Chemistry 123: Physical and Organic Chemistry
... to -10°C. Describe each step of the process and calculate the amount of energy that would need to flow in or out of the system. At each step indicate if the entropy is increasing or decreasing and under what conditions the reaction would be spontaneous. ...
... to -10°C. Describe each step of the process and calculate the amount of energy that would need to flow in or out of the system. At each step indicate if the entropy is increasing or decreasing and under what conditions the reaction would be spontaneous. ...
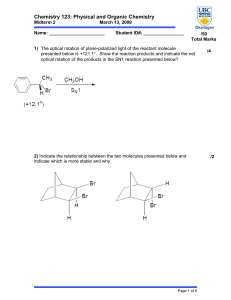
2014MSC(ORGANIC(CHEMISTRY!
... ! The!H30!molecule!happily!gives!off!one!of!the!hydrogens!due!to!the!oxygen!being!delta!plus! (partially!positively!charged).! ! The!hydrogen!is!taken!off!due!to!the!nucleophile!(the!carbons!in!the!double!bond!–!electron! rich!due!to!the!pie!orbitals!above!and!below!the!double!bond)!attacking!it!wit ...
... ! The!H30!molecule!happily!gives!off!one!of!the!hydrogens!due!to!the!oxygen!being!delta!plus! (partially!positively!charged).! ! The!hydrogen!is!taken!off!due!to!the!nucleophile!(the!carbons!in!the!double!bond!–!electron! rich!due!to!the!pie!orbitals!above!and!below!the!double!bond)!attacking!it!wit ...
Atom (A) or Ion (I)
... 84. If I have 2.5 mol of calcium carbonate in .30 L of solution, what is the molarity? 85. If I have 700.0 mL of a 5.0 M NaOH solution, how many grams of NaOH were used to make the solution? 86. What is meant by chemical equilibrium? 87. What factors affect the rate of a reaction? 88. What does a ca ...
... 84. If I have 2.5 mol of calcium carbonate in .30 L of solution, what is the molarity? 85. If I have 700.0 mL of a 5.0 M NaOH solution, how many grams of NaOH were used to make the solution? 86. What is meant by chemical equilibrium? 87. What factors affect the rate of a reaction? 88. What does a ca ...
E/F Physical Science
... 1. Is the following sentence true or false? The new substances formed as a result of a chemical reaction are called products. 2. Circle the letter of each sentence that is correct for the chemical equation: C + O2 → CO2. a. Carbon and oxygen react and form carbon monoxide. b. Carbon and oxygen react ...
... 1. Is the following sentence true or false? The new substances formed as a result of a chemical reaction are called products. 2. Circle the letter of each sentence that is correct for the chemical equation: C + O2 → CO2. a. Carbon and oxygen react and form carbon monoxide. b. Carbon and oxygen react ...
Chemical Reactions - We can`t sign you in
... Ammonia is a base that doesn’t contain a metal. Some bases are soluble in water – they are called alkalis. ...
... Ammonia is a base that doesn’t contain a metal. Some bases are soluble in water – they are called alkalis. ...
File
... An element is a pure substance which cannot be split up into two or more simpler substances by physical or chemical means. ...
... An element is a pure substance which cannot be split up into two or more simpler substances by physical or chemical means. ...
Oxidation-Reduction Reactions - An Introduction to Chemistry
... Electrons are rarely found unattached to atoms. Thus, for one element or compound to lose electrons and be oxidized, another element or compound must be there to gain the electrons and be reduced. In other words, oxidation (loss of electrons) must be accompanied by reduction (gain of electrons). In ...
... Electrons are rarely found unattached to atoms. Thus, for one element or compound to lose electrons and be oxidized, another element or compound must be there to gain the electrons and be reduced. In other words, oxidation (loss of electrons) must be accompanied by reduction (gain of electrons). In ...
Solutions
... ‣ Atoms that gain electrons (negative charges) are reduced. ‣ Atoms that loose electrons are oxidized. ‣ Electrons always end up somewhere. If something in the reaction is getting oxidized, something else is getting reduced. ‣ Red-ox processes are not an equilibrium processes — someone wins; someon ...
... ‣ Atoms that gain electrons (negative charges) are reduced. ‣ Atoms that loose electrons are oxidized. ‣ Electrons always end up somewhere. If something in the reaction is getting oxidized, something else is getting reduced. ‣ Red-ox processes are not an equilibrium processes — someone wins; someon ...
chem final review
... 30) In which of the following sets are the symbol of the element, the number of protons, and the number of electrons given correctly? A) In, 49 protons, 49 electrons B) Cs, 55 protons, 132.9 electrons C) Zn, 30 protons, 60 electrons D) He, 4 protons, 4 electrons E) F, 19 protons, 19 electrons 31) Co ...
