- Aboriginal Access to Engineering
... What are the differences between physical and chemical changes? ...
... What are the differences between physical and chemical changes? ...
final exam practice test - Clayton State University
... 3. Choose the name formula pair that does not correctly match: a.) magnesium phosphate b.) iron(II) sulfite c.) silver carbonate d.) potassium fluoride e.) copper(II) bromide ...
... 3. Choose the name formula pair that does not correctly match: a.) magnesium phosphate b.) iron(II) sulfite c.) silver carbonate d.) potassium fluoride e.) copper(II) bromide ...
CHEMISTRY
... charge. There are millions of different chemical reactions that occur in the Universe. It would be quite a task to memorize the details of all of them separately. To reduce the amount that we have to know, scientists classify reactions into types. Every reaction within a type follows a particular pa ...
... charge. There are millions of different chemical reactions that occur in the Universe. It would be quite a task to memorize the details of all of them separately. To reduce the amount that we have to know, scientists classify reactions into types. Every reaction within a type follows a particular pa ...
Document
... 3. Elements Basic substances that make up matter Cannot be broken down by ordinary chemical means http://library.thinkquest.org/3616/chem/Periodic. htm 4. Common Elements in the Human Body 5. Other Elements Lesser elements Iodine (I) Iron (Fe) Trace elements Often part of enzymes or required for enz ...
... 3. Elements Basic substances that make up matter Cannot be broken down by ordinary chemical means http://library.thinkquest.org/3616/chem/Periodic. htm 4. Common Elements in the Human Body 5. Other Elements Lesser elements Iodine (I) Iron (Fe) Trace elements Often part of enzymes or required for enz ...
Balancing chemical equations notes
... chemicals are combined together and what chemicals are made when a reaction occurs. The law of conservation of mass says that matter can neither be created nor destroyed, and this requires that all chemical reactions be balanced. Consider the following balanced equation: Cu (s) + 4 HNO3 (aq) Cu(NO ...
... chemicals are combined together and what chemicals are made when a reaction occurs. The law of conservation of mass says that matter can neither be created nor destroyed, and this requires that all chemical reactions be balanced. Consider the following balanced equation: Cu (s) + 4 HNO3 (aq) Cu(NO ...
Ch. 10 – Stoichiometry Stoichiometry – relates molar ratios between
... These molar ratios are used to 'convert' between any two compounds, whether they are reactants or products. This allows us to calculate moles of reactants needed, or products produced. ...
... These molar ratios are used to 'convert' between any two compounds, whether they are reactants or products. This allows us to calculate moles of reactants needed, or products produced. ...
Chemistry: Chemical Reactions Notes STOP
... 1. If you are given a word equation with only reactants finish the word equation by writing the chemical names of the products. Remember positive ions keep the same name as their neutral element ( ...
... 1. If you are given a word equation with only reactants finish the word equation by writing the chemical names of the products. Remember positive ions keep the same name as their neutral element ( ...
Chemical Reactions
... 1. Fe2O3 + H2 Fe + H 2O Compare the number of each atom in the reactants to the 2. Fe 21 number of the same atom in the 1. O 31 product 2. H 22 Pick one of the unequal atoms 3.Fe2O3 + H2 2Fe and multiply the compound by + H 2O a number so that the atoms are Write the skeleton equation ...
... 1. Fe2O3 + H2 Fe + H 2O Compare the number of each atom in the reactants to the 2. Fe 21 number of the same atom in the 1. O 31 product 2. H 22 Pick one of the unequal atoms 3.Fe2O3 + H2 2Fe and multiply the compound by + H 2O a number so that the atoms are Write the skeleton equation ...
CHAPTER 1 Practice Exercises 1.1 12.3 g Cd 1.3 26.9814 u 1.5
... An element is a chemical species comprised of only a single type of atom. A compound is a chemical species comprised of two or more elements in a definite and unchanging proportion. A reactant is a chemical species which is transformed in a chemical reaction A chemical reaction is a process whereby ...
