rp oc4
... 5. Circle the lone pair electrons in the following dot formula of water. 6. With respect to bonds formed between the following pairs of atoms: • Determine the electronegativity difference. SHOW WORK! • Determine the probable bond type (ionic, polar covalent, or nonpolar covalent). • Assign partial ...
... 5. Circle the lone pair electrons in the following dot formula of water. 6. With respect to bonds formed between the following pairs of atoms: • Determine the electronegativity difference. SHOW WORK! • Determine the probable bond type (ionic, polar covalent, or nonpolar covalent). • Assign partial ...
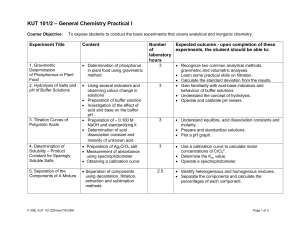
KUT 101/2 – General Chemistry Practical I
... • Preparation of Na2S2O3 solution and standardizing it • Determination of the oxidizing capacity of an unknown liquid bleach • Preparation of Cu(NO)3 and performing basic laboratory procedures • Reduction of copper with zinc • Preparation of ∼ 0.100 M NaOH and standardizing it. • Analysis of an unkn ...
... • Preparation of Na2S2O3 solution and standardizing it • Determination of the oxidizing capacity of an unknown liquid bleach • Preparation of Cu(NO)3 and performing basic laboratory procedures • Reduction of copper with zinc • Preparation of ∼ 0.100 M NaOH and standardizing it. • Analysis of an unkn ...
Aeration and Hydric Soils
... takes place and probe measures degree of reduction ( mv) As organic substances are oxidized (in respiration) Eh drops as sequence of reductions (electron gains) takes place: ...
... takes place and probe measures degree of reduction ( mv) As organic substances are oxidized (in respiration) Eh drops as sequence of reductions (electron gains) takes place: ...
Chapter 10
... will occur between the following pairs of possible reactants. If so, write the balanced chemical equation for the reaction. Chlorine gas and aqueous potassium iodide Magnesium metal and aqueous copper (II) ...
... will occur between the following pairs of possible reactants. If so, write the balanced chemical equation for the reaction. Chlorine gas and aqueous potassium iodide Magnesium metal and aqueous copper (II) ...
ChemicalBondingTestAnswers
... 7. There are no attractions (bonds) between two metal atoms. False Correct statement: There are metallic bonds between two metal atoms. ...
... 7. There are no attractions (bonds) between two metal atoms. False Correct statement: There are metallic bonds between two metal atoms. ...
pblock - Chemistry Courses
... The chemistry of carbon is unique because: • It has a valence of 4 (highest in 2nd period) • It can make stable bonds with itself • It can make multiple bonds to C, N, O • The C-H bond is nonpolar, but bonds to other elements (N, O, halogens) are polar This is why life is based on the chemistry of c ...
... The chemistry of carbon is unique because: • It has a valence of 4 (highest in 2nd period) • It can make stable bonds with itself • It can make multiple bonds to C, N, O • The C-H bond is nonpolar, but bonds to other elements (N, O, halogens) are polar This is why life is based on the chemistry of c ...
Chapter 4
... Often called a neutralization reaction Because the acid neutralizes the base. Often titrate to determine concentrations. Solution of known concentration (titrant), is added to the unknown (analyte), until the equivalence point is reached where enough titrant has been added to ...
... Often called a neutralization reaction Because the acid neutralizes the base. Often titrate to determine concentrations. Solution of known concentration (titrant), is added to the unknown (analyte), until the equivalence point is reached where enough titrant has been added to ...
Chemistry EOC Review
... 131. What is the formula for molarity? (Formula provided on EOC). 132. Calculate the molarity of a solution that contains 85 g BaCl2 in 500 mL of water. 133. What is entropy? 134. What does it mean when entropy has a negative value? 135. Indicate if the following will have a positive or negative val ...
... 131. What is the formula for molarity? (Formula provided on EOC). 132. Calculate the molarity of a solution that contains 85 g BaCl2 in 500 mL of water. 133. What is entropy? 134. What does it mean when entropy has a negative value? 135. Indicate if the following will have a positive or negative val ...
