Chemical Equations TrackStar Assignment
... 2. What is a reversible reaction and how is it indicated? 3. Write the reaction for a silver spoon tarnishing. What type of reaction is this? 4. Write the reaction for the burning of Methane gas (the gas used in Chemistry lab). What type of reaction is this? 5. Write the reaction of the neutralizati ...
... 2. What is a reversible reaction and how is it indicated? 3. Write the reaction for a silver spoon tarnishing. What type of reaction is this? 4. Write the reaction for the burning of Methane gas (the gas used in Chemistry lab). What type of reaction is this? 5. Write the reaction of the neutralizati ...
Chemical Reactions
... The other reactant will have some unchanged so it is said to be the excess reactant. For example, if you need to make 10 chicken sandwiches. You have 10 slices of bread and 10 pieces of chicken. If each sandwich requires 2 slices of bread and 1 piece of chicken, which is the limiting reactant? Exces ...
... The other reactant will have some unchanged so it is said to be the excess reactant. For example, if you need to make 10 chicken sandwiches. You have 10 slices of bread and 10 pieces of chicken. If each sandwich requires 2 slices of bread and 1 piece of chicken, which is the limiting reactant? Exces ...
Chemistry FINAL: CONTENT Review Packet
... _______________________is made from two or more substances that are physically combined ______________________________ are substances that are made up of only one type of atom _________________________________ is anything that has both mass and volume _____________________________________is a solid, ...
... _______________________is made from two or more substances that are physically combined ______________________________ are substances that are made up of only one type of atom _________________________________ is anything that has both mass and volume _____________________________________is a solid, ...
2005/6 - SAASTA
... electrode at which oxidation occurs and the cathode (+ve electrode) is the electrode at which reduction occurs. So, if the negative terminal is the electrode with greater concentration of electrons, then it serves as the anode in a voltaic cell and a cathode in an electrolytic cell. Conversely, the ...
... electrode at which oxidation occurs and the cathode (+ve electrode) is the electrode at which reduction occurs. So, if the negative terminal is the electrode with greater concentration of electrons, then it serves as the anode in a voltaic cell and a cathode in an electrolytic cell. Conversely, the ...
Chemistry Fall Final Review 2012-2013 Alchemy Unit
... Ions are atoms that have lost or gain electrons and become positive or negative charged. Cation is a positive ion and an anion is a negative ion. 12. In ionic bonds, metals tend to lose electrons and nonmetals gain electrons. What happens to these elements to achieve noble electron configuration? Wh ...
... Ions are atoms that have lost or gain electrons and become positive or negative charged. Cation is a positive ion and an anion is a negative ion. 12. In ionic bonds, metals tend to lose electrons and nonmetals gain electrons. What happens to these elements to achieve noble electron configuration? Wh ...
Name
... identify substances and determine the number of atoms of each element. Ⓡ 8.5 (E) Chemical Reactions: Students will be able to investigate how evidences of chemical reactions indicate that new substances are formed. Ⓡ 8.5 (F) Balancing Equations: Students will be able to recognize whether or not a ch ...
... identify substances and determine the number of atoms of each element. Ⓡ 8.5 (E) Chemical Reactions: Students will be able to investigate how evidences of chemical reactions indicate that new substances are formed. Ⓡ 8.5 (F) Balancing Equations: Students will be able to recognize whether or not a ch ...
Chapter 2 Practice Questions
... A) Elements are made up of tiny particles called atoms. B) Atoms are not created or destroyed in chemical reactions. C) All atoms of a given element are identical. D) Atoms are indivisible in chemical reactions. E) All of these statements are true according to modern atomic theory. 4. Avogadro's hyp ...
... A) Elements are made up of tiny particles called atoms. B) Atoms are not created or destroyed in chemical reactions. C) All atoms of a given element are identical. D) Atoms are indivisible in chemical reactions. E) All of these statements are true according to modern atomic theory. 4. Avogadro's hyp ...
Answers to Final Exam Review
... 49. Write a balanced chemical equation for each reaction (include phases). Identify the type of reaction. a. Sodium metal dropped into a beaker of water. Na (s) + 2H2O (l) Na+ (aq) + 2OH- (aq) + H2 (g) b. Silver nitrate is added to sodium chloride. AgNO3 (s) + NaCl (s) AgCl (s) + NaNO3 (s) (doubl ...
... 49. Write a balanced chemical equation for each reaction (include phases). Identify the type of reaction. a. Sodium metal dropped into a beaker of water. Na (s) + 2H2O (l) Na+ (aq) + 2OH- (aq) + H2 (g) b. Silver nitrate is added to sodium chloride. AgNO3 (s) + NaCl (s) AgCl (s) + NaNO3 (s) (doubl ...
Normality Primer
... To know how many electrons are gained or lost, redox reactions require knowledge of the half reaction associated with the reducing or oxidizing agent. These reactions are not easily predictable and generally chemists look them up in tables of reduction potentials. However, one must be particula ...
... To know how many electrons are gained or lost, redox reactions require knowledge of the half reaction associated with the reducing or oxidizing agent. These reactions are not easily predictable and generally chemists look them up in tables of reduction potentials. However, one must be particula ...
Stoichiometry
... Two compounds are involved with the cation of one compound EXCHANGING with the cation of another compound. AX + BZ AZ + BX These reactions proceed if one of the ff. is satisfied: 1. An insoluble/slightly soluble product is formed (PRECIPITATE formation) 2. A weakly ionized species is produced. The ...
... Two compounds are involved with the cation of one compound EXCHANGING with the cation of another compound. AX + BZ AZ + BX These reactions proceed if one of the ff. is satisfied: 1. An insoluble/slightly soluble product is formed (PRECIPITATE formation) 2. A weakly ionized species is produced. The ...