... 30) In which of the following sets are the symbol of the element, the number of protons, and the number of electrons given correctly? A) In, 49 protons, 49 electrons B) Cs, 55 protons, 132.9 electrons C) Zn, 30 protons, 60 electrons D) He, 4 protons, 4 electrons E) F, 19 protons, 19 electrons 31) Co ...
Key concepts of chemistry from high school chemistry
... that share the same number of protons yet differ in number of neutrons are termed isotopes. Isotopes of an element have very similar properties since they differ only in the number of neutr ...
... that share the same number of protons yet differ in number of neutrons are termed isotopes. Isotopes of an element have very similar properties since they differ only in the number of neutr ...
Sec 5.8 - 5.11 notes
... 11) The salt bridge can contain any _ELECTROLYTES________. 12) The anode will __LOSE_____(gains/loses) mass as it is _OXIDIZED___(oxidized/reduced). 13) The cathode will _GAIN____ mass as it is __REDUCED_____(oxidized/reduced). ...
... 11) The salt bridge can contain any _ELECTROLYTES________. 12) The anode will __LOSE_____(gains/loses) mass as it is _OXIDIZED___(oxidized/reduced). 13) The cathode will _GAIN____ mass as it is __REDUCED_____(oxidized/reduced). ...
Practice problems for chapter 1, 3 and 5 1) A small amount of salt
... 23) There are __________ protons, __________ neutrons, and __________ electrons in 131I-. A) 131, 53, and 54 B) 131, 53, and 52 C) 53, 78, and 54 D) 53, 131, and 52 E) 78, 53, and 72 24) Which of the following compounds would you expect to be ionic? A) SF6 B) H2O C) H2O2 D) NH3 E) CaO 25) Which pair ...
... 23) There are __________ protons, __________ neutrons, and __________ electrons in 131I-. A) 131, 53, and 54 B) 131, 53, and 52 C) 53, 78, and 54 D) 53, 131, and 52 E) 78, 53, and 72 24) Which of the following compounds would you expect to be ionic? A) SF6 B) H2O C) H2O2 D) NH3 E) CaO 25) Which pair ...
unit 7 – writing and balancing chemical equations
... (1) Write a correct formula for each of the reactants. Put a plus sign between the reactants to separate them. Note: A plus sign means “added to” or “reacts with”. (2) Draw a yield arrow (3) Write a correct formula for each of the products, putting a plus sign between them also. Note: On the product ...
... (1) Write a correct formula for each of the reactants. Put a plus sign between the reactants to separate them. Note: A plus sign means “added to” or “reacts with”. (2) Draw a yield arrow (3) Write a correct formula for each of the products, putting a plus sign between them also. Note: On the product ...
Chapter 4
... Copper wire reacts with silver nitrate to form silver metal. What is the oxidizing agent in the reaction? ...
... Copper wire reacts with silver nitrate to form silver metal. What is the oxidizing agent in the reaction? ...
Chapter 2
... reactants must be accounted for in the products. • The reactions must be “balanced”. ...
... reactants must be accounted for in the products. • The reactions must be “balanced”. ...
Catalytic Synthesis of Organophosphorus Compounds from
... catalysed by different transition metal (PdII, HgII, PtII, PtIV, AgI, FeIII, CuII) salts leading to the formation of valuable organophosphorus derivatives (trialkylphosphates, dialkylphosphites, trialkylphoshinoxides, phosphonium salts) are reviewed. The principal features of the oxidative phosphory ...
... catalysed by different transition metal (PdII, HgII, PtII, PtIV, AgI, FeIII, CuII) salts leading to the formation of valuable organophosphorus derivatives (trialkylphosphates, dialkylphosphites, trialkylphoshinoxides, phosphonium salts) are reviewed. The principal features of the oxidative phosphory ...
PERIODIC TABLE OF THE ELEMENTS
... • Transition metals are usually shiny. – E.g. silver (Ag), gold (Au), and platinum (Pt) ...
... • Transition metals are usually shiny. – E.g. silver (Ag), gold (Au), and platinum (Pt) ...
standard sample test
... 13. In the reaction of iron with nitric acid to form iron (II) nitrate and hydrogen gas (H2), if 1 mole of iron were to be reacted with 1 mole of nitric acid, how many moles of iron (II) nitrate would form? (a) 0.5 mole (b) 1 mole (c) 1.5 mole ...
... 13. In the reaction of iron with nitric acid to form iron (II) nitrate and hydrogen gas (H2), if 1 mole of iron were to be reacted with 1 mole of nitric acid, how many moles of iron (II) nitrate would form? (a) 0.5 mole (b) 1 mole (c) 1.5 mole ...