... An element is a chemical species comprised of only a single type of atom. A compound is a chemical species comprised of two or more elements in a definite and unchanging proportion. A reactant is a chemical species which is transformed in a chemical reaction A chemical reaction is a process whereby ...
Nature of chemical reaction - Environmental-Chemistry
... Energy and chemical reactions: • Chemical reactions are breaking of old bonds from reactant-molecules and formation of new bonds in product-molecules. • Chemical reactions involve changes in energy. Photosynthesis is an endothermic reaction. • Energy is released (exothermic) during formation of bon ...
... Energy and chemical reactions: • Chemical reactions are breaking of old bonds from reactant-molecules and formation of new bonds in product-molecules. • Chemical reactions involve changes in energy. Photosynthesis is an endothermic reaction. • Energy is released (exothermic) during formation of bon ...
chemistry i
... decreases. The equation E = hν means that as frequency increases, energy increases. Using this information and the reference tables, which color of visible light has the least energy? A. Red b. Yellow c. Green d. Violet 38. If an electron drops from n=6 to n=2, what type of electromagnetic radiation ...
... decreases. The equation E = hν means that as frequency increases, energy increases. Using this information and the reference tables, which color of visible light has the least energy? A. Red b. Yellow c. Green d. Violet 38. If an electron drops from n=6 to n=2, what type of electromagnetic radiation ...
File
... Perform these calculations showing ALL you work for extra credit! 31. Ammonia, NH3 is a typical ingredient in household cleaners. It is produced through a combination reaction involving nitrogen gas and hydrogen gas. If 10 moles of hydrogen gas reacts with an excess of nitrogen gas, how many moles o ...
... Perform these calculations showing ALL you work for extra credit! 31. Ammonia, NH3 is a typical ingredient in household cleaners. It is produced through a combination reaction involving nitrogen gas and hydrogen gas. If 10 moles of hydrogen gas reacts with an excess of nitrogen gas, how many moles o ...
Matter - Moodle
... • __________________ with other elements • _________________ __________________ into new substances Chemical properties include: ...
... • __________________ with other elements • _________________ __________________ into new substances Chemical properties include: ...
4.1 Writing and Balancing Chemical Equations
... The preceding chapter introduced the use of element symbols to represent individual atoms. When atoms gain or lose electrons to yield ions, or combine with other atoms to form molecules, their symbols are modified or combined to generate chemical formulas that appropriately represent these species. ...
... The preceding chapter introduced the use of element symbols to represent individual atoms. When atoms gain or lose electrons to yield ions, or combine with other atoms to form molecules, their symbols are modified or combined to generate chemical formulas that appropriately represent these species. ...
CHEM_2nd_Semester_Final_R eview
... 16. How do gases create pressure, use KMT to support your answer. 17. Explain diffusion, use KMT to support your answer. 18. Is Boyle’s law direct or inverse? Charles’s Law? Gay-Lussac’s Law? 19. If 735 L of a gas is at 3.11 atm and 34 oC, what is its temperature at 6.11 atm and 235 L? 20. If 12.2 m ...
... 16. How do gases create pressure, use KMT to support your answer. 17. Explain diffusion, use KMT to support your answer. 18. Is Boyle’s law direct or inverse? Charles’s Law? Gay-Lussac’s Law? 19. If 735 L of a gas is at 3.11 atm and 34 oC, what is its temperature at 6.11 atm and 235 L? 20. If 12.2 m ...
Chemistry 2nd Semester Final Exam Review Chemical Bonds Give
... 16. How do gases create pressure, use KMT to support your answer. 17. Explain diffusion, use KMT to support your answer. 18. Is Boyle’s law direct or inverse? Charles’s Law? Gay-Lussac’s Law? 19. If 735 L of a gas is at 3.11 atm and 34 oC, what is its temperature at 6.11 atm and 235 L? 20. If 12.2 m ...
