09 Stoichiometry WS Stoichiometry WS
... If 1.23 g of lead nitrate are consumed, what is the mass of the potassium nitrate produced? 10. A car battery produces electrical energy with the following chemical reaction: Pb(s) + PbO2(s) + 2H2SO4(aq) 2PbSO4(s) + 2H2O(l) If the battery loses 340. g of lead in this reaction, how many moles of lead ...
... If 1.23 g of lead nitrate are consumed, what is the mass of the potassium nitrate produced? 10. A car battery produces electrical energy with the following chemical reaction: Pb(s) + PbO2(s) + 2H2SO4(aq) 2PbSO4(s) + 2H2O(l) If the battery loses 340. g of lead in this reaction, how many moles of lead ...
Elements, Compounds and Chemical Reactions
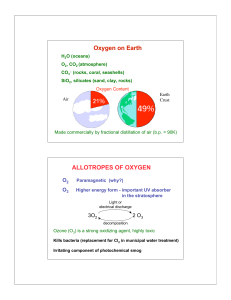
... The Earth is Made of Elements • From these last two pie charts, you should be able to see that our air is mostly Nitrogen, then oxygen, our crust is mostly oxygen and then silicon, and our bodies are oxygen and ...
... The Earth is Made of Elements • From these last two pie charts, you should be able to see that our air is mostly Nitrogen, then oxygen, our crust is mostly oxygen and then silicon, and our bodies are oxygen and ...
+ H 2 O
... Electrolytes and Nonelectrolytes Electrolytes- compounds that conduct an electric current in aqueous solution, or in the molten state – all ionic compounds are electrolytes because they dissociate into ions (they are also called “salts”) barium sulfate- will conduct when molten, but is insoluble ...
... Electrolytes and Nonelectrolytes Electrolytes- compounds that conduct an electric current in aqueous solution, or in the molten state – all ionic compounds are electrolytes because they dissociate into ions (they are also called “salts”) barium sulfate- will conduct when molten, but is insoluble ...
CHEMISTRY
... (4) Decrease the concentration of OH- ions 31. The catalyst used in Ziegler process ...
... (4) Decrease the concentration of OH- ions 31. The catalyst used in Ziegler process ...
power point notes
... that the differences in substances were the direct result of differences in the size of tiny uncuttable particles. b. During the 4th century B.C. Artistole said NO WAY! He thought that only 4 elements actually exist: water, air, fire and earth ...
... that the differences in substances were the direct result of differences in the size of tiny uncuttable particles. b. During the 4th century B.C. Artistole said NO WAY! He thought that only 4 elements actually exist: water, air, fire and earth ...
S2-2-07 - Classifying Chemical Reactions
... At the front of the class, add approximately ½ mL of CuSO4 solution to a test tube. Drop a piece of zinc into the test tube. Tell students that what is happening in the test tube is called a Single Displacement reaction. Explain that this type of reaction (using general formula) involves one reactan ...
... At the front of the class, add approximately ½ mL of CuSO4 solution to a test tube. Drop a piece of zinc into the test tube. Tell students that what is happening in the test tube is called a Single Displacement reaction. Explain that this type of reaction (using general formula) involves one reactan ...
Semester 1 Final Review Powerpoint
... 2. Construct a sentence or two to describe the following chemical: CO2 In your sentence use the following terms: Atom, element, compound molecule ...
... 2. Construct a sentence or two to describe the following chemical: CO2 In your sentence use the following terms: Atom, element, compound molecule ...
PRACTICE PROBLEMS EXAM 1,2 and 3 1311
... 23) There are __________ protons, __________ neutrons, and __________ electrons in 131I-. A) 131, 53, and 54 B) 131, 53, and 52 C) 53, 78, and 54 D) 53, 131, and 52 E) 78, 53, and 72 24) Which of the following compounds would you expect to be ionic? A) SF6 B) H2O C) H2O2 D) NH3 E) CaO 25) Which pair ...
