples of verapamil and calcium-verapamil complex solutions in methanol were studied. The following values were obtained: ∆H = -36.27 kcal/mol and ∆G = -32.42 kcal/mol ...
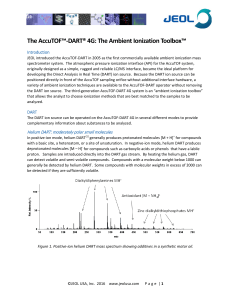
The Ambient Ionization Toolbox
... R. B. Cody , A. J. Dane. Dopant-assisted direct analysis in real time mass spectrometrywith argon gas. Rapid Communications in Mass Spectrometry, 2016, 30. A. J. Dane, R. B. Cody. Selective ionization of melamine in powdered milk by using argon direct analysis in real time (DART) mass spectrometry. ...
... R. B. Cody , A. J. Dane. Dopant-assisted direct analysis in real time mass spectrometrywith argon gas. Rapid Communications in Mass Spectrometry, 2016, 30. A. J. Dane, R. B. Cody. Selective ionization of melamine in powdered milk by using argon direct analysis in real time (DART) mass spectrometry. ...
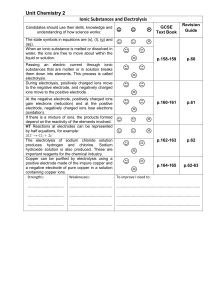
Unit_Chemistry_2_Ionic_Substances_and_Electrolysis
... The electrolysis of sodium chloride solution produces hydrogen and chlorine. Sodium hydroxide solution is also produced. These are important reagents for the chemical industry. Copper can be purified by electrolysis using a positive electrode made of the impure copper and a negative electrode of pur ...
... The electrolysis of sodium chloride solution produces hydrogen and chlorine. Sodium hydroxide solution is also produced. These are important reagents for the chemical industry. Copper can be purified by electrolysis using a positive electrode made of the impure copper and a negative electrode of pur ...
2.4 Revision 1: There were two atoms. One got hit by an extremely
... c. Conducts electricity when solid and melted? d. Conducts electricity when dissolved or melted but not as a solid? e. Forces that hold non-polar molecular solids together. f. Can conduct heat and electrical charge. g. Which of the following solids have hydrogen bonds between molecules; hydrogen chl ...
... c. Conducts electricity when solid and melted? d. Conducts electricity when dissolved or melted but not as a solid? e. Forces that hold non-polar molecular solids together. f. Can conduct heat and electrical charge. g. Which of the following solids have hydrogen bonds between molecules; hydrogen chl ...
Ion source

An ion source is a device that creates atomic and molecular ions. Ion sources are used to form ions for mass spectrometers, optical emission spectrometers, particle accelerators, ion implanters and ion engines.