A) Sn4+ → Sn2+ + 2e
... B) Anode: 2 Cl – ® Cl 2 + 2e – Cathode: 2 H 2O + 2e – ® H 2 + 2 OH – C) Anode: 2 H 2O ® O 2 + 4 H+ + 4e – Cathode: 2 Zn2+ + 4e – ® 2 Zn D) Anode: 2 H2O ® O2 + 4 H+ + 4e – Cathode: 4 H 2 O + 4e – ® 2 H 2 + 4 OH – E) Anode: Cl 2 + 2e – ® 2 Cl – Cathode: Zn ® Zn 2+ + 2e – 69. What are the anode and cat ...
... B) Anode: 2 Cl – ® Cl 2 + 2e – Cathode: 2 H 2O + 2e – ® H 2 + 2 OH – C) Anode: 2 H 2O ® O 2 + 4 H+ + 4e – Cathode: 2 Zn2+ + 4e – ® 2 Zn D) Anode: 2 H2O ® O2 + 4 H+ + 4e – Cathode: 4 H 2 O + 4e – ® 2 H 2 + 4 OH – E) Anode: Cl 2 + 2e – ® 2 Cl – Cathode: Zn ® Zn 2+ + 2e – 69. What are the anode and cat ...
2015-2016 AP CHEMISTRY MIDTERM EXAM Review
... Write a balanced chemical equation for the mentioned neutralization process. How many moles of H2SO4 are contained in the 45.7mL sample? What volume of 18M stock solution of H2SO4 is required to prepare the 45.7mL sample that is required for neutralization? e. What was the concentration of the NaOH ...
... Write a balanced chemical equation for the mentioned neutralization process. How many moles of H2SO4 are contained in the 45.7mL sample? What volume of 18M stock solution of H2SO4 is required to prepare the 45.7mL sample that is required for neutralization? e. What was the concentration of the NaOH ...
Balancing Equations
... The iron is not balanced to conserve mass. We have 1 on the left and 2 on the right, so we need to at a coefficient (small whole number that are placed in front of the formulas in an equation in order to balance it). So the equation would look like this: 2Fe + O2 Fe2O2 ...
... The iron is not balanced to conserve mass. We have 1 on the left and 2 on the right, so we need to at a coefficient (small whole number that are placed in front of the formulas in an equation in order to balance it). So the equation would look like this: 2Fe + O2 Fe2O2 ...
Test: "Chemical Equations" (General Chemistry)
... If 1 mole of Ca(OH)2 reacts with excess HCl, how many moles of CaCl2 are formed? If 2 moles of HCl react with excess Ca(OH)2, how many moles of CaCl2 are formed? If 4 moles of Ca(OH)2 react with excess HCl, how many moles of H2O are formed? If 4 moles of HCl react with excess Ca(OH)2, how many moles ...
... If 1 mole of Ca(OH)2 reacts with excess HCl, how many moles of CaCl2 are formed? If 2 moles of HCl react with excess Ca(OH)2, how many moles of CaCl2 are formed? If 4 moles of Ca(OH)2 react with excess HCl, how many moles of H2O are formed? If 4 moles of HCl react with excess Ca(OH)2, how many moles ...
Academic Chemistry Final Exam Review
... a. When solid ammonium nitrate is heated, it produces nitrogen gas, oxygen gas, and water vapor. ...
... a. When solid ammonium nitrate is heated, it produces nitrogen gas, oxygen gas, and water vapor. ...
Chem12 SM Unit 5 Review final ok
... 42. (a) In P2O5, the oxidation number of O is –2 and the oxidation number of P is +5. (b) In NO2, the oxidation number of O is –2 and the oxidation number of N is +4. (c) In Na2SO4, the oxidation number of Na is +1, the oxidation number of O is –2, and the oxidation number of S is +6. (d) In Cu(NO3) ...
... 42. (a) In P2O5, the oxidation number of O is –2 and the oxidation number of P is +5. (b) In NO2, the oxidation number of O is –2 and the oxidation number of N is +4. (c) In Na2SO4, the oxidation number of Na is +1, the oxidation number of O is –2, and the oxidation number of S is +6. (d) In Cu(NO3) ...
Atoms, Ions, and Molecules File
... • Atoms of a given element are all the same. Atoms of different elements are different. • Atoms are not changed into different atoms in a chemical reaction. • Compounds are formed when atoms of two or more elements combine. ...