... 23) There are __________ protons, __________ neutrons, and __________ electrons in 131I-. A) 131, 53, and 54 B) 131, 53, and 52 C) 53, 78, and 54 D) 53, 131, and 52 E) 78, 53, and 72 24) Which of the following compounds would you expect to be ionic? A) SF6 B) H2O C) H2O2 D) NH3 E) CaO 25) Which pair ...
Unit 3 Spiraling
... -Reduction is the gain of electrons. When an electron is added to a neutral atom it becomes an anion. It is negatively charged and has a decrease in its oxidation state. In the form of a chemical reaction, adding an electron to fluorine is written as F0 + e- F – F – is called the flouride ion ...
... -Reduction is the gain of electrons. When an electron is added to a neutral atom it becomes an anion. It is negatively charged and has a decrease in its oxidation state. In the form of a chemical reaction, adding an electron to fluorine is written as F0 + e- F – F – is called the flouride ion ...
p-Block Elements, Part 1
... Nitrogen (N2) has a triple bond N≡N (very stable molecule) But phosphorus is found in several forms (white, red, black), all of which have only single bonds. The chemistry of carbon is unique because: • It has a valence of 4 (highest in 2nd period) • It can make stable bonds with itself • It can mak ...
... Nitrogen (N2) has a triple bond N≡N (very stable molecule) But phosphorus is found in several forms (white, red, black), all of which have only single bonds. The chemistry of carbon is unique because: • It has a valence of 4 (highest in 2nd period) • It can make stable bonds with itself • It can mak ...
Chemistry Final Review 2017 1. List a set of elements
... sodium chlorate ® sodium chloride + oxygen 15. In which type of chemical reaction do two or more reactants combine to form one product, only? 16. Identify the type of reaction represented by the following: Cl2 + NaBr--->NaCl +Br2 NaCl ---> Na + Cl2 AgNO3(aq) + NaCl(aq) -->NaNO3 (aq) + AgCl(s) H2(g) ...
... sodium chlorate ® sodium chloride + oxygen 15. In which type of chemical reaction do two or more reactants combine to form one product, only? 16. Identify the type of reaction represented by the following: Cl2 + NaBr--->NaCl +Br2 NaCl ---> Na + Cl2 AgNO3(aq) + NaCl(aq) -->NaNO3 (aq) + AgCl(s) H2(g) ...
word-doc Practice for the final exam!
... d. pure substance e. solid 3. If matter is uniform throughout, cannot be separated into other substances by physical processes, but can be decomposed into other substances by chemical processes, it is ________. a. a heterogeneous mixture b. an element c. a homogeneous mixture d. a compound e. a mixt ...
... d. pure substance e. solid 3. If matter is uniform throughout, cannot be separated into other substances by physical processes, but can be decomposed into other substances by chemical processes, it is ________. a. a heterogeneous mixture b. an element c. a homogeneous mixture d. a compound e. a mixt ...
SAT Practice Test 3
... HCl is a proton donor Powdered zinc has a greater surface area NH3 is a polar substance Water boils when the vapor pressure of the water is equal to the atmospheric pressure In an exothermic reaction the products have less potential energy than the reactants Pressure and volume have a direct relatio ...
... HCl is a proton donor Powdered zinc has a greater surface area NH3 is a polar substance Water boils when the vapor pressure of the water is equal to the atmospheric pressure In an exothermic reaction the products have less potential energy than the reactants Pressure and volume have a direct relatio ...
Course Syllabus - Honors Chemistry
... j.* Spectral lines are the result of transitions of electrons between energy levels and that these lines correspond to photons with a frequency related to the energy spacing between levels by using Planck’s relationship (E = hv). 2. Biological, chemical, and physical properties of matter result from ...
... j.* Spectral lines are the result of transitions of electrons between energy levels and that these lines correspond to photons with a frequency related to the energy spacing between levels by using Planck’s relationship (E = hv). 2. Biological, chemical, and physical properties of matter result from ...
Single Replacement Reactions
... In nature, elements can occur either free, meaning uncombined with other elements, or chemically combined in a compound. The tendency of a particular element to combine with other substances is a measure of the activity of the element. The more active an element is, the more likely it is to combine. ...