Chemistry EOC Review Name
... a. dihydrogen monoxide b. tetrasulfur hexachloride c. nitrogen dioxide 64. List two differences between ionic and covalent compounds. Unit 10 (Chapter 11): 65. What do the following symbols mean: a. b. (aq) c. (g) d. (s) e. 66. Why must equations be balanced? 67. Balanced the following equations ...
... a. dihydrogen monoxide b. tetrasulfur hexachloride c. nitrogen dioxide 64. List two differences between ionic and covalent compounds. Unit 10 (Chapter 11): 65. What do the following symbols mean: a. b. (aq) c. (g) d. (s) e. 66. Why must equations be balanced? 67. Balanced the following equations ...
Chapter 11 Chemical Reactions
... For some, we will be able to: c) predict whether or not they will happen at all. ...
... For some, we will be able to: c) predict whether or not they will happen at all. ...
Slide 1
... H2O aqua The oxidation number of a metal that exhibits variable oxidation states is designated by a Roman numeral in parentheses following the name of the complex ion or molecule. If a complex is an anion, the suffix "ate" ends the name. No suffix is used in the case of a neutral or cationic compl ...
... H2O aqua The oxidation number of a metal that exhibits variable oxidation states is designated by a Roman numeral in parentheses following the name of the complex ion or molecule. If a complex is an anion, the suffix "ate" ends the name. No suffix is used in the case of a neutral or cationic compl ...
Document
... oxide. The unbalanced equation for this reaction is shown above. What are the coefficients when this equation is balanced? F 2,2,1 G 2,3,1 H 4,3,2 J 4,2,2 ...
... oxide. The unbalanced equation for this reaction is shown above. What are the coefficients when this equation is balanced? F 2,2,1 G 2,3,1 H 4,3,2 J 4,2,2 ...
Chem 30A Fa_06 FE Review
... The reaction between ammonia and carbon dioxide forms urea, CH4N2O(s), according to the following equation: 2NH3(g) + CO2(g) CH4N2O(s) + H2O(l) If 75.0 g of NH3 is reacted with 92.5 g of CO2, how many grams of urea are formed? If 115 g of urea is actually obtained, what is the percent yield? (Hint ...
... The reaction between ammonia and carbon dioxide forms urea, CH4N2O(s), according to the following equation: 2NH3(g) + CO2(g) CH4N2O(s) + H2O(l) If 75.0 g of NH3 is reacted with 92.5 g of CO2, how many grams of urea are formed? If 115 g of urea is actually obtained, what is the percent yield? (Hint ...
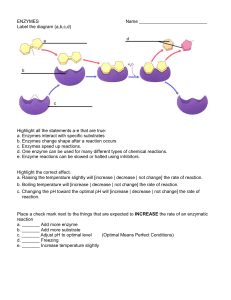
ENZYMES
... a. _______ Add more enzyme b. _______ Add more substrate c. _______ Adjust pH to optimal level (Optimal Means Perfect Conditions) d. _______ Freezing e. _______ Increase temperature slightly ...
... a. _______ Add more enzyme b. _______ Add more substrate c. _______ Adjust pH to optimal level (Optimal Means Perfect Conditions) d. _______ Freezing e. _______ Increase temperature slightly ...
Chemistry Final Exam Test Yourself I
... Predict what will happen to the rate of a reaction when you double the concentration of a reactant whose “x” has the following values: 0, 1, 2 ...
... Predict what will happen to the rate of a reaction when you double the concentration of a reactant whose “x” has the following values: 0, 1, 2 ...
File - Science With BLT
... ____ 23. A covalent bond results when ____ are shared. a. ions c. electrons b. Lewis structures d. dipoles ____ 24. Nonpolar covalent bonds are not common because a. one atom usually attracts electrons more strongly than the other. b. ions always form when atoms join. c. the electrons usually remain ...
... ____ 23. A covalent bond results when ____ are shared. a. ions c. electrons b. Lewis structures d. dipoles ____ 24. Nonpolar covalent bonds are not common because a. one atom usually attracts electrons more strongly than the other. b. ions always form when atoms join. c. the electrons usually remain ...
Atomic number
... a measure of how much solute will dissolve into the solvent Temperature will affect solubility- the solubility will be INCREASED as the temperature is increased. Stirring will increase solubility. Molecular or Particle Size will affect the solubility. The larger the molecule or particle the less sol ...
... a measure of how much solute will dissolve into the solvent Temperature will affect solubility- the solubility will be INCREASED as the temperature is increased. Stirring will increase solubility. Molecular or Particle Size will affect the solubility. The larger the molecule or particle the less sol ...
Chapter 11 Chemical Reactions
... For some, we will be able to: c) predict whether or not they will happen at all. ...
... For some, we will be able to: c) predict whether or not they will happen at all. ...
HIGHER TIER CHEMISTRY MINI-MOCK UNIT 2
... Which two sub-atomic particles are in the nucleus of an atom? ........................................................... and ............................................... ...
... Which two sub-atomic particles are in the nucleus of an atom? ........................................................... and ............................................... ...
Chemical Equation
... the relative relationship (ratio) between reactants and products in a chemical reaction. Coefficients of a balanced chemical equation gives three pieces of quantitative information about the reactants and the products. 1. The relative number of particles. 2. The relative number of moles. 3. The rela ...
... the relative relationship (ratio) between reactants and products in a chemical reaction. Coefficients of a balanced chemical equation gives three pieces of quantitative information about the reactants and the products. 1. The relative number of particles. 2. The relative number of moles. 3. The rela ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.