... 16. How do gases create pressure, use KMT to support your answer. 17. Explain diffusion, use KMT to support your answer. 18. Is Boyle’s law direct or inverse? Charles’s Law? Gay-Lussac’s Law? 19. If 735 L of a gas is at 3.11 atm and 34 oC, what is its temperature at 6.11 atm and 235 L? 20. If 12.2 m ...
2nd Semester Final Review
... 16. How do gases create pressure, use KMT to support your answer. 17. Explain diffusion, use KMT to support your answer. 18. Is Boyle’s law direct or inverse? Charles’s Law? Gay-Lussac’s Law? 19. If 735 L of a gas is at 3.11 atm and 34 oC, what is its temperature at 6.11 atm and 235 L? 20. If 12.2 m ...
... 16. How do gases create pressure, use KMT to support your answer. 17. Explain diffusion, use KMT to support your answer. 18. Is Boyle’s law direct or inverse? Charles’s Law? Gay-Lussac’s Law? 19. If 735 L of a gas is at 3.11 atm and 34 oC, what is its temperature at 6.11 atm and 235 L? 20. If 12.2 m ...
Chemistry 1- Final Exam Review
... b. F d. I ____ 68. At constant temperature and pressure, gas volume is directly proportional to the a. molar mass of the gas. c. density of the gas at STP. b. number of moles of gas. d. pressure of the gas ____ 69. Calculate the approximate temperature of a 0.50 mol sample of gas at 750 mm Hg and a ...
... b. F d. I ____ 68. At constant temperature and pressure, gas volume is directly proportional to the a. molar mass of the gas. c. density of the gas at STP. b. number of moles of gas. d. pressure of the gas ____ 69. Calculate the approximate temperature of a 0.50 mol sample of gas at 750 mm Hg and a ...
use-2012_review_sheettest_form_c_reactions
... designation of ______________ state of matter in a chemical equation. If an ionic compound does NOT dissolve in water, it is said to be _________________ and will get the designation of ____________________ state of matter in a chemical equation. ...
... designation of ______________ state of matter in a chemical equation. If an ionic compound does NOT dissolve in water, it is said to be _________________ and will get the designation of ____________________ state of matter in a chemical equation. ...
Revision Y12 Chemistry PLC
... Part 2 – Bonding & Structure (cont.) The shapes of simple molecules and ions (g) the shapes of, and bond angles in, molecules and ions with up to six electron pairs (including lone pairs) surrounding the central atom as predicted by electron pair repulsion, including the relative repulsive strengths ...
... Part 2 – Bonding & Structure (cont.) The shapes of simple molecules and ions (g) the shapes of, and bond angles in, molecules and ions with up to six electron pairs (including lone pairs) surrounding the central atom as predicted by electron pair repulsion, including the relative repulsive strengths ...
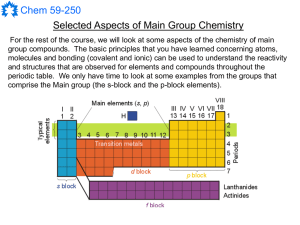
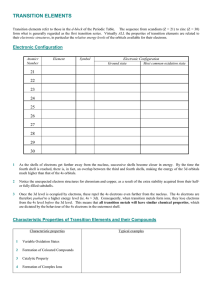
TRANSITION ELEMENTS
... Variable oxidation states of transition elements allow electron transfer between reactants and products by means of the catalyst interchanging between two oxidation states, thus facilitating an oxidation-reduction cycle. ...
... Variable oxidation states of transition elements allow electron transfer between reactants and products by means of the catalyst interchanging between two oxidation states, thus facilitating an oxidation-reduction cycle. ...
Semester Exam I Review
... 20. Chemical Change: any process determined by the atomic and molecular composition and structure of the substances involved (CGLTP) 21. Physical Change: a change from one state (solid or liquid or gas) to another without a change in chemical composition 22. Speed: distance an object travels in a ce ...
... 20. Chemical Change: any process determined by the atomic and molecular composition and structure of the substances involved (CGLTP) 21. Physical Change: a change from one state (solid or liquid or gas) to another without a change in chemical composition 22. Speed: distance an object travels in a ce ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.