... In nature, elements can occur either free, meaning uncombined with other elements, or chemically combined in a compound. The tendency of a particular element to combine with other substances is a measure of the activity of the element. The more active an element is, the more likely it is to combine. ...
Chemistry 432: Final Exam Review Sheet
... 4. Phases of matter and properties of solids, liquids, and gases. (30 questions and 4 calculations) Includes: The kinetic theory, types and properties of solutions: gaseous, liquid, and solid solutions, heating curves and calculations, solubility curves; phase diagrams, concentration including perce ...
... 4. Phases of matter and properties of solids, liquids, and gases. (30 questions and 4 calculations) Includes: The kinetic theory, types and properties of solutions: gaseous, liquid, and solid solutions, heating curves and calculations, solubility curves; phase diagrams, concentration including perce ...
Chemistry-Chapter 2 Lecture Notes Page
... Chemical bonds represent STORED ENERGY - Bond formation requires energy - Energy is released when bonds are broken ...
... Chemical bonds represent STORED ENERGY - Bond formation requires energy - Energy is released when bonds are broken ...
Name
... Essential Standard 7b: Elements are defined by the number of protons in the nucleus, which is called the atomic number. Different isotopes of an element have a different number of neutrons in the nucleus. ...
... Essential Standard 7b: Elements are defined by the number of protons in the nucleus, which is called the atomic number. Different isotopes of an element have a different number of neutrons in the nucleus. ...
Chemistry Study Guide
... 6. What kind of bond is NaCl? Ionic CO2 Covalent N2 Covalent 7. Which group forms acids with H+ ion? Halogens (Group 17) 8. How many valence electrons are in a Group 1 element? 1 Group 13? 3 9. How do positive and negative ions form? Positive ions form when an atom loses an electron, negative ions f ...
... 6. What kind of bond is NaCl? Ionic CO2 Covalent N2 Covalent 7. Which group forms acids with H+ ion? Halogens (Group 17) 8. How many valence electrons are in a Group 1 element? 1 Group 13? 3 9. How do positive and negative ions form? Positive ions form when an atom loses an electron, negative ions f ...
Practice Exam 3
... ____ 16. NO2 is the brown air pollutant molecule that is produced by high temperature N2 oxidation inside an internal combustion engine. One resonance form of the molecule contains an unpaired electron plus how many single bonds, double bonds, and lone-pairs, respectively? a. 1, 1, 5 b. 2, 0, 6 c. 0 ...
... ____ 16. NO2 is the brown air pollutant molecule that is produced by high temperature N2 oxidation inside an internal combustion engine. One resonance form of the molecule contains an unpaired electron plus how many single bonds, double bonds, and lone-pairs, respectively? a. 1, 1, 5 b. 2, 0, 6 c. 0 ...
The d-block elements are commonly known as transition
... Transition metal compounds are paramagnetic when they have one or more unpaired d electrons. Some compounds are diamagnetic. These include octahedral, lowspin, d6 and square-planar d8complexes. In these cases, crystal field splitting is such that all the electrons are paired up. Ferromagnetism occu ...
... Transition metal compounds are paramagnetic when they have one or more unpaired d electrons. Some compounds are diamagnetic. These include octahedral, lowspin, d6 and square-planar d8complexes. In these cases, crystal field splitting is such that all the electrons are paired up. Ferromagnetism occu ...
Stoichiometry Review Package Answer Key
... For a visual practice on balancing equations and limiting/excess reagents go to: https://phet.colorado.edu/en/simulation/balancing-chemical-equations https://phet.colorado.edu/en/simulation/reactants-products-and-leftovers The following questions are based on the material covered so far. I will post ...
... For a visual practice on balancing equations and limiting/excess reagents go to: https://phet.colorado.edu/en/simulation/balancing-chemical-equations https://phet.colorado.edu/en/simulation/reactants-products-and-leftovers The following questions are based on the material covered so far. I will post ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.