... • Atoms of a given element are all the same. Atoms of different elements are different. • Atoms are not changed into different atoms in a chemical reaction. • Compounds are formed when atoms of two or more elements combine. ...
CHM 2045C - State College of Florida
... Examine, sketch, and apply the basic aspects of the valence bond and molecular orbital approaches to chemical bonding, including the Lewis and valence shell electron pair repulsion (VSEPR) method of predicting molecular geometry. ...
... Examine, sketch, and apply the basic aspects of the valence bond and molecular orbital approaches to chemical bonding, including the Lewis and valence shell electron pair repulsion (VSEPR) method of predicting molecular geometry. ...
Chapter 8 Section 1 Describing Chemical Reactions
... CH3CH2OH + 3O2 2CO2 + 3H2O Synthesis Reactions • In a synthesis reaction a single compound forms from two or more reactants. • Two elements form a binary compound C + O2 CO2 2C + O2 2CO • Two compounds form a ternary compound CaO(s) + H2O(l) Ca(OH)2(s) CO2(g) + H2O(l) H2CO3(aq) Decompositio ...
... CH3CH2OH + 3O2 2CO2 + 3H2O Synthesis Reactions • In a synthesis reaction a single compound forms from two or more reactants. • Two elements form a binary compound C + O2 CO2 2C + O2 2CO • Two compounds form a ternary compound CaO(s) + H2O(l) Ca(OH)2(s) CO2(g) + H2O(l) H2CO3(aq) Decompositio ...
8th Grade Ch. 7 Chemical Reactions Study guide
... ____ 31. Each substance written to the right of the arrow in a chemical equation is a ____. A. reactant B. product C. precipitate D. catalyst ____ 32. According to the law of conservation of mass, how does the mass of the products in a chemical reaction compare to the mass of the reactants? A. There ...
... ____ 31. Each substance written to the right of the arrow in a chemical equation is a ____. A. reactant B. product C. precipitate D. catalyst ____ 32. According to the law of conservation of mass, how does the mass of the products in a chemical reaction compare to the mass of the reactants? A. There ...
6.7 – Ionic Compounds
... and Group 3A will tend to lose 3 valence electrons and become 3+ (Al3+). Transition metals will often have different charges. Anion – A nonmetal that has gained valence electrons, and is a negatively charged ion. Halogens will gain 1 valence electron and become - (F-), the oxygen family will tend to ...
... and Group 3A will tend to lose 3 valence electrons and become 3+ (Al3+). Transition metals will often have different charges. Anion – A nonmetal that has gained valence electrons, and is a negatively charged ion. Halogens will gain 1 valence electron and become - (F-), the oxygen family will tend to ...
chemistry
... The last page of the booklet is the answer sheet. Fold the last page along the perforations and, slowly and carefully, tear off the answer sheet. Then fill in the heading of your answer sheet. All of your answers are to be recorded on the separate answer sheet. For each question, decide which of the ...
... The last page of the booklet is the answer sheet. Fold the last page along the perforations and, slowly and carefully, tear off the answer sheet. Then fill in the heading of your answer sheet. All of your answers are to be recorded on the separate answer sheet. For each question, decide which of the ...
Types of Chemical Reactions Name_________________________
... How does the aluminum bromide precipitate become deposited on the watch glass covering the beaker since it does not come in contact with the bromine liquid? ...
... How does the aluminum bromide precipitate become deposited on the watch glass covering the beaker since it does not come in contact with the bromine liquid? ...
Unit 9 The p-Block Elements
... out the highest oxidation states of other elements. Reactions with iron (II) ion The reaction between fluorine and aqueous solution of Fe2+ is unsuitable to carry out because water is oxidized to oxygen and HF gas : 2 F2(g) + 2 H2O(l) → 4 HF(aq) + O2(g) (Traces of H2O2 and ozone are also formed.) Ch ...
... out the highest oxidation states of other elements. Reactions with iron (II) ion The reaction between fluorine and aqueous solution of Fe2+ is unsuitable to carry out because water is oxidized to oxygen and HF gas : 2 F2(g) + 2 H2O(l) → 4 HF(aq) + O2(g) (Traces of H2O2 and ozone are also formed.) Ch ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.