9182747 Chemistry Ja02
... If you wish to change an answer, erase your first penciled circle and then circle with pencil the number of the answer you want. After you have completed the examination and you have decided that all of the circled answers represent your best judgment, signal a proctor and turn in all examination ma ...
... If you wish to change an answer, erase your first penciled circle and then circle with pencil the number of the answer you want. After you have completed the examination and you have decided that all of the circled answers represent your best judgment, signal a proctor and turn in all examination ma ...
National 5 Chemistry Unit 3 Chemistry In Society
... The purpose of the ion bridge is to complete the circuit of this cell. The ion bridge (sometimes called a salt bridge) contains an electrolyte which takes no part in the cell reactions. The chemical reactions in cells like these are displacement reactions. Zinc can displace copper from copper(II) su ...
... The purpose of the ion bridge is to complete the circuit of this cell. The ion bridge (sometimes called a salt bridge) contains an electrolyte which takes no part in the cell reactions. The chemical reactions in cells like these are displacement reactions. Zinc can displace copper from copper(II) su ...
Metals Minitest
... Redox reactions are reactions where both oxidation and reduction are taking place. Displacement reactions are examples of redox reactions as one species is being oxidised (losing electrons) while the other is being reduced (gaining electrons). For example, if magnesium was added to copper sulphate s ...
... Redox reactions are reactions where both oxidation and reduction are taking place. Displacement reactions are examples of redox reactions as one species is being oxidised (losing electrons) while the other is being reduced (gaining electrons). For example, if magnesium was added to copper sulphate s ...
Chapter 20 Electrochemistry
... Cr2O72(aq) + 14 H+(aq) + 6 I(aq) 2 Cr3+(aq) + 3 I2(s) + 7 H2O(l) is spontaneous. A solution containing K2Cr2O7 and H2SO4 is poured into one beaker, and a solution of KI is poured into another. A salt bridge is used to join the beakers. A metallic conductor that will not react with either solutio ...
... Cr2O72(aq) + 14 H+(aq) + 6 I(aq) 2 Cr3+(aq) + 3 I2(s) + 7 H2O(l) is spontaneous. A solution containing K2Cr2O7 and H2SO4 is poured into one beaker, and a solution of KI is poured into another. A salt bridge is used to join the beakers. A metallic conductor that will not react with either solutio ...
CHEMISTRY: Practice Spring Final
... Note: Do not JUST study this practice exam; it does not contain every topic that may appear on your final exam. Be sure to look at your review guide to see a list of topics you are responsible for. Also, this practice test is broken up by topic; your final exam will not be. CHEMICAL REACTIONS 1) Cla ...
... Note: Do not JUST study this practice exam; it does not contain every topic that may appear on your final exam. Be sure to look at your review guide to see a list of topics you are responsible for. Also, this practice test is broken up by topic; your final exam will not be. CHEMICAL REACTIONS 1) Cla ...
Redox

Redox reactions include all chemical reactions in which atoms have their oxidation state changed; in general, redox reactions involve the transfer of electrons between species. The term ""redox"" comes from two concepts involved with electron transfer: reduction and oxidation. It can be explained in simple terms: Oxidation is the loss of electrons or an increase in oxidation state by a molecule, atom, or ion. Reduction is the gain of electrons or a decrease in oxidation state by a molecule, atom, or ion.Although oxidation reactions are commonly associated with the formation of oxides from oxygen molecules, these are only specific examples of a more general concept of reactions involving electron transfer.Redox reactions, or oxidation-reduction reactions, have a number of similarities to acid–base reactions. Like acid–base reactions, redox reactions are a matched set, that is, there cannot be an oxidation reaction without a reduction reaction happening simultaneously. The oxidation alone and the reduction alone are each called a half-reaction, because two half-reactions always occur together to form a whole reaction. When writing half-reactions, the gained or lost electrons are typically included explicitly in order that the half-reaction be balanced with respect to electric charge.Though sufficient for many purposes, these descriptions are not precisely correct. Oxidation and reduction properly refer to a change in oxidation state — the actual transfer of electrons may never occur. The oxidation state of an atom is the fictitious charge that an atom would have if all bonds between atoms of different elements were 100% ionic. Thus, oxidation is better defined as an increase in oxidation state, and reduction as a decrease in oxidation state. In practice, the transfer of electrons will always cause a change in oxidation state, but there are many reactions that are classed as ""redox"" even though no electron transfer occurs (such as those involving covalent bonds).There are simple redox processes, such as the oxidation of carbon to yield carbon dioxide (CO2) or the reduction of carbon by hydrogen to yield methane (CH4), and more complex processes such as the oxidation of glucose (C6H12O6) in the human body through a series of complex electron transfer processes.