* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chemical Bonding
Aromaticity wikipedia , lookup
Photoelectric effect wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Cluster chemistry wikipedia , lookup
Auger electron spectroscopy wikipedia , lookup
Heat transfer physics wikipedia , lookup
2-Norbornyl cation wikipedia , lookup
Ionic compound wikipedia , lookup
Electrochemistry wikipedia , lookup
Surface properties of transition metal oxides wikipedia , lookup
Atomic orbital wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Electron scattering wikipedia , lookup
Homoaromaticity wikipedia , lookup
Electron configuration wikipedia , lookup
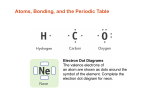
9 Sci - Chemistry Where is water? A chemical bond is an attraction between atoms. Atoms are held together by the interaction of their outer shells (valence electrons) Atoms can join together to form larger substances . Chemical bonds can be either ‘strong’ or ‘weak’ Strong Bonds » Metallic Bonding – Metal with metal » Ionic Bonding – Metal with non-metal » Covalent Bonding – Non-metal with non-metal Weak Bonds » Dipole-dipole » Hydrogen bonding » Dispersion forces An ionic bond results from the complete transfer of one or more electrons from a metal to a nonmetal. Atoms of different elements can join together to form new substances. A substance which is made up of two or more different types of atoms is known as a compound. One way this can occur is for atoms to form ions. What is it? NON-METAL METAL Metals usually want to give electrons (electron doners) » Becomes positively charged ion (cation) Non-Metals usually want to take electrons (electron receivers) » Becomes negatively charged ion (anion) » Changes name suffix to –ide (e.g.ChlorineChloride) The metal will donate the electron to the non-metal The cation and anion are opposite in charge and become attracted to each other (electrostatic forces) Example: Sodium Chloride - NaCl Example 2: Calcium Chloride + = X2 Calcium Atom Atomic Number = 20 Valence Electron = 2 Calcium gives 2 electrons to become stable Chlorine Atom Atomic Number = 17 Valence Electron = 1 Chlorine receives 1 electron to become stable Calcium Cation = Ca2+ Chloride Anion = Cl- Calcium joins with 2 Chloride ions to become: Calcium Chloride = CaCl2 Table showing Valency of Ions